Abstract
BACKGROUND:
Prostate cancer (PCa) is the second most common cancer in men worldwide. Currently, prostate-specific antigen (PSA) test and digital rectal exam are the main screening tests used for PCa diagnosis. However, due to the low specificity of these tests, new alternative biomarkers such as deregulated RNAs and microRNAs have been implemented.
OBJECTIVES:
Aberrant expressions of small heterodimer partner gene (SHP, NR0B2) and mir-141 are reported in various cancers. The aim of this study was to investigate the SHP and miR-141 expression level in tissue samples of prostate cancer.
METHODS:
The expression level of SHP gene and miR-141 was assessed by real time PCR and their relative amounts were calculated by the
RESULTS:
The miR-141 was significantly up-regulated in the samples of metastatic tumors compared to localized tumor samples (
CONCLUSIONS:
The data obtained in our study confirm the involvement of miR-141 in PCa progression and metastasis. These effects could be mediated by AR via down-regulation of its co-repressor protein, i.e., SHP.
Introduction
Prostate cancer (PCa) is the second most common cancer in men, worldwide [1]. Based on an estimate by the American Cancer Society, there were 180,890 new cases and 26,120 deaths due to the disease in the United States in 2016 [2]. Although the frequency of PCa is lower in other regions such as middle-east, the number of PCa cases in Iran has been reported to be on rise [3]. The most prevalent risk factors related to PCa are old age, race, and family history of the disease [4]. PCa is a slow progressive disease, most often with no specific signs or symptoms [5]. Therefore, screening and diagnosis of PCa at early stages are of high importance for the management of the disease [6]. Currently, prostate-specific antigen (PSA) test and digital rectal exam are the major screening tests used for PCa diagnosis. However, due to the low specificity of these tests, new alternative biomarkers such as deregulated RNAs and microRNAs have been implemented [7, 8].
MicroRNAs are short non-coding RNAs with a length of 19–22 nucleotides that play a crucial role in regulating the post-transcription of gene expression [9]. Recent investigations have shown that microRNAs contribute to cancer pathogenesis [10, 11, 12]. Several studies have also demonstrated a relationship between the changes in the expression of microRNAs and PCa, as well as other cancers [13, 14, 15, 16, 17].
MicroRNA-141 (miR-141) is one of the microRNAs that have attracted attention as potential biomarkers indicating the progression of PCa from benign prostate hyperplasia (BPH) [18]. miR-141 is a member of miR-200 family, playing a remarkable regulatory role in the epithelial-to-mesenchymal transition (EMT) process [19]. Moreover, as one of the most important regulatory microRNAs, it affects the androgen receptor (AR) signaling pathway via regulating the expression of AR gene. Although various molecular pathways have been investigated in PCa, androgen receptor pathway is directly involved in the pathogenesis of prostate cancer [20, 21].
Small heterodimer partner (SHP), also known as SHP1 and NR0B2, is a member of the nuclear receptor (NRs) family that lacks DNA-binding domain [22]. Considering their remarkable biological role, NRs have been suggested as potential cancer biomarkers as well as candidates for therapeutic targets [23, 24]. In vitro studies have shown that SHP acts as a co-repressor for the AR cascade and serves as a target for miR-141. This decomposition is carried out by a selective hybridization of miR-141 at 3
Previous studies have indicated aberrant expression of SHP gene in different cancers including hepatocellular carcinoma and breast cancer [27, 28, 29]. In the present study, we investigated the expression profile of miR-141 and SHP gene in androgen receptor pathway in tissue samples of PCa patients. To our knowledge, these data represent the first study to evaluate changes of SHP gene expression in PCa clinical samples.
Materials and methods
Tissues collection
In the present case-control study, 68 prostate tissue samples (including 30 tumor tissues, 30 BPH tissues, and 8 tumor-adjacent normal prostate tissues) were examined. The samples were collected from the patients, referred to Hashemi Nejad Kidney Center in Tehran, Iran during May 2015 to March 2016 in collaboration with the Oncopathology Research Center at Iran University of Medical Sciences. Each patient provided a written informed consent form approved by the Ethics Committee of the Research Center. Demographics and clinicopathological features of each patient, including age, PSA, and Gleason score were recorded. None of the patients had history of chemotherapy, radiotherapy, or hormone therapy. The mean patients’ age were 61.16
Bioinformatic analysis for the prediction of has-miR-141-3p target genes
To predict the most relevant gene targets for miR-141 in silico, we investigated the has-miR-141-3p targets by online prediction tools including the miRanda (http://www.microrna.org/), miRWalk 2.0 (http://zmf. umm.uni-heidelberg. de/apps/zmf/mirwalk2/), miRDB (http://www.mirdb.org/miRDB/), TargetScan (http://
The sequence of the primers used for SYBR Green real-time PCR assays
The sequence of the primers used for SYBR Green real-time PCR assays
Oligonucleotide primers, probes and stem-loops used for the analysis of microRNA expression by TaqMan real-time PCR assay
For the extraction of total RNA containing microRNAs, 100 mg of tissue sample was lysed using the Qiazol lysis reagent (Qiazol, USA) as recommended by the manufacturer’s instruction with minor modification. The concentration and quality of the extracted RNA was determined using a NanoDrop spectrophotometer (Implen, Munich, Germany). A small number of samples were excluded from the study because of low quality of their RNA. The extracted RNAs were used directly for cDNA synthesis or preserved at
Quantitative real-time PCR
Stem loop and primer/probe design was performed based on has-miR-141-3p sequence obtained from the miRBase (http://www.mirbase.org) as the reference microRNA database. Also, the reference sequence of SHP (NM_021969.2), GAPDH (NM_001289746.1) and U6 (NR_125730.1) genes were acquired from the NCBI website (https://www.ncbi.nlm.nih.gov). Theprimers were designed using AlleleID
Real-time PCR assay for SHP gene expression was performed using SYBR Green I dye. Each reaction contained 10
Validated SHP/miRNA interactions assessed by miRWalk 2.0. online prediction tool
Validated SHP/miRNA interactions assessed by miRWalk 2.0. online prediction tool
MIMATid; microRNA accession ID at miRbase data base PMID; Related reference ID at NCBI/PUBMED.
Cumulative scores for the interaction between has-miR-141-3p and SHP gene according to the data obtained from indicated online prediction tools
The relative expression of miR-141 in metastatic tumors compared to the localized tumor samples
TRG: Target gene; REF: Reference gene.
IHC was performed in the Pathology Department of Iran University of Medical Sciences (IUMS, Tehran, Iran). Twenty Paraffin-embedded tissue blocks including 10 tumors and 10 BPH samples were obtained from the patients. Four-micron slice cuts were prepared from the paraffin blocks, and a slice cut of healthy human liver tissue was considered as a positive control. IHC slides were prepared as follows: Initially, the slides were placed in Tris-EDTA buffer (pH 9) and autoclaved at 120
Statistical analysis
Relative expression of the study genes was investigated using the REST 2009 software (version 2.0.13) according to the threshold cycle (Ct) comparison method (
Results
MicroRNA gene target prediction
Assessment by miRWalk 2.0 revealed that has-miR-141-3p was significantly associated with androgen receptor pathway (
The results of real-time PCR assay for miR-141 expression in clinical tissue samples from the PCa patients with metastatic, localized tumors or BPH. A: Box and whiskers plot representing the relative expression of has-miR-141-3p normalized to U6 mRNA in localized samples compared to metastatic tissues. B: Scatter plot shows the mean dispersion of has-miR-141-3p expression 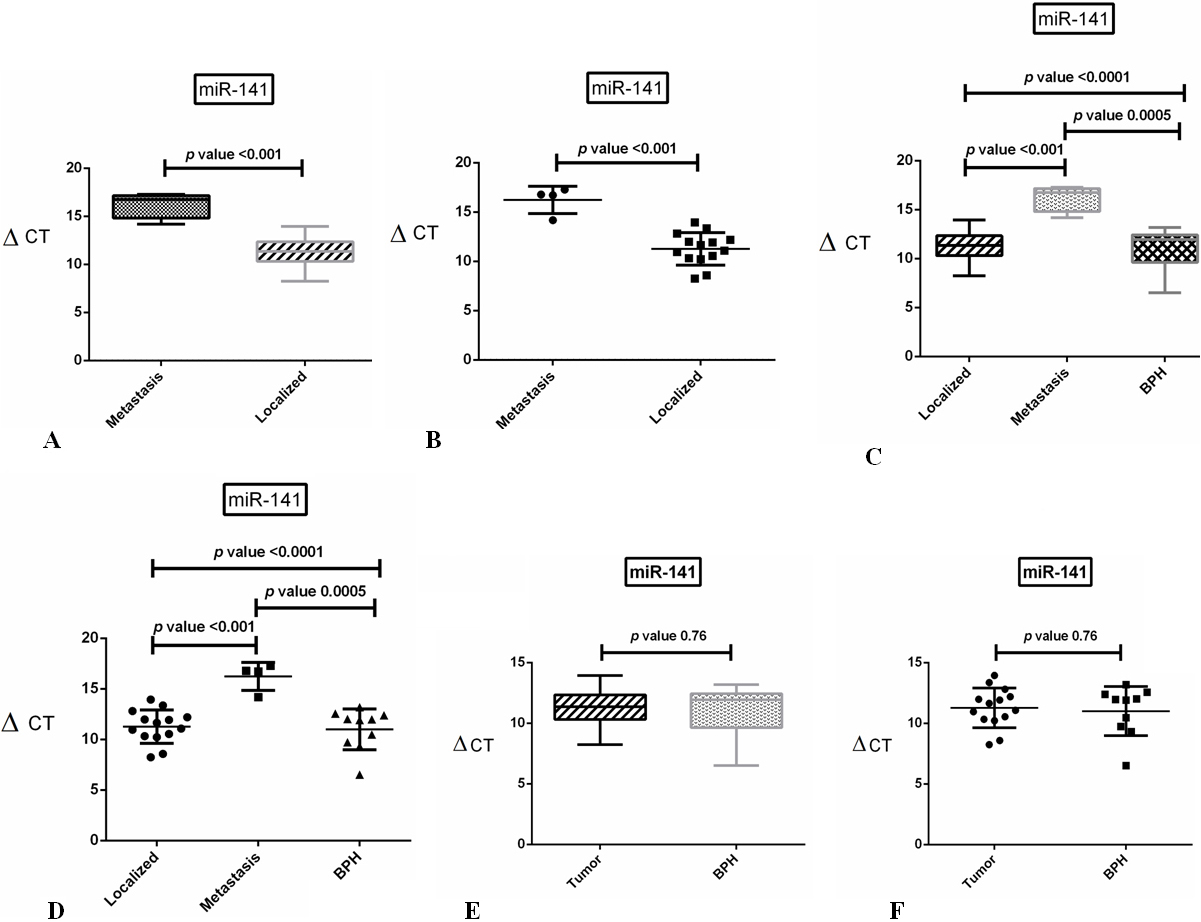
miR-141 was significantly up-regulated in samples of metastatic tumor as compared to the samples from localized tumor. In metastatic tumor samples included in this study, a significant difference (
The relative expression of miR-141 and SHP gene in PCa clinical samples
The relative expression of miR-141 and SHP gene in PCa clinical samples
TRG: Target gene; REF: Reference gene.
The comparison of SHP gene expression level in PCa tumor, tumor-adjacent normal and BPH tissue samples. A: The box plot represents the relative expression of SHP gene normalized to GAPDH mRNA in tumor samples compared to BPH. B: The scatter plot shows the mean dispersion of SHP expression 
SHP gene expression level was significantly different in PCa tumor compared to BPH tissue samples (
Immunohistochemichal staining of SHP protein in PCa tumor and BPH tissues. A (25
ROC curve analysis for the evaluation of diagnostic value of SHP expression in prostate cancer. Data were derived from the results of SHP expression changes and their capability for the discrimination between PCa and BPH patient’s samples. A: ROC curve analysis for SHP expression at mRNA level (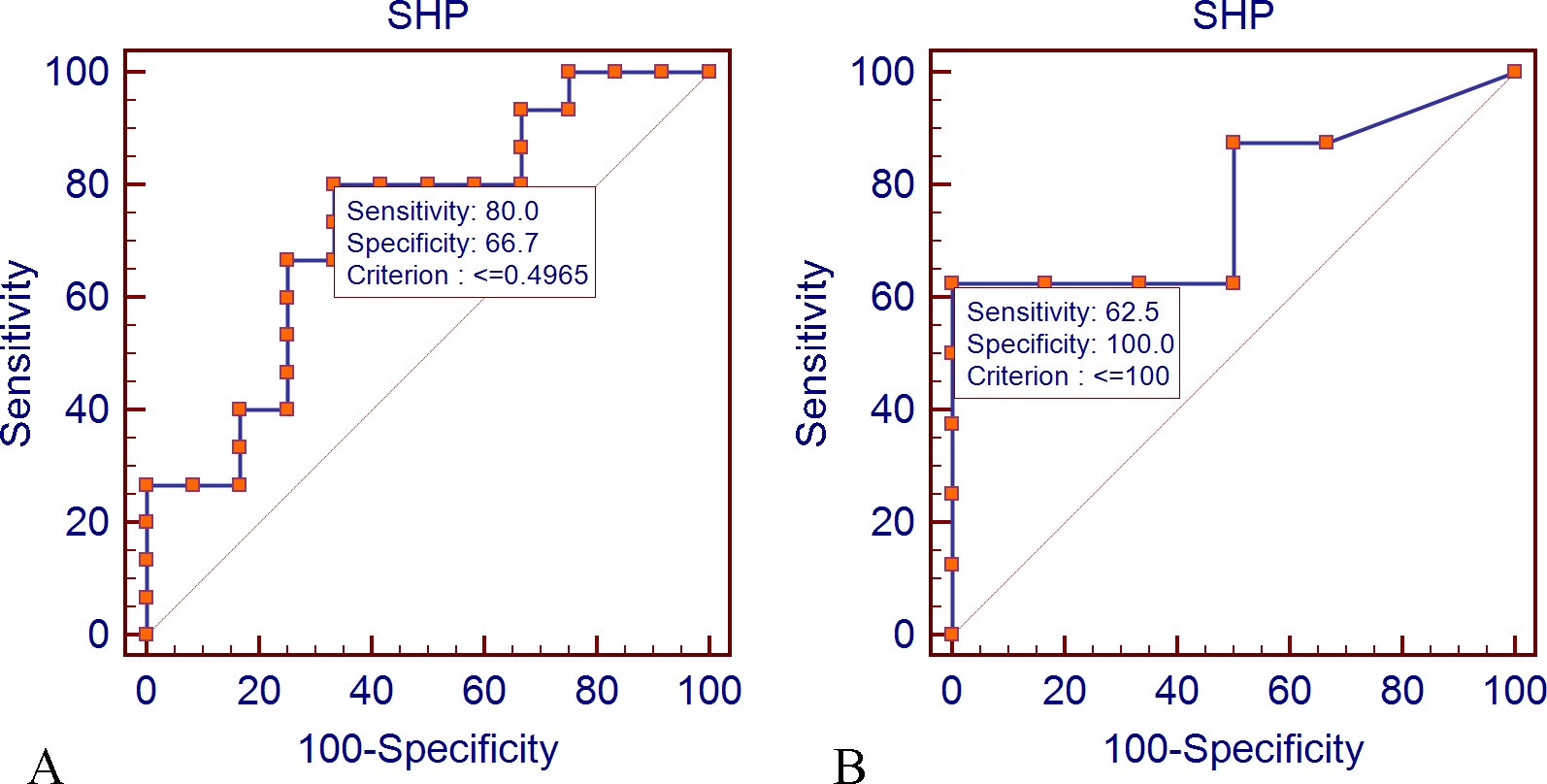
IHC analysis was used to investigate the expression level of SHP protein in the PCa and BPH tissue samples. Following staining, the cells were evaluated via light microscopy and the data obtained for SHP by immunohistochemical semiquantitation method was reported as H-score. The intensity of stained tumor cells was marked as 0 (none), 1 (weak), 2 (moderate), and 3 (strong). H-score was determined as the intensity of the stained cells multiplied by the percentage of positive cells. At least 500 tumor cells from each slide were evaluated. Quantitative IHC scores determined by SHP protein expression analysis were 91.25
Correlation between miR-141 and SHP gene expression and clinicopathological features of the patients
The assessment of correlation between miR-141 expression and clinicopathological characteristics in the PCa patients was performed using chi-square test. The results showed that miR-141 expression was correlated with the metastatic status of the tumor samples, but not with other features such as PSA level and Gleason score of the patients. Furthermore, there was a linear reverse correlation between the overall expression of miR-141 and SHP among the tumor samples. Chi-square analysis showed no relationship between SHP and clinicopathological characteristics, including PSA, Gleason score, and the metastasis of the tumor in the patients.
Discussion
Early screening and diagnostic tests for PCa need to be improved by implementation of more specific alternatives [33, 34]. To this end, the use of specific biomarkers in combination with PSA can contribute to the screening and diagnosis of PCa patients [35]. Currently, with better understanding of the role of signaling pathways in molecular pathogenesis of PCa, multiple biomarkers at either RNA or protein level have been suggested for early detection or the assessment of the disease prognosis [36]. For this purpose, the deregulation of various microRNAs including miR-141 has been reported in PCa patients [9, 37]. Mitchell et al. found a significant difference in miR-141 expression level between the patients with metastatic Vs localized PCa [38]. Furthermore, Gonzales et al. reported that miR-141 expression level has a significant correlation with the disease stage and the PSA levels in metastatic PCa patients [39]. Another study showed increased miR-141 expression levels in clinical samples of the patients with PCa [40]. Nevertheless, Kachakova and co-workers reported the down-regulation of miR-141 expression level in the plasma of PCa patients compared to BPH patients [41]. Also, Agaoglu et al. found no difference in the expression level of miR-141 between normal individuals and PCa patients but significant difference was observed between metastatic and non-metastatic PCa patients [42]. Consistent with the previous reports, we showed miR-141 expression level was deregulated in the PCa tissue samples. Our result revealed a significant difference in the expression level of miR-141 between metastasis and non-metastasis tissue samples while there was a slight increase in the level of miR-141 expression in PCa compared to BPH tissues. Similar data regarding the down-regulation of miR-141-3p expression in early PCa and its up-regulation in advanced tumors (Gleason score
Multiple cancer-related genes have been suggested as miR-141 targets based on in silico or in vitro experiments, but only few studies have been carried out to validate their clinical significance. Some of these targets are NRs with aberrant expression in the signaling pathways involved in PCa [45]. SHP belongs to NRs proteins and is located downstream to the AR signaling pathway [46]. Xiao et al. evaluated the effect of miR-141 on SHP expression level in epithelial cancer cell lines [26]. They reported that SHP gene expression level was down-regulated by miR-141 in prostate cancer epithelial cell lines. Here, for the first time, we investigated the aberrant expression of SHP gene in the clinical samples of PCa and BPH. At mRNA level, we observed down-regulation of SHP expression in PCa tissue compared to BPH samples. Interestingly, this finding was consistent with the data at protein level obtained by IHC assessment on comparable tissue samples. Also, ROC curve analysis was used to investigate the ability of SHP mRNA and protein expression for the discrimination of prostate cancer and BPH samples. ROC curve results indicated that the sensitivity and specificity of the assay were within acceptable ranges (
MiR-141-3p is androgen regulated in PCa and modulates transcriptional activity of AR by targeting SHP. A recent study has revealed that miR-141 can also directly affect AR expression by targeting its transcript, which makes the contribution of this microRNA in PCa carcinogenesis more difficult to elucidate [47].
In conclusion, the data achieved in our study confirm the involvement of miR-141 in PCa progression and metastasis. These effects can be mediated by AR via down-regulation of its co-repressor protein, i.e., SHP. However, the expression profile of SHP in PCa and its importance for modulating androgen receptor signaling pathway has yet to be fully described.
Footnotes
Acknowledgments
This paper represents the results of a PhD thesis in Molecular Medicine performed by M. Khorasani. This project has been supported in part by a research grant from Qazvin University of Medical Sciences.
