Abstract
BACKGROUND:
Cisplatin-based concurrent chemoradiotherapy is recommended for nasopharyngeal carcinoma (NPC) at advanced stages. Excision repair cross-complementation group 1 (ERCC1) plays an important function in the repair of DNA damage that is a critical process of chemo- and radiotherapy.
OBJECTIVE:
This study aimed to investigate the clinical significance of ERCC1 expression in NPC treated with cisplatin-based concurrent chemoradiotherapy in locoregionally advanced NPC.
METHODS:
The expression level of ERCC1 and its association with clinicopathological characteristics in 205 locoregionally advanced NPC patients receiving cisplatin-based concurrent chemoradiotherapy were analyzed retrospectively.
RESULTS:
The correlation analysis revealed that the treatment-sensitive patients displayed dramatically lower ERCC1 expression than treatment-resistant cases did. Furthermore, the Kaplan-Meier plots revealed lower ERCC1 expression was significantly associated with better survival. Multivariate analysis further showed that the ERCC1 expression was an independent predictor of NPC patients’ survival.
CONCLUSIONS:
ERCC1 expression might be a useful predictive marker in patients with locoregionally advanced NPC receiving cisplatin-based concurrent chemoradiotherapy.
Introduction
Nasopharyngeal carcinoma (NPC) is one of most common malignancies in South China. Most NPC patients presented locoregionally advanced disease at diagnosis. In 1998, the phase III randomized Intergroup 0099 study had showed that concurrent chemoradiotherapy (CCRT) with adjuvant chemotherapy provided a 31% increase in 3-year overall survival [1]. Currently, this regimen is the standard treatment approach for advanced NPC [2]. Cis-platinum, which not only inhibited the recovery of tumor cells from sub-lethal and potentially lethal radiation damage but also had minor myelosuppression effect, has become the standard concurrent chemotherapy drug for advanced NPC [2]. Recently, several clinical trials have confirmed the value of the addition of chemotherapy on survival for advanced NPC [3, 4, 5, 6, 7, 8]. However, the treatment outcomes varied greatly, and were associated with some adverse effects as well [3, 4, 5, 6]. Therefore, it is very important and necessary to identify factors that can predict which patients benefit from CCRT with adjuvant chemotherapy.
Repair of DNA damage is an important response to chemo- and radiotherapy. Thus, the expression and activity of proteins related to DNA damage repair could affect the outcomes of the treatment [9]. Excision repair cross-complementation group 1 (ERCC1) gene plays as a rate-limiting enzyme in the nucleotide excision repair (NER) pathway, one important mechanisms that involves in the repair of non-specific DNA damage induced by radiation and chemicals [10, 11, 12]. Recently, increasing experimental and clinical evidences suggested that the up-regulated mRNA or protein level of ERCC1 was highly associated with platinum resistance in lung cancer, ovarian cancer and gastric cancer, whereas the patients with low RECC1 expression level could gain the survival benefit from platinum-based chemotherapy [11]. Moreover, ERCC1 has been suggested as a prognostic marker in patients of triple negative breast cancer, urothelial cancer, N2 non small-cell lung cancer and squamous cell carcinoma of the head and neck treated with platinum-based CCRT [12, 13, 14, 15, 16, 17]. However, the expression and significance of ERCC1 in NPC is still unclear, with confused conclusions by different groups, especially in NPC that received cisplatin-based CCRT [18, 19, 20]. Thus, our study analyzed the protein level of ERCC1 in NPC patients treated with standard platinum-based CCRT, and discussed the significance of ERCC1 in tumor response and cancer-specific survival.
Association between ERCC1 expression and clinical features of 205 NPC patients
Association between ERCC1 expression and clinical features of 205 NPC patients
Human NPC tissue specimens
All procedures performed in this study involving human participants were in accordance with the ethical standards of the ethics committee of the First Affiliated Hospital of Sun Yat-sen University and with the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards. Informed consent was obtained from each patient.
Human NPC tissues were collected from 205 patients who underwent CCRT at the Guangzhou General Hospital of Guangzhou Military between 2001 and 2009. All cases were pathologically diagnosed as locoregionally advanced nasopharyngeal cancer. The median age for the 205 NPC patients was 44 years and the sex ratio (male v.s. female) was 2.5:1. There was no distant metastasis in all patients at diagnosis. The TNM classifications of 205 patients were T
The chemoradiotherapy regimen consisted of100 mg/m
All patients received magnetic resonance imaging (MRI) of pars nasalis pharyngis and neck 12 weeks after completion of treatment, and tumor response was evaluated according to the Response Evaluation Criteria in Solid Tumors (RECIST). Based on the tumor response, all 205 cases were divided into complete response group (CR), partial response group (PR), stable disease group (SD) or progressive disease group. The response rate (RR) was calculated as the ratio of CR and PR cases in all patients.
All patients were followed to assess survival rates, and monitor for recurrence and metastases by chest X-ray, abdomen B ultrasonic analysis or MRI of pars nasalis pharyngis and neck every 3 months for the first year and every 6 months for the following years.
According to the imaging examination, patients that with tumor residual (33 cases) or distant metastasis (30 cases) or local relapse (15 cases) after therapy were divided into the CCRT-resistant group while the others (127 cases) were divided into CCRT-sensitive group.
Analysis of gene expression
Immunohistochemistry was used to assess ERCC1 expression in nasopharyngeal tumor tissues. Immunostaining was performed with the ChemMate DAKO EnVision Detection Kit for Peroxidase/DAB/Rabbit/ Mouse (code K 5007, Dako Cytomation, Glostrup, Denmark). Mouse monoclonal antibodies for ERCC1 (cat. Sc-56386, Santa Cruz) were used for Immunohistochemical staining.
Immunohistochemical staining of ERCC1. ERCC1 is expressed in the nucleus of NPC cells with different intensities indicated by H-score. A brown signal was considered to be a positive ERCC1 stain. Images were captured at a magnification of 200X. Scale bar, 50 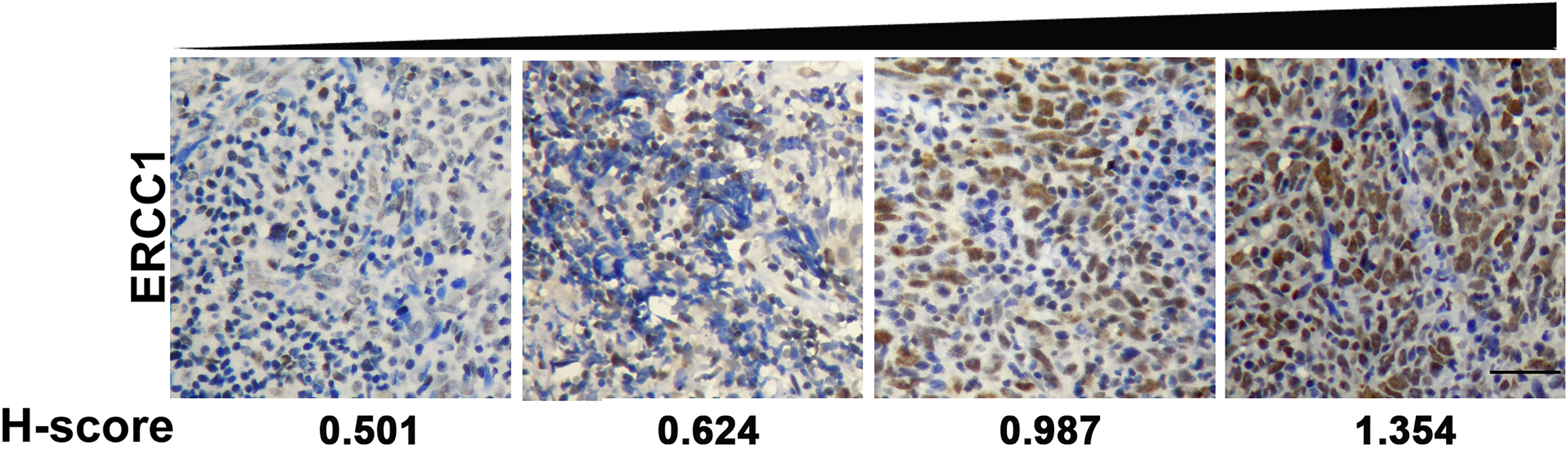
The ERCC1 expression in the tissues was evaluated under a light microscope at a magnification of 200X by two molecular pathologists independently. Five images of random areas were acquired for each specimen, and a total of 500 tumor cells were counted. IHC scoring was performed according to a modified Histo-score, which included a semi-quantitative assessment of both the fraction of positive cells and the intensity of staining. The intensity score was defined as no staining (0), weak (1), moderate (2) or strong (3) staining. The fraction score was based on the proportion of positively stained cells. The intensity and fraction scores were multiplied to obtain an H-score that represented the expression level of ERCC1. The median H-score of all 205 cases was chosen as the cutoff point for separating ERCC1-low-group (100 out of 205 cases) from ERCC1-high-group (105/205).
The failure-free survival (FFS) was calculated from the date of enrollment to the date of the first failure at any site. The overall survival was defined as the time from start of treatment to death from any cause. The Kaplan-Meier plots and the Cox proportional hazard regression analysis, which were applied to identify the prognostic factors, were performed with SPSS, version 17.0 (SPSS Inc., Chicago, IL, USA). Associations between the FFS or OS and the molecular changes or clinical characteristics were analyzed initially by a univariate Cox proportional hazards regression analysis. Significant prognostic factors found in the univariate analysis were evaluated further by a multivariate Cox regression analysis. The differences between groups were analyzed using
CCRT-sensitive group displayed lower ERCC1 expression levels. Patients without tumor residual or distant metastasis or local relapse after CCRT therapy were divided into the CCRT-sensitive group while the others were divided into CCRT-resistant group. The central horizontal line represents the mean value; error bars represent the SEM. CCRT, concurrent chemoradiotherapy. **, 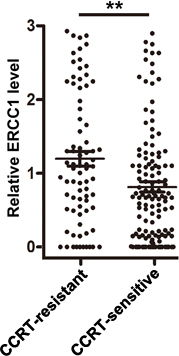
The expression pattern of ERCC1 in human NPC tissues
ERCC1 was mainly expressed in the nucleus of nasopharyngeal carcinoma (NPC) cells (Fig. 1). The positive expressions of ERCC1 were observed in 175 out of 205 tumor samples (85%) with different intensities (Fig. 1). The ERCC1 expression levels in 205 tumors were further quantified and divided into low- and high-expression groups according to the median expression value in all cases. There was no statistically significant correlation between the ERCC1 expression and the clinicopathological features of 205 patients, as shown in Table 1.
Relationship between treatment response and ERCC1 expression
A total of 202 NPC patients were responsive to the concurrent chemoradiotherapy (CCRT) (overall reponse rate (RR), 98.2%), including complete response (CR) in 162 patients (79%), partial response (PR) in 40 patients (19.5%), stable disease (SD) in 3 patients (1.5%) and none with progressive disease (PD). The response rates in ERCC1-high-expression group and ERCC1-low-expression group were 97.1% (102/105) and 100% (100/100), respectively. And there was no statistical difference between them (
Univariate analysis of factors associated with FFS and OS
Univariate analysis of factors associated with FFS and OS
Multivariate analysis of factors associated with OS and FFS
Higher expression of ERCC1 in NPC tissues is associated with a worse prognosis for NPC patients. (A) Kaplan-Meier plots revealed an association of higher ERCC1 levels with a shorter failure-free survival. (B) Kaplan-Meier plots revealed an association of higher ERCC1 levels with a shorter overall survival. The levels of ERCC1 were analyzed by IHC, and the median value of all 205 cases was chosen as the cut-off point for separating the ERCC1-low-expression and ERCC1-high-expression group.
The median follow-up time for all patients was 36.3 months (range from 7 to 90 months, 95% CI). The five-year FFS and OS rate were 56.0% and 51.4%, respectively. In univariate analysis, the median FFS for ERCC1-low expression group was 58.1 months, compared with 47.7 months for ERCC1-high expression group (
Discussion
In the present study, the retrospective analysis revealed that in NPC patients receiving cisplatin-based concurrent chemoradiotherapy (CCRT), the cases with lower expression level of ERCC1 presented significantly better survival than those with higher expression level. Moreover, compared with CCRT-resistant group, patients in CCRT-sensitive group displayed significant lower expression of ERCC1. Ours findings implicate the potential application of ERCC1 in prediction of therapy outcome and prognosis of NPC patients receiving cisplatin-based concurrent chemoradiotherapy.
To date, few reports have tried to explore the prediction value of ERCC1 in NPC. In 2010, studies from two groups have indicated a prediction value of ERCC1 in NPC patients who were treated with cisplatin-based CCRT in Korea. Lee et al. [18] and Huang et al. [19] reported that high expression of ERCC1 predicted poor OS of NPC treated with CCRT. Nevertheless, the sample size of both studies was relatively small (
The 5-year OS rate in the present study was 50.9% which was inferior compared with some phase III trials (
The principal reason for radiotherapy or chemotherapy resistance in cancers is the enhanced repair capacity of DNA damage in tumor cells. It was known that ERCC1 was required for the repair of DNA lesions induced by electrophilic compounds such as cisplatin. Increasing experimental and clinical evidences suggested a correlation between up-regulated ERCC1 expression and platinum resistance in multiple tumors, including ovarian cancer, cervical cancer, testicular cancer, bladder cancer and NSCLC [21]. And functional studies have revealed that suppression of ERCC1 in tumor cells resulted in enhanced chemotherapy-sen- sitivity of cisplatin-resistant lung cancers in vitro [22]. Base on the above observations on the function of ERCC1, we suggested that low level of ERCC1 indicated an inferior repair capacity of DNA damage, in turn rendered the sensitivity for chemotherapy or/and radiotherapy in NPC cells.
In several studies of cDNA microarray analysis, ERCC1 displayed significant up-regulation in radio-resistant cancer cells but down-regulation in radiosensitive cancer cells [23, 24]. Moreover, over-expression of ERCC1 reduced the sensitivity of radiation treatment, suggesting the anti-radiation function of ERCC1 in tumor cells. Combined with our present results, it can be assumed that ERCC1 expression might be also a predictor of radio-sensitivity. However, the conclusion remains to be elucidated.
ERCC1 has been shown to predict the effectof cisplatin-based concurrent chemoradiotherapy inNSCLC [17]. According to the findings from this study and others, it is intriguing to find that ERCC1 can also predict the effect of cisplatin-based concurrent chemoradiotherapy in NPC. It is remarkable that the conclusion should be validated prospectively on a large number of patients in a multi-center study.
Conclusions
ERCC1 expression might be a useful predictive marker in patients with locoregionally advanced NPC receiving cisplatin-based concurrent chemoradiotherapy.
Footnotes
Acknowledgments
This study was supported by Science and Technology Planning Project of Guangdong Province, China (2010B031600266, 2014A020212083).
Conflict of interest
The authors declared that they have no conflict of interest.
