Abstract
BACKGROUND:
Chronic hepatitis C (CHC) is a contagious liver disease that results from infection with the hepatitis C virus (HCV). The most serious consequence of CHC is HCV-related hepatocellular carcinoma (HCC).
OBJECTIVE:
To illustrate the clinical significance of lncRNA HEIH expression in serum and exosomes in the development of HCV-related HCC.
METHODS:
Thirty-five CHC, twenty-two HCV-induced cirrhosis and ten HCV-related HCC patients in Huzhou Central Hospital from January 2016 to September 2016 were recruited in the present study. Basic patient information, clinical serological indicators, and clinical imaging data were investigated and analyzed. Serum samples were collected from patients after receiving informed consent. Exosomes were extracted from the serum, and electron microscopy was used to observe the ultrastructure of exosomes. Quantitative PCR was used to detect lncRNA HEIH gene expression in serum and exosomes.
RESULTS:
The changes in the ALT, GGT, HDL, INR, Alb and AFP levels in the patients with HCV-induced cirrhosis and HCV-related HCC were statistically significant. In patients with HCV-related HCC, lncRNA-HEIH expression in serum and exosomes was increased, but the ratio of lncRNA-HEIH expression in serum versus exosomes was decreased compared to patients with CHC.
Introduction
The incidence of chronic hepatitis C (CHC) is increasing worldwide, and Central and East Asia and North Africa have a high prevalence of this disease and the morbidity is more than 3.5% [1]. Patients with CHC are at high risk for liver cirrhosis and hepatitis C virus (HCV)-related hepatocellular carcinoma (HCC). Both HCV-induced cirrhosis and HCV-related HCC remain serious end-stage consequences of CHC [2] despite the progress that has been made in CHC diagnosis and treatment [3]. CHC, HCV-induced cirrhosis and HCV-related HCC are the three distinct stages of HCV infection. These stages are traditionally diagnosed in clinical practice using the combination of radiographic appearance, serum biochemistry indicators and histologic features. The present study investigated and analyzed liver function and liver related serological indices of patients with hepatitis C, HCV-induced cirrhosis and HCV-related HCC.
The exosome is a discoid vesicle that is 40–150 nm in diameter. Exosome production begins with invagination of the lysosome and formation of the multivesicular endosome, which fuses with the cellular membrane and is then released into the extracellular matrix (ECM) [4, 5]. The exosome was first detected in the 1980s and was considered to be a carrier to excrete metabolites. Recent studies have focused on its biological source, material constitution, intracellular and intercellular signal transduction and distribution in bodily fluids [6, 7]. These studies show that the exosome is involved in the regulation of multiple physiological and pathological functions, including the body’s immune response, antigen presentation, activation of T lymphocytes, cell migration, cell differentiation and tumor invasion and metastasis [8, 9, 10].
In cancer, exosomes are involved in information exchange between cancer cells and basal cells and play a synergistic role in prompting angiogenesis and metastasis [11, 12]. Long non-coding RNAs (lncRNAs) are defined as a class of large and heterogeneous RNA transcripts which are more than 200 nucleotides in length, and cannot code for protein [13]. Some of these transcripts were shown to be potential functions of regulating cellular differentiation and proliferation, as well as having oncogenic functions in cancers [14]. Accumulating studies of cancer-associated lncRNAs have reported lncRNAs regulate biological functions in a variety of cancers and are a potential diagnostic/prognostic marker and therapeutic target [15]. The hepatocellular carcinoma up-regulated EZH2-associated long non-coding RNA (HEIH) lncRNA-HEIH was first reported to be as an oncogenic ncRNA that promotes tumor progression by Zhang et al. [16]. It is an lncRNA of 2.2 kb whose gene locates in chromosome 5q35.3. lncRNA-HEIH, as a key regulatory gene in HCC, is specific to and highly expressed in hepatocellular carcinoma cell lines and tissues. More, lncRNA-HEIH was up-regulated in HBV-related HCC and it was significantly associated with recurrence and prognosis [17, 18]. The lncRNA-HEIH is a single-stranded RNA that is unstable in serum due to the abundance of RNA-degrading enzymes. Exosomes may protect lncRNA from degradation [19, 20, 21]. In the present study, expression of lncRNA-HEIH was detected in exosomes extracted from serum. Accumulating evidence suggests that the exosome transfer of functional lncRNAs between cells might play an important role in cancer development and anti-cancer drug resistance by altering and/or regulating local cellular microenvironments [22, 23, 24]. Recent studies have shown that exosomes altered HCC growth, metastasis and resistance to antitumor agents by serving as vehicles for the transfer of proteins, viruses, lipids and RNA between cells [25, 26, 27]. The molecular mechanism of exosome- and lncRNA-mediated regulation in HCC remains unclear.
HCV is thought to disrupt the immune system, leading to chronic liver disease-chronic hepatitis C, liver cirrhosis, liver failure and hepatic cellular carcinoma [8]. Accumulating evidence suggests that exosomes and lncRNA can regulate the immune system [28, 29, 30]. However, the roles of exosomes and lncRNA remain unclear and require further study. Most studies were performed in vitro and have not been confirmed in vivo. We detected and analyzed the lncRNA-HEIH genes in serum and exosomes from patients with CHC, HCV-induced cirrhosis and HCV-related HCC. These results provide valuable insight on the mechanism of HCC development.
Materials and methods
Inclusion criteria and exclusion criteria
Patients with CHC, HCV-induced cirrhosis and HCV-related hepatocellular carcinoma in Huzhou Central Hospital from January 2016 to September 2016 were studied. The clinical trials involving the patients and the informed consents were approved by the Ethics Committee of Huzhou Central Hospital (No.2016.31). The inclusion criteria were as follows: (1) For all patients, the serum HCV antibody was positive, and the number of copies of serum HCV-RNA were higher than 1000 copies per milliliter. (2) Diagnoses of cirrhosis and HCC were made based on ultrasonography, CT, MRI scans, and pathological examination. (3) Diagnoses of CHC, HCV-induced cirrhosis and HCC were made by at least three doctors, including a chief physician and imaging doctor. (4) At the time of enrollment, all patients signed informed consents under guidelines approved by the Ethics Committee of Huzhou Central Hospital at enrollment. (5) All patients were not taking any medications that might cause liver damage in the past three months. (6) The patients with cirrhosis and HCC did not undergo liver transplants. The exclusion criteria were as follows: (1) Patients co-infected with other hepatitis viruses. (2) The main causes of liver cirrhosis and HCC were not HCV infection. (3) The patients suffered from serious heart problems, severe respiratory disease, diabetes, infections or other tumors. (4) Pregnant or lactating women. (5) Patients with a history of drug or Chinese herb use within the last 6 months.
Collection of clinical data and serum samples
Basic information, clinical serological indicators, and clinical imaging data were obtained from the medical record management system of Huzhou Central Hospital after informed consent. Negative pressure evacuation tubes containing coagulant were used to collect at least 10 ml of whole blood from all participants. The samples were incubated for 30 minutes at 25
Exosomes extracted from the serum
The serum samples were thawed in a 25
Electron microscopy analysis of the exosome ultrastructure
Exosomes from the serum were fixed in 2.5% ice-cold glutaraldehyde for 2 h and then rinsed for 15 minutes in phosphate buffer solution. This process was repeated 3 times. The samples were post-fixed with 1% OsO4 in cacodylate buffer for 2–3 h, dehydrated with ethanol, and embedded using acetone and embedding reagent for 3–4 hours at room temperature. The embedded tissue was cut with an LKB-1 ultramicrotome, stained with uranyl acetate and lead citrate, and then observed and photographed under the electron microscope (JEM 1011 transmission electron microscope; JEOL, Peabody, MA, USA).
Quantitative real-time RT-PCR
Quantitative real-time RT-PCR assays were performed to evaluate the expression of the lncRNA-HEIH gene. Total RNA from the serum and exosomes was isolated with Trizol reagent (15596-026; Invitrogen, Carlsbad, USA). The First-Strand cDNA Synthesis kit (K1622; Thermo Fisher, USA) was used to synthesize the first-strand cDNA. The GAPDH gene was used as an internal control. Quantitative real-time RT-PCR was performed using a 20
Statistical analysis
SPSS software (V: 16.0; SPSS Inc., Chicago, IL) was used to analyze the data. The data are expressed as the mean
Characteristics of study participants
Characteristics of study participants
The 35 chronic hepatitis C, 22 HCV-induced cirrhosis and 10 HCV-related hepatocellular carcinoma from the Huzhou Central Hospital were recruited in the present study. The chi-squared-test was used to calculate the categorical variables, and the SNK test were used to analyze the data of pairwise comparison among the groups. The risk of disease increased with age and was more common in women than men. The 1b hepatitis C virus was the main genotype in Huzhou. The rate of virus replication did not increase with the disease progression.
Abdominal magnetic resonance imaging of patients. Panels A, B and C show the abdominal magnetic resonance imaging (MRI) results from patients with CHC, HCV-induced cirrhosis and HCV-related HCC, respectively. MRI of patients with CHC showed no major differences compared to images of healthy liver. MRI of patients with HCV-induced cirrhosis showed that the liver volume was reduced, the left and right hepatic lobes were disproportionate, the hepatic contour was matte, and the inter-lobar fissure was widened. MRI of patients with HCV-related HCC showed that the liver surface was uneven, the inter-lobar fissure was markedly widened, the signal in liver was inhomogeneous. There were one or more abnormal signals in the liver.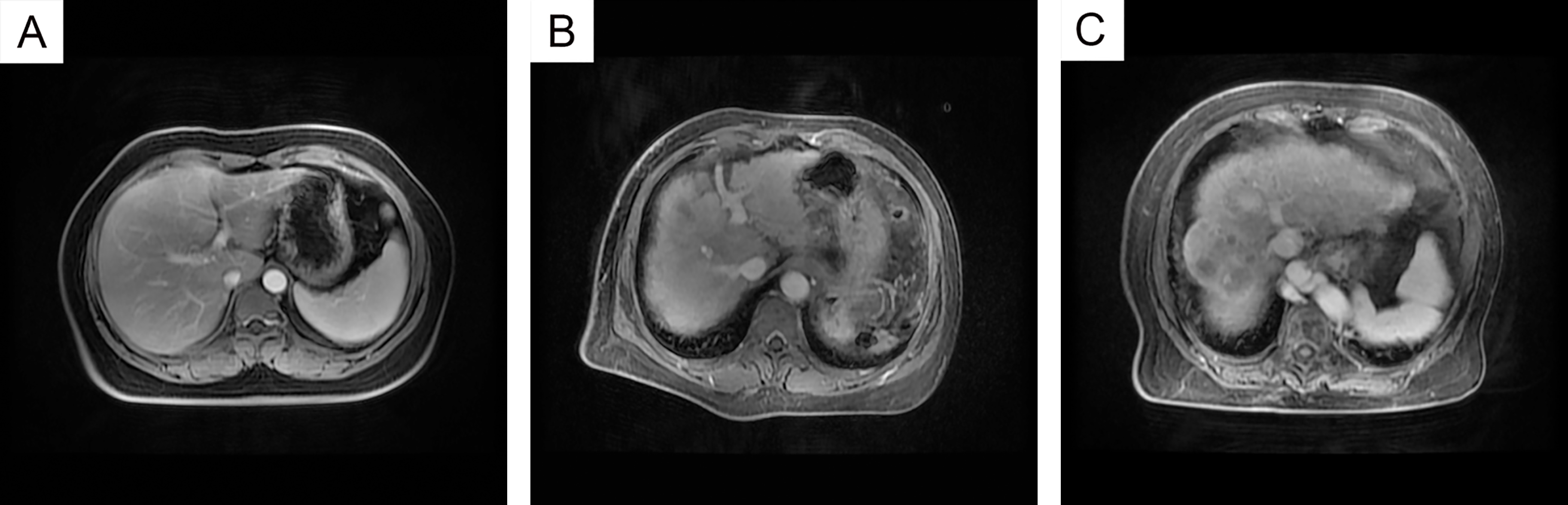
Basic characteristics of the patients
In the present study, 35 CHC, 22 HCV-induced cirrhosis and 10 HCV-related HCC patients were recruited from the Huzhou Central Hospital according to strict inclusion and exclusion criteria. The characteristics of the patients are shown in Table 1. The risk of progressive disease increased with age and was more common in women than in men. Hepatitis C virus 1b was the main genotype in Huzhou. The rate of viral replication did not increase with disease progression. In Fig. 1, panels A, B and C represent the abdominal magnetic resonance imaging (MRI) data from patients with CHC, HCV-induced cirrhosis and HCV-related HCC, respectively. MRI of patients with CHC showed a glossy liver appearance, well-proportioned hepatic lobes, and a clear texture in the intrahepatic vessels. There were no major differences in comparison to MRI from a healthy liver. MRI of patients with HCV-induced cirrhosis showed that the liver volume was reduced, the left and right hepatic lobe were disproportionate, the hepatic contour was matte, and the inter-lobar fissure was widened. MRI of patients with HCV-related HCC showed an uneven liver surface, an obvious widening of the inter-lobar fissure, and an inhomogeneous signal. One or more abnormal signals were detected in the liver. The dynamic enhancement scanning displayed hyperechoic enhancement in the arterial phase and hypoechoic enhancement in the portal and delayed phases.
The liver-related serological indicators
The liver-related serological indicators
The data of liver-related serological indicators including bilirubin, Liver enzymes, blood proteins, fat and cholesterol levels, platelets, blood coagulation function, ferritin and AFP was collated and analyzed. SNK test was used to analyze the data of pairwise comparison among the groups. The different symbol like
Liver-related serological indicators, including bilirubin, liver enzymes, blood proteins, blood fat and cholesterol levels, blood coagulation function, ferritin and AFP, were assessed and analyzed. As shown in Table 2. The levels of bilirubin and liver enzymes in patients with CHC were within the normal range. Compared to patients with CHC, the ALT levels, and ALP (alkaline phosphatase) in patients with HCV-related HCC and GGT levels in patients with HCV-induced cirrhosis and HCV-related HCC were higher. The level of Alb (albumin), HDL (high density lipoprotein) and platelets in patients with HCV-induced cirrhosis and HCV-related HCC was lower. ATPP (activated partial thromboplastin time) in patients with HCV-induced cirrhosis and HCV-related HCC was somewhat prolonged, but the INR (International Normalized Ratio) in HCV-related HCC patients was dramatically decreased. The AFP (alpha fetal protein) levels were higher in patients with HCV-induced cirrhosis and markedly higher in patients with HCV-related HCC.
Ultrastructure of the exosomes
The ultrastructural characteristics of the exosomes were studied to validate the observations made in extracted exosomes from serum by using an electron microscope. As shown in Fig. 2, panels A, B and C represent scanning electron microscope images of exosomes from patients with CHC, HCV-induced cirrhosis and HCV-related HCC, respectively. The ultrastructural characteristics of the exosomes showed a circular or elliptical shape, a size of approximately 30–100 nm in diameter and an integrated membrane system. However, we could not distinguish among the exosomes from patients with different diseases.
Ultrastructure of the exosomes. The ultrastructural characteristics of the exosomes were studied using electron microscopy to validate the observations made in extracted exosomes from serum. Panels A, B and C represent the scanning electron microscope images of exosomes from patients with CHC, HCV-induced cirrhosis and HCV-related HCC, respectively. The exosomes had a circular or elliptical shape, a size of approximately 30–100 nm in diameter and an integrated membrane system.
lncRNA-HEIH expression in serum and exosomes. Panels A, B and C represent lncRNA-HEIH expression in the serum and exosomes from patients with CHC, HCV-induced cirrhosis and HCV-related HCC, respectively. Ten patients were recruited in each group. Quantitative PCR was used to detect the expression of lncRNA-HEIH. Panel D shows the ratio of lncRNA-HEIH expression in serum versus exosomes in each example. The mean ratio in patients with CHC, HCV-induced cirrhosis and HCV-related HCC was 0.78, 1.85 and 3.62, respectively. And the IQR (Inter-Quartile Range) in patients with CHC, cirrhosis and HCC was (0.66–0.97), (1.77–1.98) and (2.91–3.88), respectively.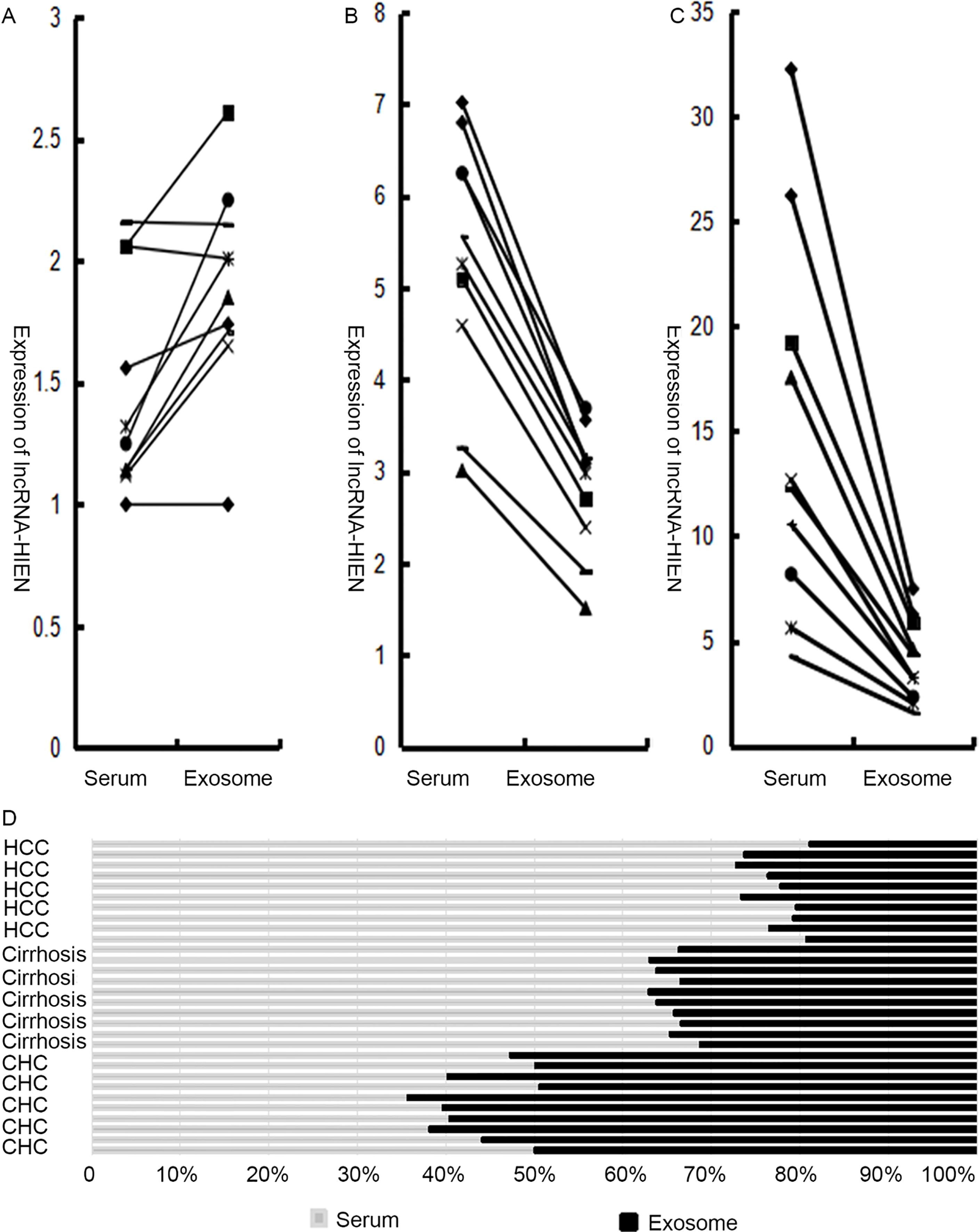
Panels A, B and C in Fig. 3 represent lncRNA-HEIH expression of serum and exosomes from the patients with CHC, HCV-induced cirrhosis and HCV-related HCC, respectively. Ten patients were recruited in each group. lncRNA-HEIH expression in serum from the group of HCV-related HCC patients was higher than that in HCV-induced cirrhosis patients, and both were higher than that in CHC patients. According to statistical analysis (q-test), there was a significant difference among the groups (
Discussion
CHC is a blood-borne infectious disease that often lacks symptoms and can result in chronic liver inflammation, cirrhosis and hepatocellular carcinoma (HCC) over several decades [30]. CHC develops into hepatocellular carcinoma through irreversible processes and thus is a serious public health problem. CHC was included in the chronic and unusual disease administration of basic medical insurance in Huzhou city. Hepatitis C patients were concentrated in our hospital and received free medical treatment. The focus of hospitalization was on HCV-induced cirrhosis and HCV-related HCC because CHC patients often lacked symptoms.
The pathologic examination had many limitations; for example, it was an invasive procedure and small lesions were hard to obtain. Imaging studies demonstrate some characteristic manifestations of the disease, and imaging examinations can provide grounds for clinical diagnosis in the process of HCV transformation into liver cirrhosis or hepatocellular carcinoma. However, atypical lesions and small lesions are often misdiagnosed when only imaging tools are used. Large studies have indicated that liver-related serological markers might provide some indication of the disease diagnosis and prognosis.
Serological indicators, including bilirubin, liver enzymes, blood proteins, blood fat and cholesterol levels, platelets, coagulation function, ferritin and AFP, were investigated. The changes in the levels of ALT, GGT, HDL, INR, Alb and AFP in patients with HCV-induced cirrhosis and HCV-related HCC were statistically significant. Most of these changes were consequences rather than causes of the disease.
Immune modulation is considered to be the most important mechanism underlying HCV as well as the development of liver cirrhosis and hepatocellular carcinoma [31]. The gene variations, gene expression and molecular signal transduction pathways in the immune reactions are involved in the susceptibility to and prognosis of HCV-induced HCC [32]. Previous studies carried out in HCC cell lines and HCC tissues reported that lncRNA-HEIH might provide a potential target in the diagnosis and therapy of HCC [16, 17, 18]. By studying the expression of lncRNA-HEIH in the serum of HCV, HCV-induced cirrhosis and HCV-related HCC patients, we found that the lncRNA-HEIH may also serve as a potential target in serum. Some important clinical values relevant to the early diagnosis, therapy, monitoring and prognosis of HCV-related HCC were identified.
We found that lncRNA-HEIH expression in the serum from patients with HCV-related HCC was higher than that in serum from CHC patients. The detection of lncRNA-HEIH in serum suggests that it can be utilized as a noninvasive serum-based potential biomarker for the diagnosis and screening of HCV-related HCC. However, the ratio of lncRNA-HEIH expression in serum versus exosomes was lower in patients with HCC than in CHC patients. This finding may be an abnormal phenomenon. Nevertheless, the exosomes in serum not only act as gene transfer vectors [33] but also play a regulatory role in cancer metastasis [34]. lncRNA-HEIH expression in serum and exosomes was higher but the ratio of lncRNA-HEIH expression in serum versus exosomes was lower in HCC. Did lncRNA-HEIH in exosomes release into serum? To explain this phenomenon, we raises bold hypothesis that the exosomes in serum have two functions: release and storage. lncRNA stored in exosomes cannot regulate downstream gene expression until the exosomes release lncRNA into the serum. The data in the present study have not yet be reached at this hypothesis and it requires a significant amount of experimental data to support the interesting hypothesis.
It is not difficult to diagnose CHC, liver cirrhosis or HCC using a combination of pathologic examination, imaging and serum biochemistry indicators in clinical practice. However, it remains of great importance to elucidate the mechanism by which CHC develops into HCC. On the one hand, not all HCV-infected individuals are destined to develop serious liver cirrhosis or HCC, and it is very difficult to predict the outcome. On the other hand, there is a shortage of effective interventions to inhibit the development of CHC into liver cirrhosis or HCC. The clinical serological indicators and differential expression of lncRNA-HEIH in serum and serum-to-exosomes in the process of CHC transformation into HCC were investigated in the present study. Elucidating the mechanisms of CHC development may provide a novel way to predict the risk of disease progression and lead to new preventative methods. Moreover, these findings provide new insights into the development of HCC.
Expect for the research findings, there are also research limitations, such as lack of pathogenesis study, data limitation, and inadequate interviews, etc. More, follow-up visits should be paid and more molecular experiment should be done to make explicit the associations of exosomes with liver-related outcomes.
Footnotes
Acknowledgments
We thank the patients and volunteers for their contributions to the imaging and sample collection. This work was supported by the Public Welfare Technology Application Research Program of Huzhou (No. 2016GYB14) and the Natural Science Foundation of Zhejiang province (No. LY17H100002).
Conflict of interest
The authors declare no conflicts of interest.
