Abstract
STAT3 is an important molecule in Janus kinase (JAK) signal transducer and activator of transcription (STAT) signal pathway, and facilitates expression of various oncogenic genes such as Bcl-2, thus is correlated with tumor onset, progression and drug resistance. MiR-29a down-regulation is associated with the pathogenesis of nasopharyngeal carcinoma. Bioinformatics analysis demonstrated a complementary binding between miR-29a and 3’-UTR of STAT3. This study aims to investigate the role of miR-29a in regulating STAT3, as well as in Taxol resistance of nasopharyngeal carcinoma CNE-1 cells. Dual luciferase reporter gene assay showed a regulatory relationship between miR-29a and STAT3. Rhodamine 123 repository in CNE-1 and CNE1/Taxol drug resistant cells was measured together with the expression of miR-29a, STAT3, and p-STAT3. Flow cytometry was used to measure cell apoptosis and PCNA expression under Taxol treatment. CNE-1/Taxol cells were treated with miR-29a mimic and or si-STAT3, followed by measuring the expression of miR-29a, STAT3, and p-STAT3 and cell apoptosis. CCK-8 assay was performed to evaluate cell proliferation. MiR-29a inhibited STAT3 expression. Significantly lower Rhodamine 123 repository, miR-29a expression and apoptosis and higher expression of STAT3, p-STAT3 and PCNA were observed in CNE-2/ Taxol cells than those in CNE-1 cells. Transfection of miR-29a mimic and/or si-STAT3 decreased STAT3, p-STAT3 and PCNA expression, inhibited proliferation and promoted cell apoptosis. MiR-29a down-regulation is correlated with drug resistance of nasopharyngeal carcinoma cell line CNE-1 and MiR-29a up-regulation decreases Taxol resistance of nasopharyngeal carcinoma CNE-1 cells possibly via inhibiting STAT3 and Bcl-2 expression.
Introduction
Nasopharyngeal carcinoma (NPC) is an epithelial derived malignant tumor which is frequently occurred in nasopharyngeal cavity upper and lateral walls. As a common malignant tumor in Southeastern Asia and Southern China, NPC has a relatively higher incidence [1]. Currently, radiotherapy is often used to treat NPC in clinic. Combined chemotherapy helps to improve treatment efficacy and prognosis for those NPC patients with recurrence or metastasis. However, some patients present drug resistance for chemotherapy drugs, mainly contributing to the failure of chemotherapy and mortality [2].
In Janus kinase (JAK)-signal transducer and activator of transcription (STAT) signal transduction pathway, STAT protein family consists of 7 members, in which STAT3 and STAT5 are closely associated with human tumor pathogenesis, especially for STAT3 [3]. Potential expression and activity of STAT3 is closely correlated with the onset, progression, invasion and metastasis of breast cancer [4], colon cancer [5] and gastric carcinoma [6], and affects patient survival and prognosis. The role of STAT3 in chemotherapy resistance of lung cancer [7], lymphoma [8] and prostate cancer [9] has also been reported.
MicroRNA is a family of endogenous non-coding small molecular single stranded RNA with a length of 22
Materials and methods
Major reagents and materials
Human NPC cell line CNE-1 and immortalized nasopharyngeal epithelial cell line NP69 were purchased from Xinyu Biotech (Zhangjiakou, Hebei, China). DMEM and 1640 medium, streptomycin-penicillin and fetal bovine serum (FBS) were purchased from Gibco BRL. Co. Ltd. (Grand Island, NY, USA). RNA extraction kit Rneasy MiNi Kit was purchased from Qiagen (Hilden, Germany). FuGENE HD Transfection Reagent was purchased from Roche Pharma (Basel, Switzerland). PrimerScript™ RT reagent Kit and SYBR Green dye were purchased from Takara (Dalian, Liaoning, China). MiR-29a mimic and miR-NC were synthesized by Ruibo Bio. Technol. Co. Ltd. (Guangzhou, Guangdong, China). Mouse anti-human STAT3, p-STAT3, Bcl-2 and
Cell culture
CNE-1 and NP69 cells were cultured in 1640 medium containing 10% FBS and 1% streptomycin in a 37
Establishment of CNE-1/Taxol model and drug resistance index
CNE-1/Taxol cell line was established by gradient induction. In brief, cells at log-growth phase were treated with 0.1
CNE-1/Taxol and CNE-1 cells were treated with 0, 0.25, 0.5, 1, 2.5, 5, 10, and 20
Measurement of PCNA expression
CNE-1/Taxol and CNE-1 cells were cultured in Taxol with IC
Rhodamine 123 refusal staining assay
CNE-1/Taxol and CNE-1 cells were re-suspended in 1640 medium containing 0.1% Rhodamine solution. After incubation at 37
Dual luciferase activity assay
Full length or mutant fragment of 3’-UTR of STAT3 gene was amplified by PCR, digested by SacI and XbaI endo-nuclease, and sub-cloned into pLUC dual luciferase gene reporter plasmid, which was named as pLUC-STAT3-wt. Luciferase reporter vector containing mutant form of the 3’-UTR of STAT3 gene was also constructed as pLUC-STAT3-mut. FuGENE HD Transfection Reagent was used to transfect pLUC-STAT3-wt (or pLUC-STAT3-mut) and miR-29a mimic into HEK293T cells. After 48 h, dual luciferase activity was measured by a commercial kit according to the manufacturer’s instructions.
Cell transfection and grouping
qRT-PCR for gene expression
PrimeScript™ RT reagent kit was used to synthesize cDNA by reverse transcription using RNA which was extracted using Rneasy MiNi Kit. Using cDNA as the template, PCR amplification was performed. Primer sequences were designed as follows: miR-29aP
Western blot
RIPA lysis buffer was used to extract protein, followed by examination of the quality and concentration. Forty
Analysis of cell apoptosis
Cells were collected and digested by trypsin, and re-suspended in Binding Buffer. Five
Cell proliferation analysis
Cells from all groups were cultured in 1640 medium containing 2.0
Statistical analysis
SPSS18.0 was used for data analysis. Measurement data were presented as mean
Results
MiR-29a inhibits STAT3 expression
Bioinformatics analysis showed a complementary binding relationship between miR-29a and the 3’-UTR of STAT3 mRNA (Fig. 1A). Dual luciferase gene reporter assay showed that transfection of miR-29a mimic significantly decreased the relative luciferase activity in HEK293T cells (Fig. 1B), indicating that miR-29a targeted the 3’-UTR of STAT3 mRNA to suppress its expression.
MiR-29a inhibited STAT3 expression. (A) Binding sites between miR-29a and 3’-UTR of STAT3 mRNA; (B) Dual luciferase gene reporter assay. *, 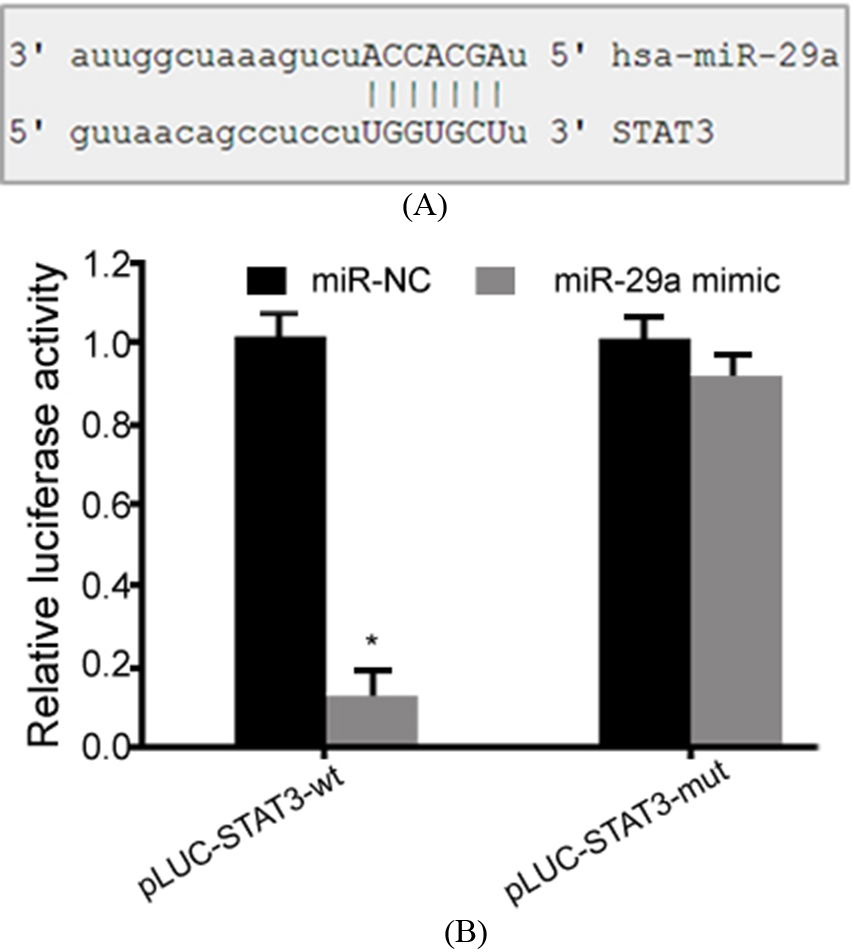
qRT-PCR analysis showed significantly lower miR-29a expression in CNE-1 cells compared to NP96 cells, whilst STAT3 mRNA level was elevated (Fig. 2A). Comparing to CNE-1 cells, CNE-1/Taxol cells had significantly lower miR-29a expression and higher STAT3 mRNA expression (Fig. 2A). Consistently, western blot analysis showed significantly increased STAT3 and p-STAT3 expression in CNE-1/Taxol cells compared to those in CNE-1 cells, which had higher STAT3 and p-STAT3 expression than NP96 cells (Fig. 2B). Rhodamine 123 repository assay showed more dyes in CNE-1 cells and lower repository in CNE-1/Taxol cells, indicating refusal of staining (Fig. 2C). Flow cytometry demonstrated that under the treatment of equal concentration of Taxol, CNE-1/Taxol cells had higher PCNA expression than CNE-1 cells (Fig. 2D), but with lower cell apoptotic rate (Fig. 2E).
MiR-29a up-regulation and STAT3 down-regulation reduces drug resistance of CNE-1/Taxol cells
Next, we investigated the changes of drug resistance in CNE-1/Taxol cells with miR-29a up-regulation and/or STAT3 down-regulation under the treatment of 2.0
MiR-29a down-regulation was involved in CNE-1 cell drug resistance. (A) qRT-PCR for gene expression; (B) Western blot for protein expression; (C) Rhodamine 123 refusal staining assay; (D) Flow cytometry for PCNA expression; (E) Flow cytometry for cell apoptosis. *, 
MiR-29a up-regulation and STAT3 down-regulation reduced drug resistance of CNE-1/Taxol cells. (A) qRT-PCR for gene expression; (B) Western blot for protein expression; (C) CCK-8 assay for cell proliferation activity; (D) Flow cytometry for cell apoptosis. a, 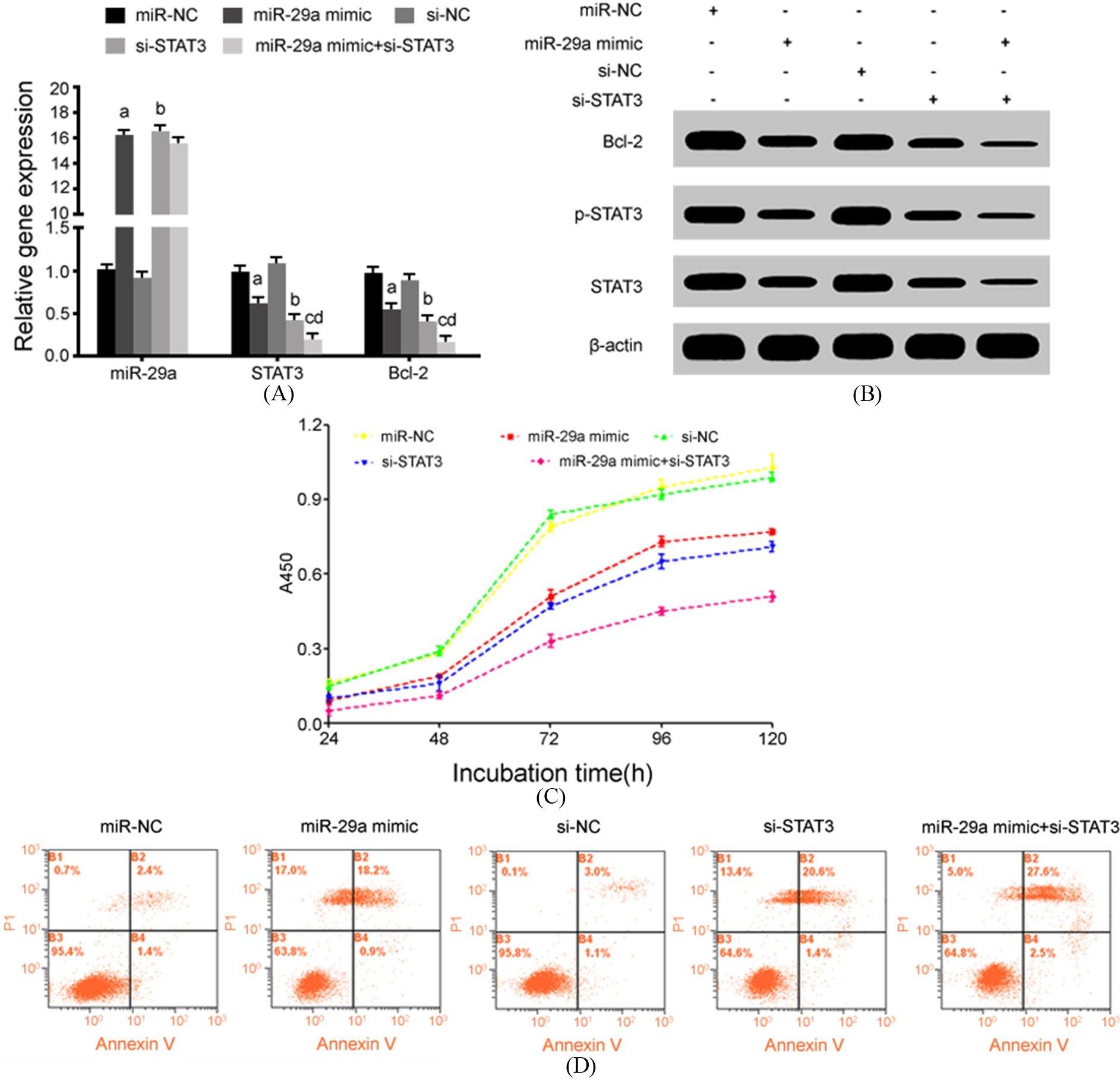
JAK-STAT signal pathway integrates multiple extra-cellular signal pathways including cytokines, growth factor and mitogen, and plays an important role in regulating cell growth, mitosis, proliferation or apoptosis [3]. STAT transcription factor protein family consists of 7 members, in which STAT3 and STAT5 are closely associated with human tumor occurrence, especially for STAT3. Under the activation of JAK-STAT3 signal pathway, dimerization of membrane receptor can phosphorylate JAK kinase, which then phosphorylates the receptor, leading to recruitment of STAT3 onto the membrane receptor and subsequent phosphorylation of STAT3 under JAK direction. Phosphorylated and activated STAT3 detaches from the receptor, resulting in dimerization and subsequent transportation from cytoplasm into the nucleus, where it exerts functions on specific DNA fragment to mediate gene expression. Enhanced cytoplasm and activity of STAT3 can potentiate transcription and expression of various oncogenic-like genes including Bcl-2, Bcl-xl, and is closely associated with the onset, progression, invasion and metastasis of multiple tumors including breast cancer [4], colon cancer [5] and gastric carcinoma [6], leading to shortened overall survival and poor prognosis. Moreover, elevated expression and activity of STAT3 is also an important factor endowing drug resistance for tumor cells [9].
Recently, a study illustrated the close correlation between miR-29a down-regulation and the occurrence, progression and prognosis of NPC [11, 12]. In addition, various studies have indicated critical roles of miR-29a expression in mediating drug resistance of tumor cells [13, 14]. Bioinformatics analysis revealed a complementary binding association between miR-29a and 3’-UTR of STAT3 mRNA, indicating a possible regulatory relationship. Taxol is a tetracyclic diterpene compound extracted from Taxus brevifolia. As a cell cycle specific drug, it can inhibit denature of micro-tubulin and spindle fiber formation during mitosis, thus suppressing cell proliferation and facilitating apoptosis. As a cytotoxic drug, Taxol has been used for treating various tumors including ovarian cancer [15], esophageal squamous carcinoma [16] and breast cancer [17]. Therefore, this study established a Taxol-resistant cell model by drug induction, to investigate the role of miR-29a in mediating STAT3 expression as well as in Taxol drug resistance of NPC.
In this study, we showed that compared to NP96 cells, miR-29a expression in NPC cell line CNE-1 was significantly decreased, accompanied with elevated STAT3 and p-STAT3 expression, indicating that NPC cells had potentiated phosphorylation of STAT3, plus it up-regulation, in which miR-29a down-regulation might play a role in enhancing STAT3 and p-STAT3 expression as well as in facilitating NPC pathogenesis. Rhodamine 123 staining showed more repository in CNE-1 cells compared to CNE-1/Taxol cells, suggesting drug resistant mechanism involving exclusion of drugs. In addition, IC50 value of CNE-1/Taxol cells (14.57
In this study, up-regulation of miR-29a inhibited drug resistance of tumor cells, consistent with Yu et al. [14]. Xu et al. showed significantly reduced NPC C666-1 cell proliferation, migration and invasion after STAT3 inhibition, plus potentiated cell apoptosis [18]. Tsang et al. showed that after drug intervention on STAT3 activity,
There are some limitations in the present study. Firstly, the other NPC cells with low expression of miR-29a and high expression of STAT3 have not been investigated in this study, which should be investigated in further studies to provide more constructive conclusion regarding the role of miR-29a and STAT3 in the pathogenesis of NPC. Secondly, the present study only knockdown the STAT3 gene by siRNA, however, the effects of siRNA mediated gene knockdown is temporary [22]. Therefore, knockout of STAT3 should be performed in the following study to confirm the role of STAT3. Thirdly, as almost all cells in the
Conclusion
MiR-29a down-regulation is correlated with NPC cell line CNE-1 drug resistance. Up-regulation of miR-29a decreases Taxol drug resistance of CNE-1 cells possibly via inhibition of STAT3 and Bcl-2 expression.
Footnotes
Acknowledgments
This work was supported by the National Natural Science Foundation of China (No. 81472988).
Conflict of interest
None.
