Abstract
BACKGROUND:
Arylsulfatase B (ARSB) removes the 4-sulfate group from chondroitin 4-sulfate (C4S) and dermatan sulfate and is required for their degradation. Prior work showed that ARSB immunohistochemical scores were lower in malignant prostate tissue, and were associated with higher Gleason scores and recurrence.
OBJECTIVE:
This study aims to confirm that ARSB immunostaining of prostate tissue obtained at the time of radical prostatectomy is prognostic for prostate cancer recurrence.
METHODS:
Intensity and distribution of ARSB immunostaining were digitally analyzed in a large, well-annotated, prostate cancer tissue microarray (TMA). Scores were calculated for stroma and epithelium and compared for 191 cases, including 36 recurrences, defined as PSA
RESULTS:
Epithelial scores were significantly lower in the recurrences (
CONCLUSIONS:
Study findings validate previous findings and provide further evidence that lower ARSB is associated with prostate cancer recurrence. Additional studies are required to assess if there are specific cutoff values that may help predict recurrence.
Keywords
Introduction
Arylsulfatase B (ARSB; a N-acetylgalactosamine-4-sulfatase) is the enzyme that removes 4-sulfate groups from the non-reducing end of the sulfated glycosaminoglycans (GAGs) chondroitin 4-sulfate (C4S) and dermatan sulfate (DS). ARSB was considered to be only a lysosomal enzyme, but has been shown to be present in the cell membranes of epithelial and endothelial cells [9, 17, 24]. In addition to association with the congenital, lysosomal storage disease Mucopolysaccharidosis VI, we have reported decline in ARSB in prostate, mammary, melanoma, and colon malignancies and in cystic fibrosis [7, 10, 13, 15, 16, 23]. Since chondroitin 4-sulfate acts as a receptor for the malarial parasite, and lower sulfation facilitates parasite adhesion, regulation of ARSB activity may be of evolutionary importance in protection from infectious disease [12]. Activity of ARSB requires post-translational modification by the formylglycine modifying enzyme and molecular oxygen, and hypoxia reduced ARSB activity [5, 6, 18]. ARSB activity is also reduced by exposure to increased concentrations of chloride and phosphate and to estrogen, indicating responsiveness to ambient conditions [8, 13, 14]. Acquired ARSB deficiency mediates transcriptional events by reduced binding of galectin-3 or by increased binding of SHP2 to the more highly sulfated C4S present when ARSB declines [19, 20, 21]. Increased binding of other vital molecules to more highly sulfated C4S has also been shown, including increased binding of Interleukin-8, high molecular weight kininogen, and bone morphogenetic protein-4. In addition, increased aerobic glycolysis, the Warburg effect, occurs when ARSB is deficient [22]. These findings demonstrate that decline in ARSB leads to profound effects on critical cell processes related to cell signaling, transcription, metabolism, inflammation, proliferation, and differentiation.
In prostate, ARSB immunostaining and activity were reduced in malignant compared to normal tissue, and intensity of immunostaining was less in cases with higher Gleason scores [10]. An evaluation of paired prostate cancer cases, matched by age,
A possible mechanism by which ARSB acts as a tumor suppressor involves the extracellular matrix proteoglycan versican, a chondroitin sulfate proteoglycan, which was previously shown to be a marker of poor prognosis in prostatic malignancy [2, 3, 4, 10, 19]. Increased mRNA expression of versican followed ARSB silencing in cultured human prostate epithelial and stromal cells. This increase was mediated by chondroitin 4-sulfation, galectin-3, and AP-1. Cell proliferation, measured by BrdU incorporation, increased following exposure to exogenous epidermal growth factor (EGF) when ARSB was reduced, and was attributable to the increased availability of cellular EGF receptor (EGFR), due to inhibition of binding with the EGF-like motifs of versican when C4S was increased.
With this background, further investigation of the role of ARSB as a tumor suppressor and of decline in ARSB as a possible biomarker for more aggressive prostate cancer was undertaken. In this communication, we report the analysis of ARSB immunohistochemistry in a well-annotated prostate cancer array which was designed to identify biomarkers of prostate cancer recurrence.
Materials and methods
The prostate cancer tissue array was developed as part of the Prostate Cancer Biorepository Network (PCBN) and consisted of prostate tissue from 217 patients [1, 11]. The array is described as an intermediate-density tissue microarray, containing tumor and non-neoplastic samples from localized prostate cancercases, as well as benign samples from cases of benign prostatic hyperplasia (BPH). The cases were all obtained from the New York University site of the PCBN, in accord with approval from the Institutional Review Board and with established procedures. The microarray of 204 cases of localized prostate cancer from radical prostatectomies and 13 BPH cases from suprapubic prostatectomies was assembled using an MTA 1 microarrayer (Beecher instruments, Sun Prairie, Wisconsin), and was constructed over 5 blocks. Each case was arrayed in quadruplicate with a total of 4 tumor cores per case over each of the blocks (from A–D). The cores were 0.6 mm in diameter and 5
ARSB Epithelial H-Scores in association with biochemical recurrence, Gleason Score, PSA, age, and modified CAPRA. A. Lower epithelial H-scores were associated with biochemical recurrences in 191 prostate cancer cases (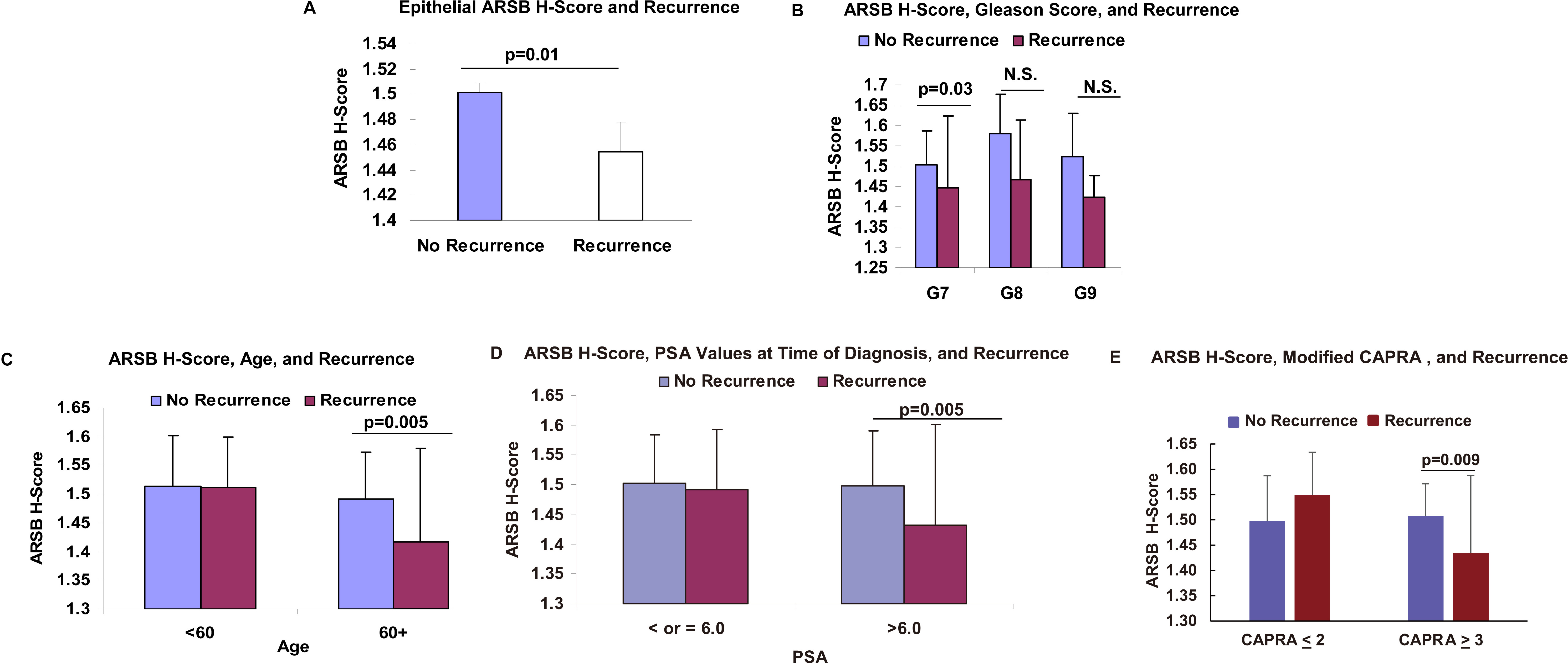
Slides were immunostained with ARSB polyclonal rabbit antiserum (Open Biosystems, ThermoFisher Scientific, Huntsville, AL, USA; 1:100), as previously [10, 17, 24]. Digital scanning of slides was performed at 200x with Aperio ScanScope
H-scores were matched with accompanying de-identified patient data, including age, race, Gleason score in order of secondary or primary score and total score, Prostate-Specific Antigen (PSA) at the time of diagnosis, pathological TNM stage, description of surgical margins, extracapsular extension, seminal vesicle involvement, vital status, and clinical outcome (either no recurrence or biochemical recurrence defined as PSA
Epithelial H-scores and recurrence
Of 204 prostate cancer cases on the arrays, immunostaining of 191 prostate cancer cases had evaluable H-scores, no residual tumor from the time of prostatectomy, and followup clinical data. The sections on the array which could not be analyzed were folded, torn, or otherwise damaged, so that digitized analysis could not be performed. The H-scores for epithelial, stromal, and combined were tabulated and compared for the cases that relapsed vs. the cases that did not relapse. The mean epithelial H-scores were significantly higher for No Recurrence (1.50
ARSB Immunostaining in Gleason 9 cancer images shows decline in epithelial ARSB membrane staining in association with recurrence. A–C. In the representative No Recurrence Gleason 9 cases, epithelial membrane retains prominent cell membrane immunostaining for ARSB. Marker is 40 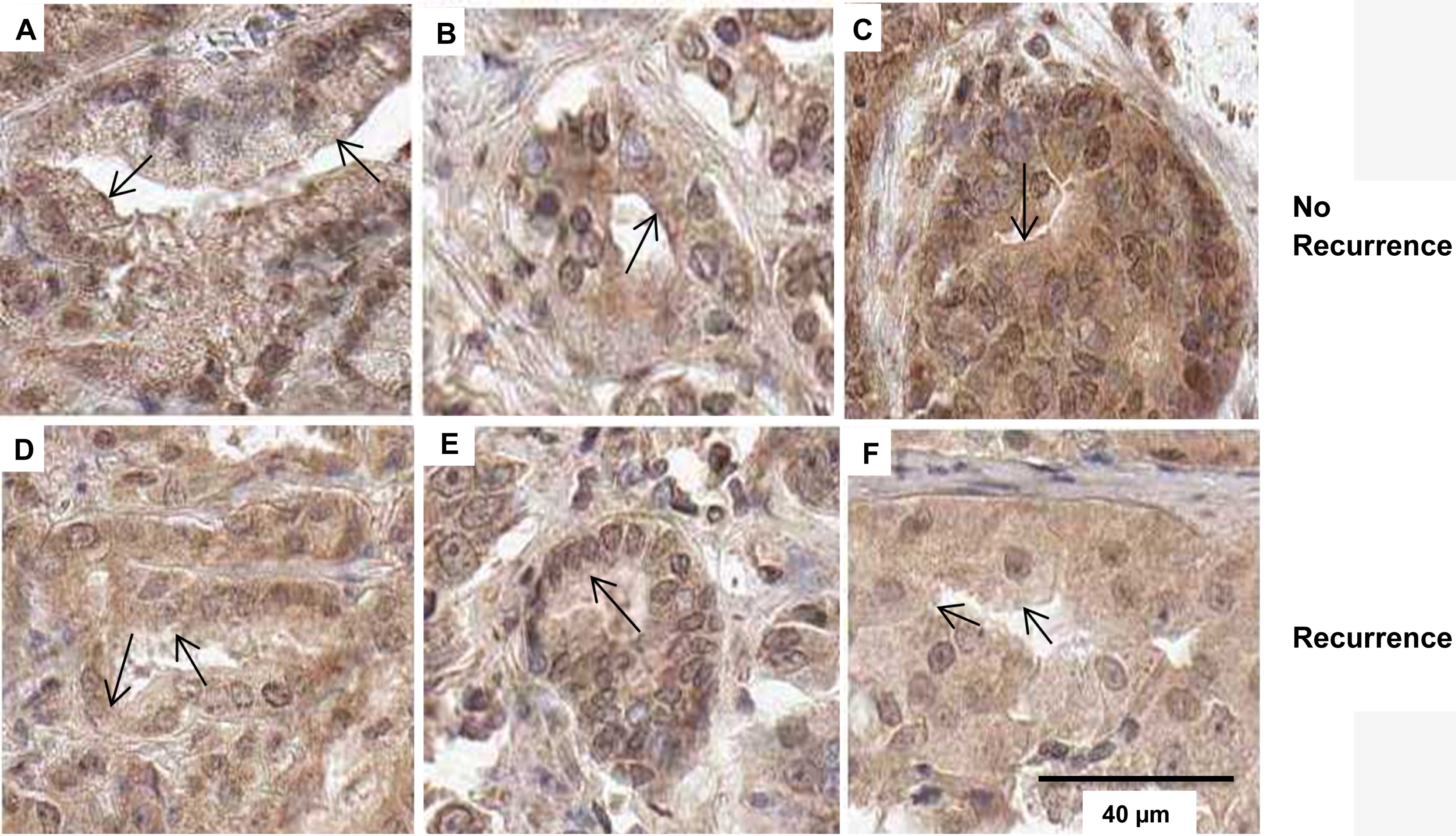
Among Gleason 7 cases, the average epithelial score for No Recurrence was significantly higher than for Recurrence (1.50
The average epithelial score for patients over 60 years old was significantly higher for No Recurrence: (1.49
The average No Recurrence epithelial H-score for patients with PSA at diagnosis greater than 6.0 ng/ml was significantly higher (1.50
The percentage of tumor in the cores was not included in the original dataset, so was arbitrarily set at either 0 (for
Other statistical tests of relationships between combined epithelial and stromal ARSB H-scores or stromal ARSB H-scores and clinical parameters were not significant.
In a multifactorial analysis with the established risk factors of age, Gleason score, tumor stage, and PSA, addition of the ARSB epithelial H-score did not improve the prediction of recurrence. Only the Gleason score was an independent predictor of recurrence in this model.
ARSB epithelial membrane immunostaining staining in Gleason 9 cores
Analysis of the images of Gleason 9 cancers indicates that presence vs. absence of luminal membrane staining of the epithelial cells may be a distinguishing feature that is associated with worse prognosis (Fig. 2). Epithelial membrane staining is darker and more continuous in the Gleason 9 cases with No Recurrence (Fig. 2A–C), than in the recurrences (Fig. 2D–F), in which the epithelial membrane staining is less intense and more fragmented.
Discussion
The study findings indicate that in prostate cancers sampled at prostatectomy, lower epithelial ARSB expression is associated with biochemical recurrence of prostate cancer. This result confirms findings of lower ARSB in association with biochemical recurrences in 44 paired cases reported previously [24]. Lower epithelial H-scores in the 204 cases of this report were also associated with specific clinical features in the recurrent cases, including age over 60, Gleason score of 7, and initial PSA over 6.0 ng/ml. ARSB showed no independent association when several clinical predictors were controlled simultaneously in a multivariable model. Further evaluation in a larger number of patients may help to identify specific cutoff values for H-scores, or specific cellular localization sites, and may help in the development of a risk of recurrence model that includes ARSB. A more complex model will also need to consider the impact of increasing age on decline in ARSB scores. These considerations will help in the assessment of how ARSB determinations can aid in the prediction of biochemical recurrence or non-recurrence.
Previously, in frozen prostate tissue samples obtained at the time of diagnosis, the ARSB activity was markedly less in malignant than in normal tissue [10]. The measurement of activity may provide an alternative approach to assessment of ARSB, since it does not require digitized imaging and quantitative analysis of microscopic images. Acquisition and normalization of the information obtained from digitized images may be obstacles to their routine usefulness in the clinical setting. Small volumes of frozen prostate tissue can be used for determination of ARSB activity using spectrophotometric or fluorometric methods [10, 14]. Alternatively, the analysis of epithelial membrane immunostaining for ARSB may provide a shortcut assessment of ARSB decline and may inform prognosis in prostate cancers. The mRNA expression of ARSB may not be informative, since ARSB must undergo post-translational modification for activation [5]. For this reason, genome-wide association studies may not identify ARSB as reduced in prostate cancers. Further investigation of the role of ARSB as a tumor suppressor and biomarker will help to clarify the role of ARSB in prostate cancer biology and in the prediction of prostate cancer recurrence.
Footnotes
Conflict of interest
The authors have no conflicts of interest with regard to the content.
