Abstract
BACKGROUND AND AIM:
Hypercalcemia is a potentially fatal and not rare complication of hepatocellular carcinoma (HCC), and its underlying mechanism remains unclear. Parathyroid hormone (PTH) is the most important regulator of the concentrations of calcium and phosphate in blood; parathyroid hormone-related protein (PTHrP) was the most frequent cause of humoral hypercalcemia of malignancy; parathyroid hormone 1 receptor (PTH1R) is the common receptor for PTH and PTHrP. The aim of this study is to investigate the expression of PTH, PTHrP, and PTH1R in HCC tissues, and their relationship with clinical pathological characters in HCC.
METHODS:
First, a meta-analysis based on online Oncomine Expression Array database was conducted to compare the different mRNA expression of PTH1R, PTH and PTHrP between hepatocellular carcinoma and normal tissues. Then, the protein expression level of differentially expressed gene was examined by immunohistochemistry staining in 223 HCC tissues and 102 non-tumorous liver tissues controls. The relationship between the protein expression and clinicopathological parameters was analyzed by
RESULTS:
PTH1R mRNA expression was significantly lower in HCC tissues compared with normal tissues, while the expression of PTH and PTHrP showed no significant difference between HCC tissues and normal tissues. High PTH1R protein expression was found in 90/102 cases of adjacent non-tumorous liver tissues, and in 91 of 223 cases of HCC tissues. PTH1R expression was significantly related to tumor size, Edmondson Grade, AFP, and overall survival.
CONCLUSIONS:
PTH1R may be the major cause of hypercalcemia in HCC, and the decreased PTH1R expression was a poor prognosis in HCC.
Introduction
G-protein coupled receptors (GPCRs) constitute a large protein family of receptors. Based on sequence homology and functional similarity, GPCRs can be grouped into 6 classes: Class A (Rhodopsin- like), Class B (Secretin receptor family), Class C (Metabotropic glutamate/pheromone), Class D (Fungal mating pheromone receptors), Class E (Cyclic AMP receptors), and Class F (Frizzled/Smoothened). GPCRs are involved in and regulate various physiological functions [1, 2, 3]. Dysfunction of GPCRs may lead to the occurrence and development of many diseases. Also, GPCRs are the targets of approximately 40% of all modern medicinal drugs [4, 5].
Parathyroid hormone 1 receptor (PTH1R), belongs to the class B G-protein coupled receptor family, is the common receptor for parathyroid hormone (PTH) and parathyroid hormone-related protein (PTHrP). PTH is the most important regulator of the concentrations of calcium and phosphate in blood. It is mainly produced by the parathyroid glands, as a 115-amino acid precursor (pre-pro-PTH), and cleaved to pro-PTH with a prosequence of 6 amino acids, and then to the 84-amino acid mature PTH [6, 7]. Mature PTH is then secreted into blood and acts primarily on kidney and bone, promotes osteoclastic bone resorption and renal tubular calcium reabsorption, and inhibits renal tubular phosphate reabsorption [8, 9]. The biological actions of PTH are mediated by binding of its N-terminal region (aa 1–34) to PTH1R, which was highest expressed in kidney and bone [7, 10]. PTHrP, also called parathyroid hormone like hormone (PTHLH), was first identified from tumors associated with the syndrome of humoral hypercalcemia of malignancy, and considered as the most frequent cause of humoral hypercalcemia of malignancy [11, 12]. PTHrP was also produced by non-hypercalcemic tumors, and responsible for the osteolysis [13, 14, 15]. Sequence analysis showed that PTHrP was significant sequence homology within the first 13 amino acid residues to PTH, and bind to the same receptor PTH1R [16]. Further studies showed that PTHrP was not confined in tumors; it was also widely expressed in normal tissues and with multiple biological activities [17, 18]. PTH1R was the common receptor of PTH and PTHrP. When PTH1R bind with PTH or PTHrP, it was internalize activated, and subsequently activated the adenylyl cyclase/protein kinase A (AC/PKA) pathway and the phospholipase C/protein kinase C (PLC/PKC) pathway [19, 20, 21]. Defects in PTH1R was the cause of chondrodysplasia and primary failure of eruption [22, 23], while over-expression of PTH1R was found in osteosarcoma and breast cancer, and conferred an aggressive phenotype [24, 25].
Hypercalcemia, hypophosphataemia, bone thinning and fractures, are the common complications of chronic liver disease [26], while the mechanism was still not well understood. In this study, we first performed a meta-analysis on Oncomine Expression Array database to evaluate the expression of PTH1R, PTH and PTHrP, the three most important regulators of calcium and phosphate, and found only expression of PTH1R was significantly reduced in HCC tissues when compared with normal liver tissues; then, we examined PTH1R expression in 223 hepatocellular carcinoma (HCC) tissues and 102 non-tumorous liver tissues by immunohistochemistry staining, and investigated the relationships between PTH1R expression and clinicopathological parameters, and the overall survival rate. This study might provide further insight into the cause of bone thinning and fractures, hypercalcemia and hypophosphataemia in HCC cancers.
Materials and methods
Oncomine database analysis
A meta-analysis based on online Oncomine Expression Array database (
Patients and tissue samples
All the human tissues were obtained from surgical resection specimens of HCC patients at Zhejiang Provincial People’s Hospital, Hangzhou, China. This study was approved by the Ethics Committee of Zhejiang Provincial People’s Hospital, Hangzhou, China. Written informed consent was provided by the patients.
Expression of PTH1R in normal liver and hepatocellular carcinoma tissues based on Oncomine database.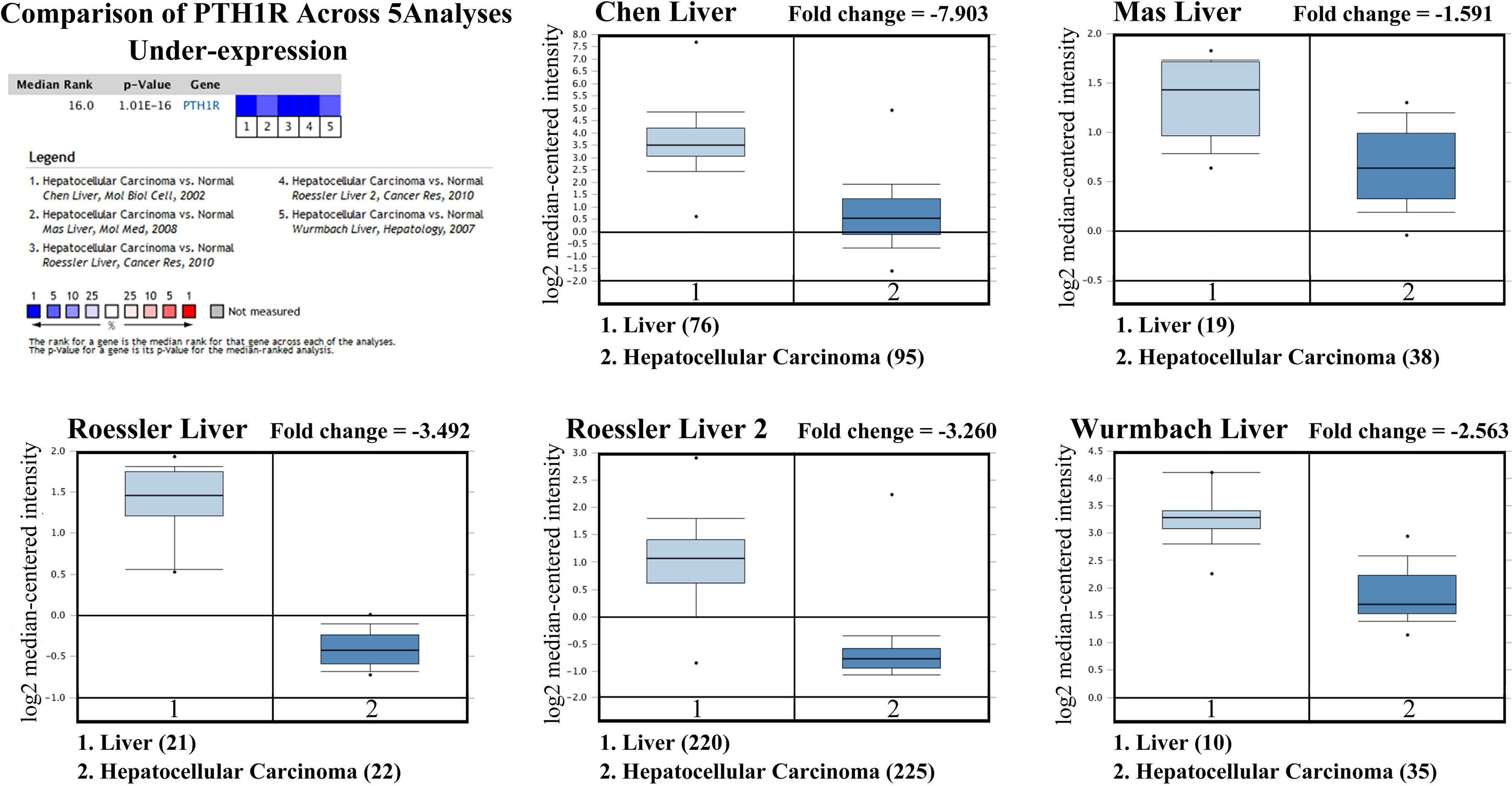
Two hundred and twenty-three cases of paraffin-embedded HCC tissues and 102 non-tumorous liver tissues were obtained from April 2008 to September 2014. The patient cohort consisted of 177 males and 46 females, with a median age of 58.15 years (range: 25–90) at the time of surgery. The survival time was calculated from the date of surgery to the follow-up deadline or the date of death. All tissues were used for tissue microarray (TMA), constructed by Shanghai Biochip Co., Ltd, Shanghai, China.
Immunohistochemical staining was performed using Histostain-Plus IHC Kit (Invetrogen, USA), according to the manufacturer’s instructions. Briefly, 5
The immunohistochemical stain of PTH1R was scored independently by two pathologists, based on the intensity and the proportion of positively stained cells. Staining intensity was evaluated with a four-tiered grading system: 0
Statistical analysis
Statistical analysis was performed using Statistical Program for Social Sciences (SPSS) software 13.0 (SPSS Inc., Chicago, IL, USA). The
Expression of PTH in normal liver and hepatocellular carcinoma tissues based on Oncomine database.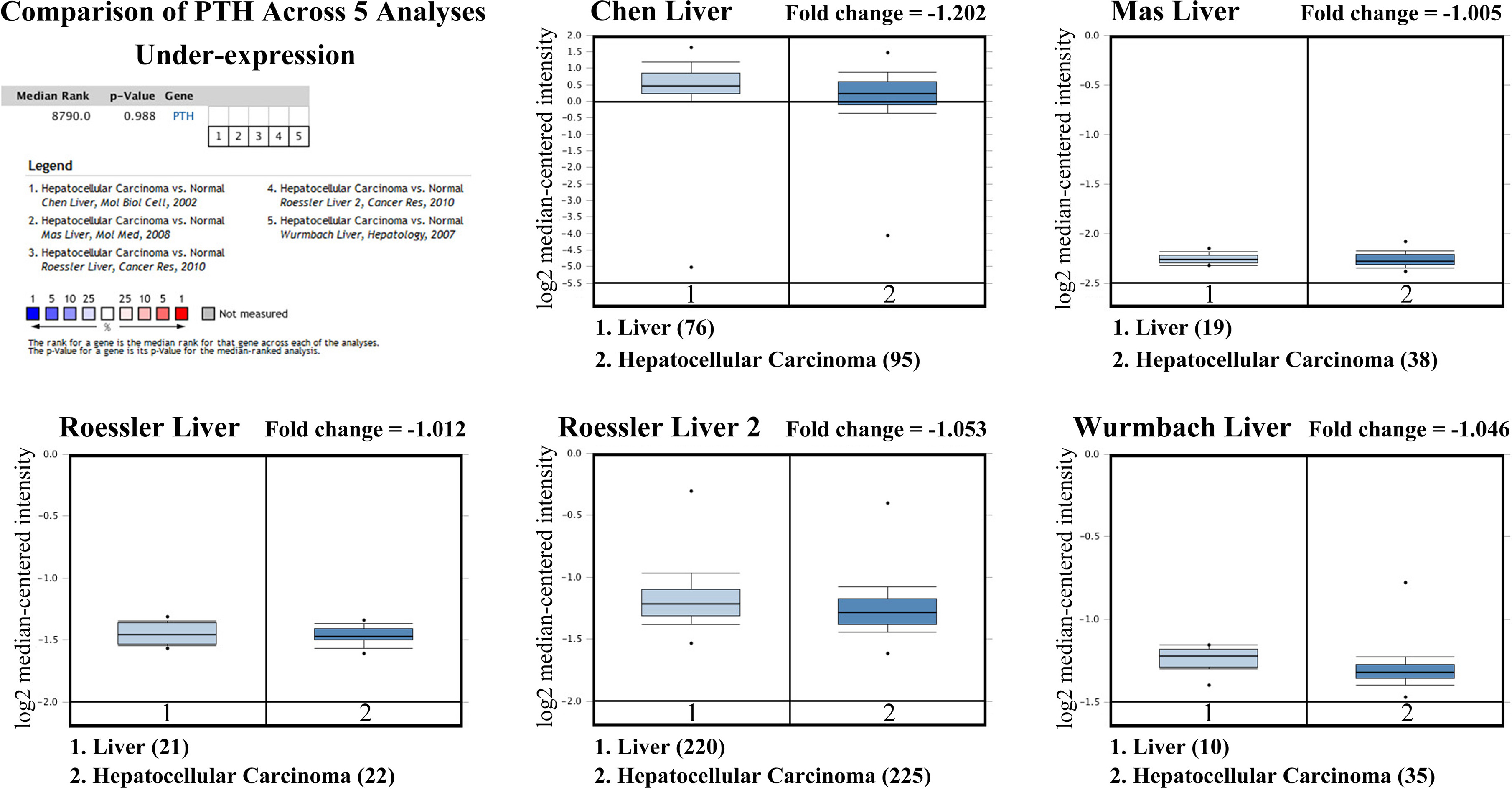
Expression of PTHLH (PTHrP) in normal liver and hepatocellular carcinoma tissues based on Oncomine database.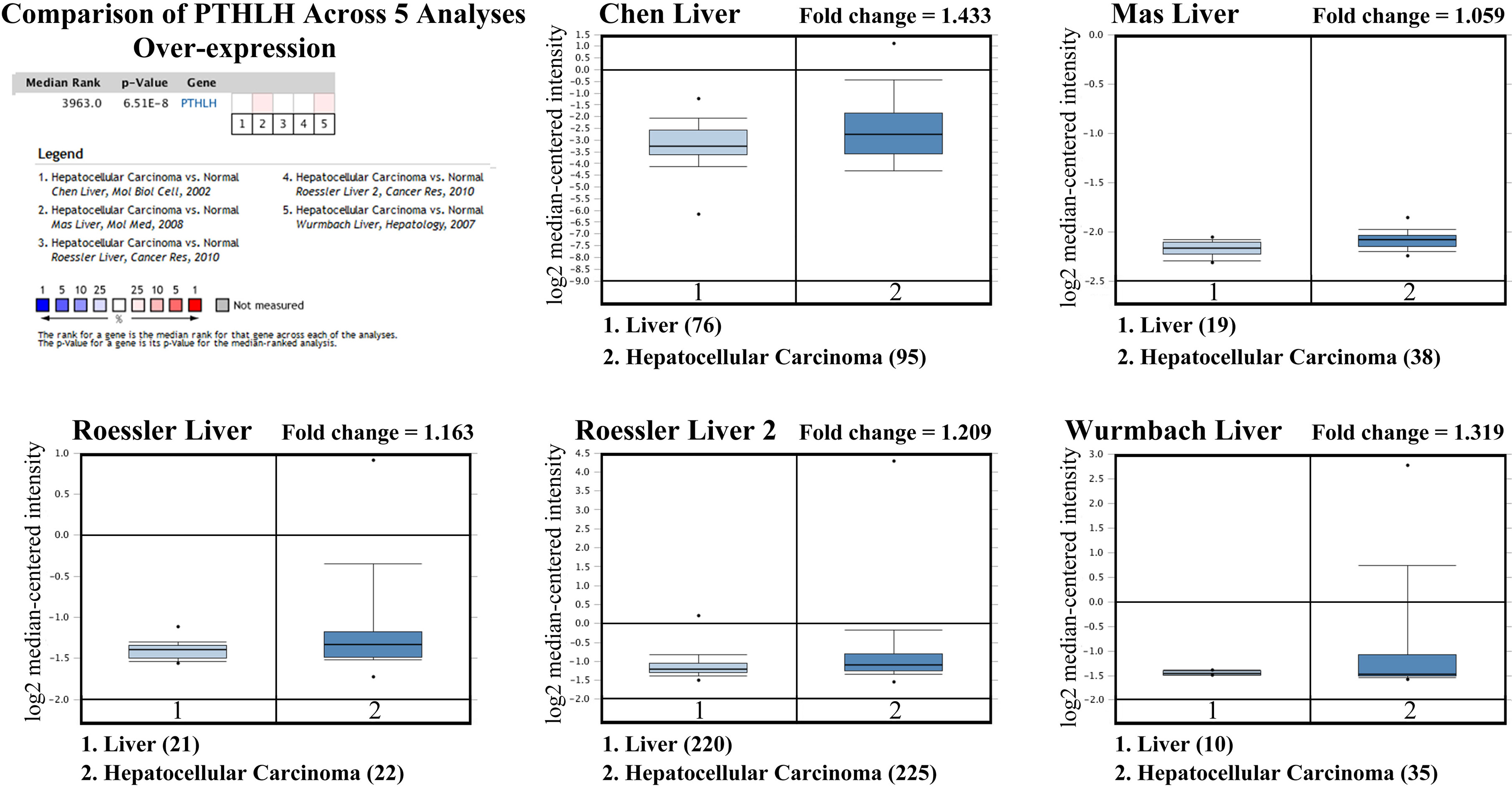
Analysis of PTH1R, PTH, PTHrP expression based on Oncomine database
Firstly, we analyzed the Oncomine databases to compare PTH1R, PTH, PTHrP mRNA expression in hepatocellular carcinoma vs. normal tissues, and found that PTH1R mRNA expression was significantly lower in HCC tissues compared with normal tissues (Fig. 1, all
Immunohistochemical staining of PTH1R in non-tumorous liver tissues. A: normal liver tissue; B: Inflammatory tissues; C: Cirrhosis tissues.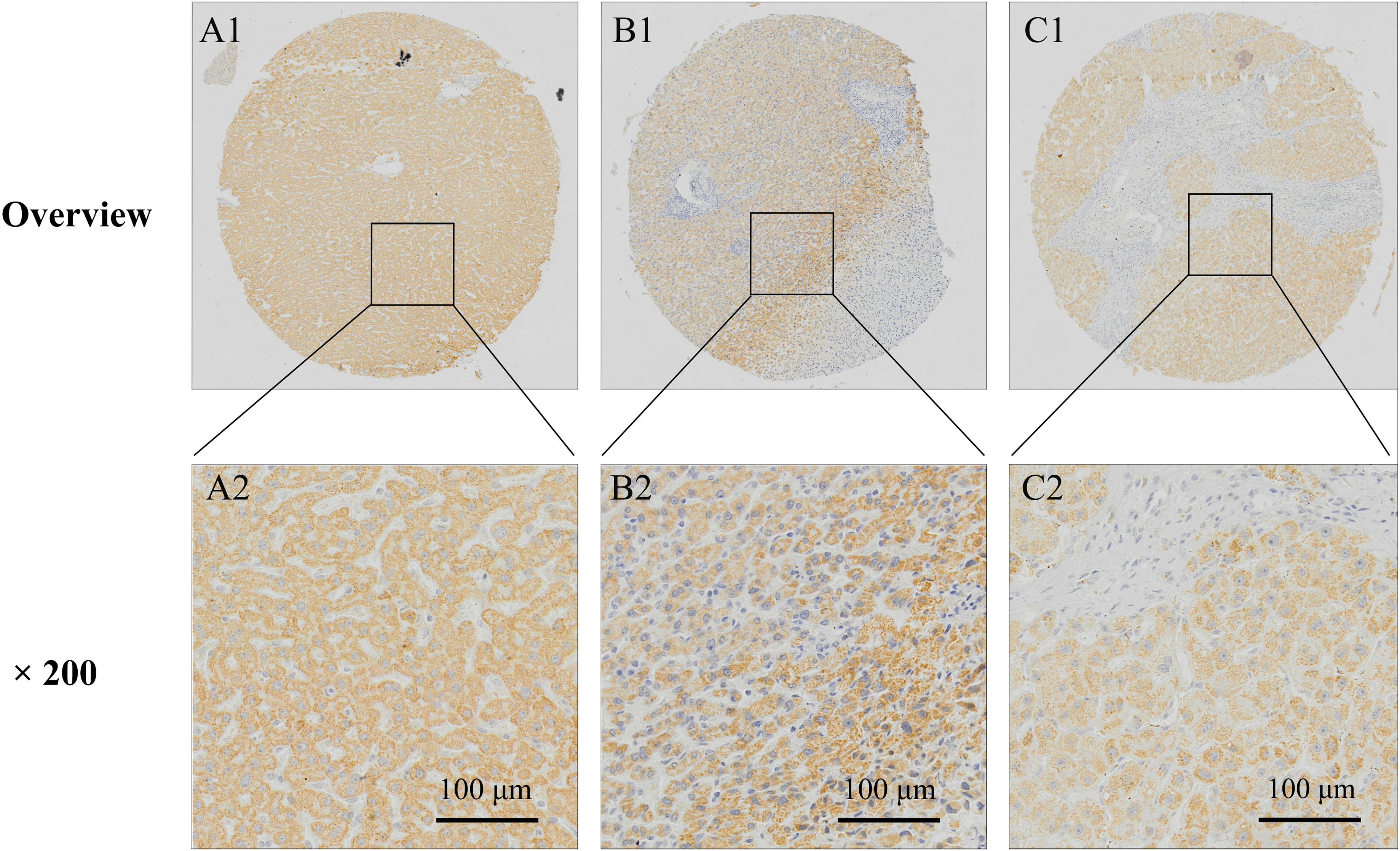
Strong (A), Moderate (B), and Negative (C) expression of PTH1R in HCC tissues.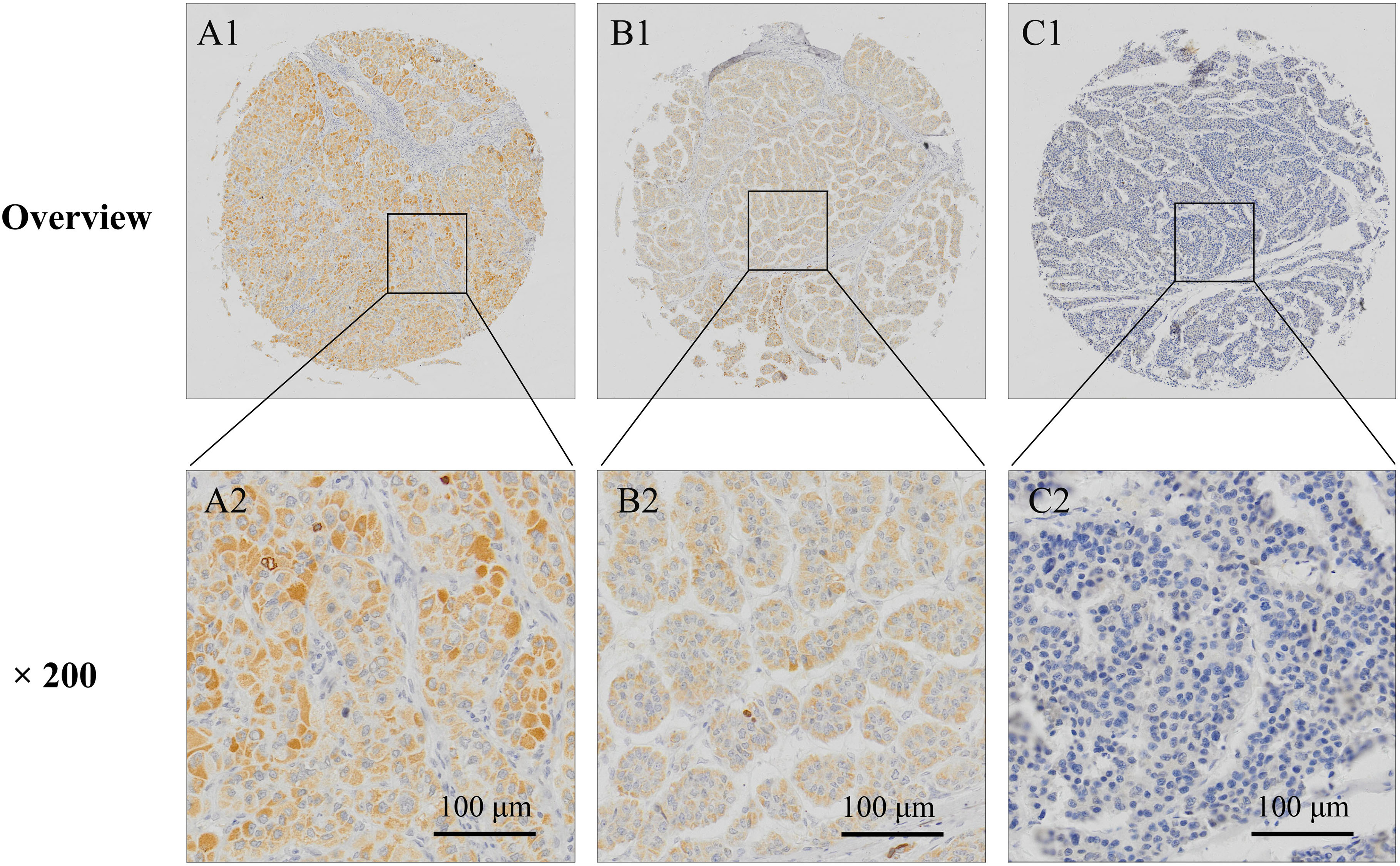
Expression of PTH1R in non-tumorous liver tissues
Expression of PTH1R in non-tumorous liver tissues
Immunostaining of PTH1R was positive in cytoplasm and negative in nucleus in non-tumorous liver and HCC tissues. PTH1R was found highly expressed in 90 of the 102 (88.23%) cases of adjacent non-tumorous liver tissues (29 cases of normal liver tissues, 36 cases of inflammatory liver tissues, and 25 cirrhotic liver tissues), only 12 cases were PTH1R low expression (Table 1 and Fig. 4). The expression of PTH1R was significantly reduced in HCC tissues. High expression of PTH1R was found in 91 of 223 (40.81%) HCC tissues (Fig. 5).
Relationship between PTH1R expression and pathological parameters of HCC
The correlation between expression of PTH1R and clinical variables was shown in Table 2. The PTH1R expression was significantly related to tumor size, Edmondson Grade and AFP. The expression of PTH1R in tumor with big size, high Edmondson Grade, or high AFP level was significantly lower than that in tumor with small size, low Edmondson Grade, or AFP level. There was no significant correlation between PTH1R expression and other clinicopathologic parameters.
Survival analysis
Kaplan-Meier survival analysis showed that the 5-year cumulative survival rate for patients with low PTH1R expression was 59.2%, whereas of the patients with high PTH1R expression was 73.5%. The mean survival time of the patients with low PTH1R expression was 40.08
Kaplan-Meier survival curves of the HCC patients with high or low PTH1R expression.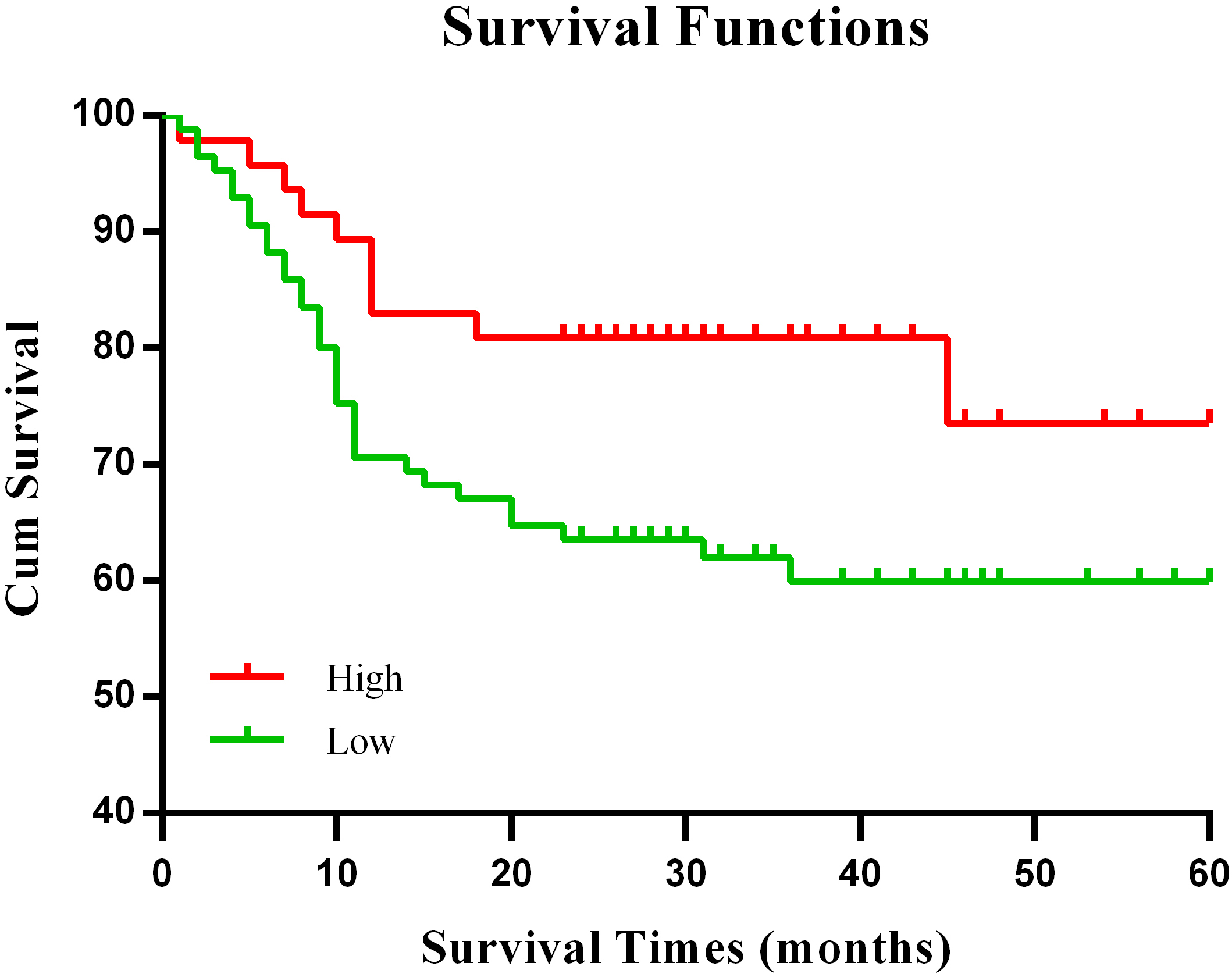
Hepatocellular carcinoma (HCC) is a common malignant tumor of digestive system, ranks fourth in cancer incidence and third in cancer mortality in China [31]. Hypercalcemia is a potentially fatal and not rare complication of HCC. The most common cause of hypercalcemia is excess PTH released by the parathyroid glands. Sometimes, certain cancers, such as breast cancer, lung cancer, and certain blood cancers, could release PTHrP and result calcium release from the bones into the bloodstream [11, 12]. In addition, metastasis, or spreading of a cancer to the bones also can increase the risk of hypercalcemia. In this study, we analyzed the expression of PTH1R, PTH, and PTHrP, the three most important regulators of calcium and phosphate, in HCC. A meta-analysis on online Oncomine Expression Array database showed that the expression of PTH1R in 415 cases of HCC tissues was significantly lower than that in 346 cases of normal liver tissues. Further immunohistochemical staining also confirmed the decreased expression of PTH1R in HCC tissues. There was no significant difference between the expression of PTH in HCC and normal liver tissues. Although there are a few case reports and studies with small sample size showed that high PTHrP expression in HCC patients with hypercalcemia [32, 33, 34, 35], the meta-analysis showed that PTHrP mRNA was slightly higher in HCC tissue than in normal tissues, with no statistical significance. These results suggested that decreased expression of PTH1R may be the major cause of hypercalcemia in HCC.
The underlying mechanism of hypercalcemia in HCC is still unclear. As previous studies reported, both liver and kidneys contribute to the clearance of circulating intact PTH, which may involve receptor-triggered endocytosis mediated by the PTH1R. It was reported that Kupffer cells could uptake intact PTH, proteolyse N-terminal portion of the PTH, and generate various circulating C-terminal PTH peptide (CPTH) fragments; while hepatocytes and sinusoidal cells could uptake both intact PTH and N-PTH fragments [36, 37, 38]. Kidneys are crucial in clearance from blood of both intact PTH and, especially, CPTH fragments [36, 39]. In combination with existing reports and our results, we present hypotheses: decreased expression of PTH1R in HCC tissues reduced the clearance of circulating PTH, which result in retention of PTH in blood, and the subsequently hypercalcemia, hypophosphataemia, bone thinning and fractures. On the other hand, failure of clearance of circulating PTH by liver and the subsequently hypercalcemia increased kidney burden.
Besides, the decreased expression of PTH1R in HCC is also contrary to the previous studies in other malignant tumors. Over-expression of PTH1R has been detected in numbers of neoplastic tissues, including colorectal carcinoma, prostate cancer, renal cell carcinoma and osteosarcoma [10, 24]. Over-expression of PTH1R conferred an aggressive phenotype in osteosarcoma, and PTHR1 knockdown resulted in a profound inhibition on invasion and growth in osteosarcoma cells [24, 40]. PTH1R was also frequently expressed in breast cancer bone metastases, and drived proliferation via cAMP and ERK pathways, while PTH1R silencing suppressed cell proliferation [25]. However, in this study, we found decreased expression of PTH1R was significantly related to big tumor size, high Edmondson Grade, high serum AFP level and poor overall survival. Similar to HCC, meta-analysis based on Oncomine database also showed significantly down-regulation of PTH1R in renal carcinoma, and survival analysis with TCGA data showed that low expression of PTH1R was a poor prognosis in patients with renal carcinoma (data not shown). This may due to the biological roles of PTH1R in liver and kidneys in the clearance of PTH. The mechanisms involved in PTH1R down-regulation and downstream regulation pathway in HCC remain explore.
In conclusion, our results indicated that the decreased expression of PTH1R may be the main cause of hypercalcemia in HCC. Decreased expression of PTH1R was associated with tumor size, Edmondson Grade, serum AFP level and poor overall survival, and was a poor prognosis in HCC.
Footnotes
Acknowledgments
This work was supported by the grants from the National Science Foundation of China (81602174 and 81672430), Zhejiang Provincial Natural Science Foundation of China (LY16H160042 and LY17H160062), Funds of Science Technology Department of Zhejiang Province (No. GF18H160059, GF18H160061, 2015C37089), Zhejiang Province Bureau of Health (Nos. 2018ZZ002, WKJ-ZJ-1710, and 2015ZA009).
Conflict of interest
None.
