Abstract
Background
To examine the prognostic relevance of pan-immune-inflammation value (PIV) in locally advanced nasopharyngeal carcinomas (LA-NPC) patients treated with concurrent chemoradiotherapy (CCRT) definitively.
Methods
We used receiver operating characteristic (ROC) curve analysis to determine an optimal PIV cutoff that could effectively divide the patient cohort into two distinct groups based on distant metastasis-free (DMFS) and overall survival (OS) results. For this purpose, receiver operating characteristic (ROC) curve analysis was employed. Our primary and secondary endpoints were to investigate the potential correlations between pre-CCRT PIV measurements and post-CCRT OS and DMFS outcomes, respectively.
Results
This retrospective cohort study included 179 LA-NPC patients. The optimal PIV cutoff was 512 (area under the curve: 74.0%; sensitivity: 70.8%, specificity: 68.6%; J-index: 0.394) in ROC curve analysis, creating two patient groups: Group-1: PIV < 512 (N = 108); vs Group-2: PIV ≥ 512 (N = 71). In the comparative analysis, although there were no significant differences between the two groups regarding the patient, disease, and treatment characteristics, the PIV ≥ 512 group had significantly poorer median OS [74.0 months vs not reached yet (NR); HR: 2.81;
Conclusions
The present findings indicate that PIV, which classifies these patients into two groups with significantly different DMFS and OS, might be a potent prognostic biological marker for LA-NPC patients.
Introduction
Nasopharyngeal carcinomas (NPCs) are highly aggressive cancers that develop from the squamous epithelial cells lining the lateral wall of the nasopharynx, which are most prevalent in endemic areas like China and northern Africa. 1 NPCs have distinct epidemiology, pathology, clinical characteristics, and treatment outcomes compared to other head and neck cancers. The current standard treatment options for medically fit, locally advanced LA-NPC patients consist of concurrent chemoradiotherapy (CCRT) with intensity-modulated radiotherapy (IMRT) or induction chemotherapy followed by IMRT-based CCRT.2-4 Despite significant advancements in diagnostic and staging tools and the successful implementation of concurrent chemotherapy and IMRT, which have resulted in loco-regional tumor control rates exceeding 90%,2,3 up to 20% of all LANPC cases still experience distant metastasis (DM) during the course of the disease.2-5
The TNM (tumor-node-metastasis) staging system is the most comprehensive current framework for predicting the prognosis of patients presenting with locoregionally advanced nasopharyngeal carcinoma (LA-NPC). However, clinical outcomes may significantly vary among patients even if they receive similar anti-cancer interventions for identical disease stages, which may stem from inherent tumor- and host-related biological disparities.6,7 These biological differences and their unpredictable impact on clinical results underscore the necessity for novel and efficacious biomarkers to complement conventional TNM staging for more accurate prognostic classification of these patients. Persistent systemic inflammation and deficient antitumor immunity are crucial factors that trigger various phases of cancer progression, including cellular transformation, tumor cell survival, growth, invasion, extravasation, metastatic dissemination, and immune evasion. 8 Consequently, several combinations of blood-borne monocytes, platelets, neutrophils, and lymphocytes have been studied to ascertain their prognostic relevance in NPC patients. Multiple research studies consistently show that specific biomarkers involving the ratio of two or three types of cells, such as the neutrophil-to-lymphocyte ratio (NLR),5,9 monocyte-to-lymphocyte ratio (MLR), 9 platelet-to-lymphocyte ratio (PLR),10,11 systemic immune-inflammation index (SII), 12 and systemic immune response index (SIRI), 13 can robustly predict the prognosis of patients with LA-NPC who receive systemic chemotherapy or concurrent chemoradiotherapy (CCRT).
In 2020, Fucà and colleagues introduced a novel comprehensive biological marker, the pan-immune-inflammation value (PIV), which integrates the platelet, monocyte, neutrophil, and lymphocyte counts.
14
The authors investigated its prognostic utility in 438 stage IV colorectal cancer patients who received systemic chemotherapy. The findings of the investigation revealed that patients with higher PIV exhibited inferior progression-free survival (PFS) (
Patients and Methods
Study Design and Participants
The reporting of this study conforms to REMARK guidelines. 25 The present investigation was a retrospective cohort study conducted at the Department of Radiation Oncology of the Baskent University Medical Faculty with a single-center design. Records of LA-NPC patients who had CCRT at our facility between June 2010 and December 2019 were reviewed retrospectively. The study’s eligibility criteria encompassed the following requirements: patients aged 18 to 80 years, an Eastern Cooperative Oncology Group (ECOG) performance score of 0-2, and a BMI of at least 18.5 kg/m2. Participants had to have accessible pretreatment clinical ear-nose-throat (ENT) examinations, head and neck magnetic resonance imaging (MRI) scans, fluorodeoxyglucose-positron emission computerized tomography (PET-CT), clinical/radiological T3-4N0-3M0 or T1-4N1-3M0 disease according to the AJCC 8th edition, documented non-keratinizing or undifferentiated squamous cell histology, no history of other cancers, chemotherapy or RT, infectious diseases, or immune suppressive drug use in the past 30 days before the commencement of CCRT. Patients need to have received one or more cycles of platinum-based concurrent chemotherapy. Accessible RT and chemotherapy charts, available baseline complete blood count test records, records of follow-up ENT examinations, and MRI and PET-CT scans were also required.
Ethics and Consent
The Institutional Review Board of the Baskent University Medical Faculty approved the study design before collecting any patient data (Project no: D-KA-2058), which adhered to the exact specifications stipulated in the Declaration of Helsinki and its subsequent updates. Participants who met the eligibility requirements for the study gave their informed consent for the collection and analysis of blood samples and pathological specimens, and for the publication of the research results through academic channels. Preliminary results of these data were published as an oral presentation at the 8th International Black Sea Coastline Countries Scientific Research Conference, August 29-30, 2022, Sofia, Bulgaria. 26
Chemoradiotherapy Protocol
All study participants underwent definitive CCRT utilizing previously documented dosages of RT and chemotherapy. 27 In brief, we employed intensity-modulated radiotherapy (IMRT) administered in daily fractions over 7 weeks, 5 days per week. Concurrently, chemotherapy was delivered with cisplatin dosages of 75-80 mg/m2 at 3-week intervals. After CCRT, adjuvant chemotherapy consisted of two cycles of cisplatin-based regimens at 3-week intervals were advised if deemed tolerable, contingent upon treatment protocol approval. Additionally, appropriate supportive care measures, including the provision of antiemetic medications and oral/enteral nutritional supplementation, were administered as clinically indicated.
Measurement of Pan-Immune-Inflammation Value
The pre-CCRT PIV was estimated for each patient using the original Fucà formula: PIV = [P × M × N] ÷ L, with P, M, N, and L representing the counts (per microliter) of platelets, monocytes, neutrophils, and lymphocytes acquired on day 1 of CCRT. 14
Response Assessment
Although the research design was retrospective, the CCRT response evaluations were prospective under the norms of our institution. In the initial 2-year and 3- to 5-year periods of CCRT, patients underwent evaluations every 3-month and every 6-month intervals, respectively. Then, the patients underwent annual or more frequent assessments as deemed necessary based on clinical indications. A meticulous endoscopic ENT examination was conducted at each visit to identify potential local or regional tumor recurrences and the likelihood of second primary tumors. In addition, PET-CT scans were acquired to evaluate the probable distant metastasis (DM) and determine the type of treatment response. The PET Response Criteria for Solid Tumors (PERCIST) was the standard tool for response evaluations. When a complete metabolic response was ascertained, head and neck MRI and/or CT scans were used instead of PET-CT scanning from that point on. Supplementary imaging modalities were exclusively utilized for the detection of suspected lesions or the reevaluation of recurrent tumors.
Clinical Endpoints and Statistics
This study’s primary objective was to ascertain if there was a correlation between the pre-CCRT PIV and OS (the interval between the commencement of CCRT and the death or last follow-up records. The secondary objective was to evaluate the same relationship with DM-free survival (DMFS: The duration between the initiation of CCRT and the occurrence of any distant or non-regional lymph node relapses, death, or the final visit. Continuous data were represented using medians and ranges, whereas frequency distributions characterized categorical variables. We used the student’s t-test, Chi-square test, or Spearman correlations to compare frequency distributions across different groups. We utilized receiver operating characteristic (ROC) curve analysis to evaluate the availability of an optimal pre-CCRT PIV cutoff value that would divide the entire study population into two groups with significantly different DMFS and OS results. Kaplan-Meier curves and log-rank tests were employed to explore the potential interactions between the tested risk factors and the results of DMFS and OS. The Cox proportional hazards model was applied to carry out multivariate analyses to assess the independence of variables that demonstrated univariate significance. All comparisons between groups were conducted using a two-tailed test, and statistical significance was determined at a level of
Results
Pretreatment Patients and Disease Characteristics.
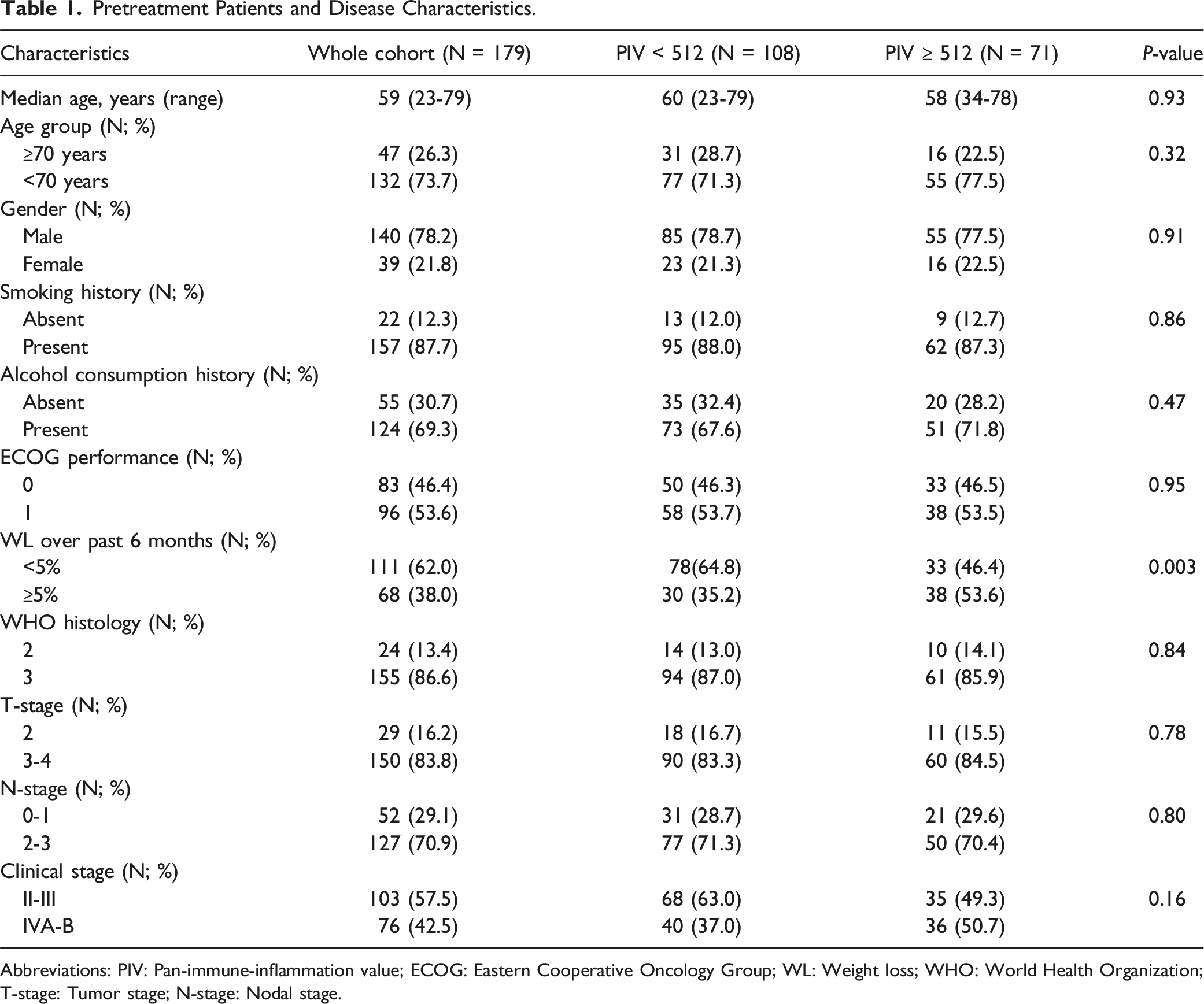
Abbreviations: PIV: Pan-immune-inflammation value; ECOG: Eastern Cooperative Oncology Group; WL: Weight loss; WHO: World Health Organization; T-stage: Tumor stage; N-stage: Nodal stage.
Treatment and Survival Results per Pan-Immune-Inflammation Value Group.

Abbreviations: PIV: Pan-immune-inflammation; OS: Overall survival; DMFS: Distant metastasis-free survival.
We searched to find the optimal PIV cut-off values that could effectively stratify the study cohort into two groups with significantly different prognoses. ROC curve analyses yielded two ideal cut-off points, namely 512 [area under the curve (AUC): 74.0%; sensitivity: 70.8%; specificity: 68.6%; J-index: 0.394] and 508 (AUC: 83.3%; sensitivity: 72.9%; specificity: 70.7%; J-index: 0.436), which were found to be significantly correlated with the OS and DMFS results, respectively (Figure 1). Based on the proximity of the two cut-off values, a decision was made to select 512 as the PIV cut-off point for categorizing patients into two distinct groups: Group 1: PIV < 512 (N = 108) and Group 2: PIV ≥ 512 (N = 71). Considering the pretreatment and treatment characteristics, WL ≥ 5% was found to be meaningfully more frequent in the PIV ≥ 512 cohort than in the PIV < 512 cohort (53.6% vs 35.2%; Results of receiver operating characteristic curve analyses: (a) Distant metastasis-free survival, (b) Overall survival.
After a median follow-up period of 85.7 months (95% CI: 67.9-103.5 months), 134 out of 179 participants (74.9%) were still alive, and 96 (53.6%) were free from the disease. Among the 45 deaths, 30 (66.7%) were due to cancer and its treatment, with 28 cases attributed to uncontrolled LA-NPC progression and 2 cases due to late toxicities. Non-NPC factors caused the remaining 15 deaths (33.3%). Locoregional control (LRC) was attained in 161 (89.9%) patients. Although not statistically significant, the group with PIV ≥ 512 showed a numerically lower LRC rate compared to the PIV < 512 group (87.9% vs 93.0%;
The median DMFS and median OS times were not reached during the final analysis for the whole research cohort, as indicated by the Kaplan-Meier survival estimates. The DMFS and OS rates at 10 years were 50.2% and 67.9%, respectively (Table 2). When the two PIV cohorts were compared, the PIV ≥ 512 patients had significantly poorer median DMFS [27.0 months (95% CI: 14,3-39.7) vs not reached (NR); Comparative survival outcomes between the pan-immune-inflammation value groups, (a) Distant metastasis-free survival (b) Overall survival (dark blue line: low pan-immune-inflammation value; red line: high pan-immune-inflammation value).
Results of Uni- and Multivariate Analysis.

Abbreviations: OS: Overall survival; DMFS: Distant metastasis-free survival; HR: Hazard ratio; ECOG: Eastern Cooperative Oncology Group; WHO: World Health Organization; T-stage: Tumor stage; N-stage: Nodal stage; PIV: Pan-immune-inflammation.
Discussion
Identifying new biological markers in patients with LA-NPC who receive radical CCRT may enhance patient selection and customize treatment protocols. Consequently, given the lack of comparably designed antecedents, this retrospective study aimed to evaluate the prognostic value of the novel PIV in a cohort of 179 LA-NPC patients who received CCRT. The results revealed that LA-NPC patients with PIV ≥ 512 displayed decreased tolerance to the prescribed concurrent and adjuvant chemotherapy cycles. Furthermore, this patient group exhibited significantly inferior median and long-term DMFS and OS outcomes in comparison to those with PIV < 512. The significance of these findings lies in their indication of a direct association between a chronically compromised immune-inflammatory state and decreased tolerance to treatment protocol, as well as unfavorable clinical outcomes. This information could prove beneficial in precisely risk stratification of such patients and selecting the optimal individualized treatment modalities.
The highly inflammatory nature of NPC is attributed to the spatial arrangement of stromal cells within tumor compartments. These cells reside in proximity to NPC cells and exhibit physical contact with them, as opposed to being situated in the surrounding regions.
28
Additionally, NPCs show a higher level of stromal infiltration than most other solid tumors due to the abundance of secondary and tertiary lymphoid structures, creating a unique environment that promotes a significant influx of inflammatory cells and associated chemokines/cytokines.
29
While the precise mechanisms remain complex, it is plausible that these inflammatory cells may promote various aspects of tumor-cell behavior, including proliferation, survival, invasiveness, migration, and metastasis. Furthermore, they have the potential to increase resistance to apoptotic stimuli and anti-neoplastic therapies while also impeding the tolerability of chemotherapy and RT, thereby resulting in elevated rates of both acute and chronic toxicity.
30
Elevated levels of regulatory T-cells, myeloid-derived platelets, monocytes, and neutrophils can have both inflammatory and immunosuppressive effects, potentially reducing the effectiveness and tolerance of therapy. T-cells possess distinct antitumor immune and anti-inflammatory properties compared to other cell types, thereby enhancing treatment efficacy across various tumor categories, including LA-NPCs.
29
In this respect, our findings reasserted the existing fundamental and clinical evidence by highlighting that, patients in the PIV ≥ 512 group demonstrated a reduced ability to tolerate both concurrent (85.9% vs 94.4%;
Several immune-inflammation indices, such as lymphocyte counts, NLR,
9
PLR,
10
MLR,
11
SII,
12
and SIRI,
13
have previously been utilized to predict the prognosis of LA-NPC patients undergoing CCRT. However, our current study is the first to investigate the prognostic value of PIV, a novel comprehensive four-cell immune-inflammation index, in LA-NPC patients receiving radical CCRT. The study findings revealed that pretreatment PIV measurements were independently prognostic in the patient population under investigation. Specifically, a PIV value of ≥ 512 was found to be significantly associated with shorter median DMFS (27.0 months vs NR;
We acknowledge that certain limitations constrain our research. First, the current findings may be susceptible to selection bias due to the retrospective nature of the cohort analysis, which was conducted at a single center with a relatively modest cohort size. Second, our results may not be universally applicable to all patients with LA-NPC, particularly those with an ECOG 2 performance score or those undergoing different RT or chemotherapy regimens, as our study exclusively included patients with an ECOG performance score of 0-1 who were staged and treated uniformly. Third, as components of PIV may fluctuate dramatically over time, the pretreatment PIV cutoff utilized in this investigation and the results reported may only represent a snapshot in time, as we did not collect additional data throughout or after the C-CRT course. Fourth, the absence of Epstein-Barr virus (EBV)–related parameters in most patients poses a challenge in correlating PIV groups with these parameters in our study. However, a recent investigation by Zhang and colleagues into the correlation between EBV and PIV statuses revealed no significant relationships between the two parameters. 38 Consequently, if EBV status does exert an influence on the PIV groups, it is likely to be negligible or non-existent. 33 And fifth, the lack of an internal and/or external validation cohort may have underrated specific crucial parameters, such as the well-established prognostic relevance of the presenting T-stage. Therefore, we concede these obstacles and advocate viewing current findings as hypothesis-generating rather than firm recommendations until additional data from well-designed future research addressing these constraints may validate them. Nevertheless, when viewed through the lens of Clark’s well-respected prognostic factor definition, 39 our findings appear to point to the novel PIV as a dependable prognosticator, as it is a biological parameter that is simple to obtain and compute, objectively measurable, replicable, inexpensive and provides data regarding the probable outcome of LA-NPC patients regardless of their clinical conditions.
Conclusion
Notwithstanding the limitations of a retrospective cohort study conducted at a single institution, our current findings emphasize the efficacy of the novel PIV as a dependable prognostic marker for patients with locally advanced nasopharyngeal carcinoma (LA-NPC). Given the PIV ≥ 512 cohort’s drastically reduced treatment tolerance and worse survival outcomes, if further research confirms, these findings may serve valuably in precisely selecting the best-suited treatment options for such patient.
Footnotes
Author Contributions
E.T., U.S., D.O., S.S, and N.K.D. conceived the study, participated in the study’s design, and performed clinical examination and statistical analysis. All authors contributed significantly and equal, and all authors approved the final form of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Statement
Data Availability Statement
The present data belongs to and is stored at the Baskent University Faculty of Medicine; it cannot be shared without permission. Researchers who meet the criteria for access to confidential data should contact the Baskent University Department of Radiation Oncology at
