Abstract
BACKGROUND:
Globo H is a tumor-associated carbohydrate antigen exclusively expressed in cancer cells rather than normal tissue. Globo H has been found on many cancers of epithelial origins, and become an attractive target for cancer vaccine.
OBJECTIVES:
We aimed to study the expression of Globo H in non-small cell lung cancer (NSCLC) patients, and correlated its expression with common driver mutations, clinical outcomes, and status of immune checkpoint, programmed death-ligand 1 (PD-L1).
METHODS:
The study enrolled 228 patients with surgically resected stage I NSCLC, including 139 patients with adenocarcinoma (ADC) and 89 patients with squamous cell carcinoma (SqCC). Using immunohistochemistry, tumors with moderate to strong membranous staining in
RESULTS:
Positive Globo H expression was detected in 88 of the 228 (38.6%) patients. These included 51 of 139 (36.7%) patients with ADC and 37 of 89 (41.6%) patients with SqCC. Positive Globo H expression was significantly associated with EGFR mutation and PD-L1 expression in the ADC group, and PI3KCA overexpression in the SqCC group. The survival analysis showed that Globo H expression was not an independent prognostic factor in stage I NSCLC.
CONCLUSIONS:
Globo H expression was correlated with specific driver mutations in ADC and SqCC NSCLC tumors, as well as PD-L1 status. Immunotherapy targeting Globo H may have potential application in lung cancer treatment.
Keywords
Introduction
Aberrant glycosylation in tumor cells is involved in several biological processes that occur during tumorigenesis, including inflammation, immune surveillance, cell-cell adhesion, cell-matrix interaction, signaling transduction and cellular metabolism [1]. Immunogenic carbohydrate epitopes are expressed exclusively in tumor cells and generated through aberrant glycosylation [2], These carbohydrate epitopes are so-called tumor-associated carbohydrate antigens (TACAs) which have been studied as potential targets of cancer vaccines [3]. Globo H, a hexasaccharide glycosphingolipid TACA, was first identified in a human breast cancer cell line MCF-7 [4, 5]. The expression of Globo H has been reported in various neoplasms, including pancreatic, breast, gastric, and endometrial cancers [6, 7]. Cancer vaccines targeting Globo H have shown promise in several clinical trials [8, 9], especially a large phase II/III clinical trial exhibiting the efficacy of Globo H-based vaccine in patients with metastatic breast cancer [10]. Therefore, Globo H may be considered as an ideal immunotherapeutic target.
Treatment strategies for non-small cell lung cancer (NSCLC) have moved from platinum-based chemo-therapy and molecular target therapy to immunotherapy [11], especially immune checkpoint inhibitors targeting programmed death-ligand 1 (PD-L1) and PD-1 axis [12, 13, 14, 15, 16], Besides PD-1/PD-L1 inhibitors, a number of studies have also investigated the efficacy of cancer vaccines in lung cancer, including MAGE-A3 targeting melanoma associated antigen A3 (MAGE-A3) [17], tecetomide (L-BLP25) targeting mucin 1 [18], and CIMAvax targeting epidermal growth factor (EGF) [19]. However, Globo H expression in NSCLC and its value as a potential target for cancer vaccine have not been well studied.
This study aimed to evaluate the expression of Globo H in a large cohort of resected stage I NSCLCs, and to assess its association with clinicopathological features, clinical outcomes, common driver mutations of lung cancer, and the immune checkpoint, PD-L1 expression. The driver mutations included EGFR, Kirsten rat sarcoma viral oncogene homolog (KRAS), v-raf murine sarcoma viral oncogene homolog B (BRAF), anaplastic lymphoma kinase (ALK), phosphoinositide 3-kinase catalytic alpha (PI3KCA), and fibroblast growth factor receptor 1 (FGFR1).
Materials and methods
Patient population
This study enrolled a total of 228 patients with stage 1 NSCLC who underwent complete surgical resection at National Taiwan University Hospital between 1990 and 2013. Patients who had received neo-adjuvant, adjuvant chemotherapy or radiation therapy were excluded. Otherwise, these patients belonged to the same cohort we reported previously for PD-L1 studies though 24 ADC patients and 16 SqCC patients were excluded due to unavailable specimens [20, 21]. The study protocol was approved by the Research Ethics Committee of the hospital, and all patients provided written informed consent. All the patients had been regularly followed by chest computed tomography every six months for at least five years after their surgery.
Formalin-fixed, paraffin-embedded (FFPE) tumor specimens were sectioned for microscopic examination and stained with hematoxylin and eosin. All histological and pathological analyses were independently performed by two pathologists (C-T W and Y-L C), and pathologic staging was based on the seventh edition of the American Joint Committee on Cancer guidelines.
Immunohistochemical analysis of Globo H, FGFR-1, PI3KCA and ALK expressions
For immunohistochemical (IHC) staining of tumor tissue, 4
Demographic data, clinicopathological characteristics, driver mutations and PD-L1 status in the stage I NSCLC study cohort
Demographic data, clinicopathological characteristics, driver mutations and PD-L1 status in the stage I NSCLC study cohort
*: All the EGFR mutations in the SqCC cohort are non-classical and non-EGFR TKI sensitizing mutations. Abbreviations: ALK, anaplastic lymphoma kinase; BRAF, v-raf murine sarcoma viral oncogene homolog B; EGFR, epidermal growth factor receptor; FGFR1, fibroblast growth factor receptor 1; K-ras, Kirsten rat sarcoma viral oncogene homolog; NSCLC, non-small cell lung cancer; PI3KCA, Phosphoinositide 3-kinase catalytic alpha; PD-L1, programmed cell death-ligand 1.
Globo H IHC staining was interpreted based on the percentage of moderate to strong membranous staining tumor cells in the whole section. We chose 1% as a threshold to define positive Globo H expression for sample size balance, while other cutoff values (Table S2) were also tested. For PD-L1, samples with membranous staining
Immunohistochemical stainings of PD-L1, PI3KCA, FGFR1 overexpression, and Globo H expression. (A) Positive PD-L1 membranous staining. (B) Positive staining of PI3KCA. (C) Positive staining of FGFR1. (D) Positive membranous staining of Globo H in adenocarcinoma. (E) Positive membranous staining of Globo H in squamous cell carcinoma. FGFR1, fibroblast growth factor receptor 1; PD-L1, programmed death-ligand 1; PI3KCA, phosphoinositide 3-kinase catalytic alpha.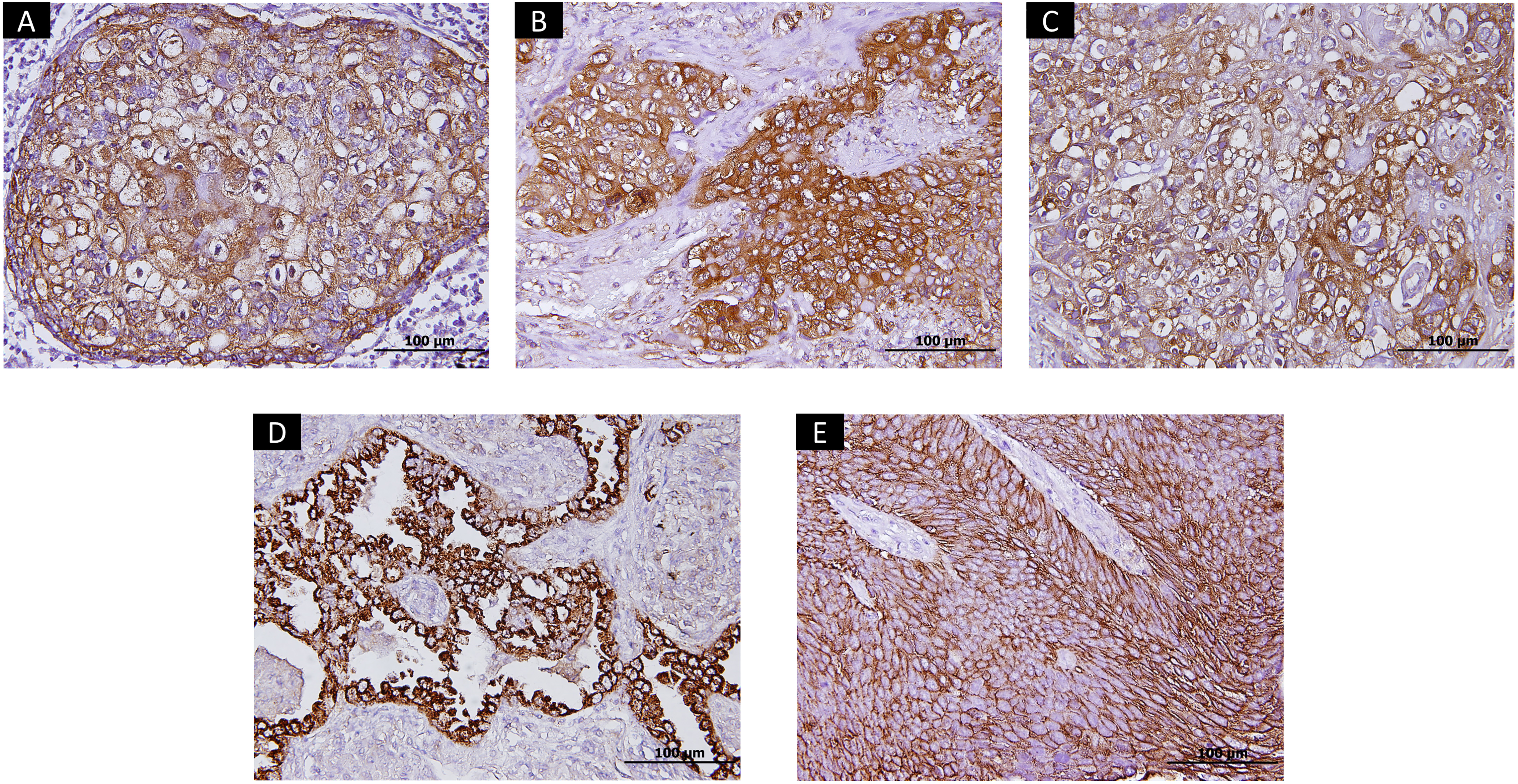
Four 10
Statistical analysis
The correlations between Globo H expression and various clinicopathologic variables were analyzed by chi-squared test, Fisher’s exact test, and Mann-Whitney U test. Logistic regression model was used to analyze factors associated with Globo H expression. Disease-free survival (DFS) and overall survival (OS) were tested by the Kaplan-Meier survival plots, while log rank test was used for comparison. The prognostic factors were analyzed by Cox regression methods, and variables with
Results
Patient demographics, driver mutations, and PD-L1 status
Patient demographic data, clinicopathological features, and status of driver mutations are summarized in Table 1. Our stage 1 NSCLC cohort included 139 patients with ADC and 89 patients with SqCC. Compared to the ADC group, the SqCC group had a higher proportion of elderly patients (median age, SqCC vs ADC: 71 vs 60 years old,
In ADC patients, the most common driver mutations were EGFR (84/139, 60.4%) and KRAS mutations (8/139, 5.8%). Other identified mutations included the BRAF mutation (6/139, 4.3%) and ALK translocation (3/139, 2.1%). The prevalence of these mutations was as expected in Eastern Asian populations. In SqCC patients, the most common genetic alterations were PI3KCA (33/89, 37.1%) and FGFR1 overexpression (14/89, 15.7%). Thirteen of the 89 SqCC patients were positive for EGFR mutations, but all of these were non-sensitizing mutations. PD-L1 expression was detected in 56 of 139 ADC patients (40.3%) and in 48 of 89 SqCC patients (53.9%) when the 5% threshold was chosen as cutoff value. Compared with the results of Dako 28-8 in phase III trials of nivolumab [12, 13] (PD-L1
The association between Globo H expression and clinicopathological characteristics, driver mutations, and PD-L1 status
The association between Globo H expression and clinicopathological characteristics, driver mutations, and PD-L1 status
Abbreviations: ALK, anaplastic lymphoma kinase; BRAF, v-raf murine sarcoma viral oncogene homolog B; EGFR, epidermal growth factor receptor; FGFR1, fibroblast growth factor receptor 1; K-ras, Kirsten rat sarcoma viral oncogene homolog; PI3KCA, Phosphoinositide 3-kinase catalytic alpha; PD-L1, programmed cell death-ligand 1.
Globo H expression was detected in 88 of 228 (38.6%) patients of the entire cohort while tumors with moderate to strong membranous staining in
Distribution of Globo H expression in stage I non-small cell lung cancer and the correlation of Globo H with driver mutation and PD-L1 expression. (A) The expression of Globo H was stratified as 5 quarters. (B) The expression of Globo H was stratified by histology. (C) Globo H expression was higher in ADC patients positive for EGFR mutation (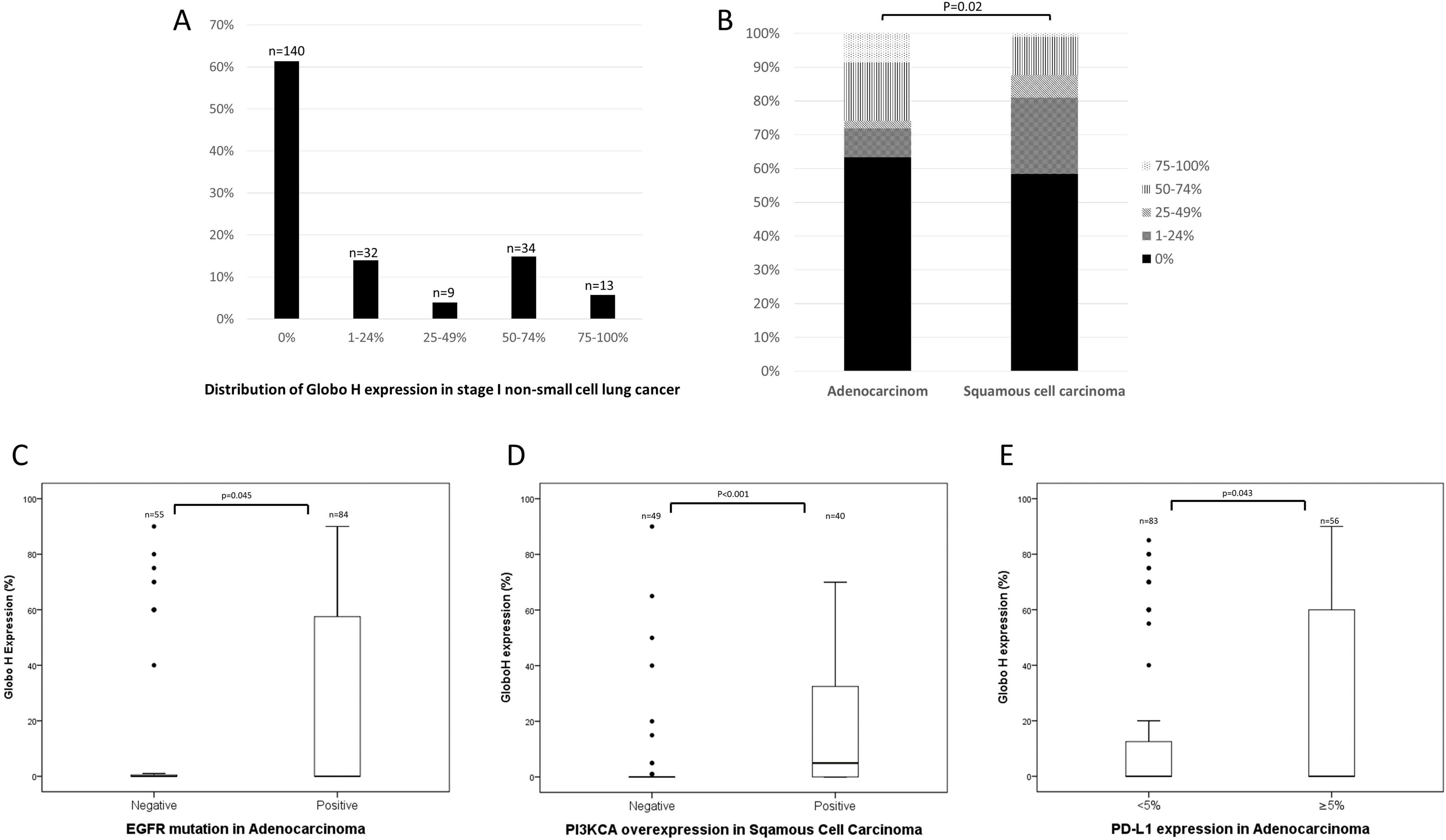
The association between Globo H and clinicopathological features, driver mutations, and PD-L1 expression are described in Table 2. In ADC patients, Globo H expression was significantly lower in patients with stage IB compared to patients with stage IA (Globo H+ in stage IB vs IA: 2/15 vs 49/144,
Regarding genetic alterations, Globo H expression was significantly associated with EGFR mutation in ADC patients and PI3KCA overexpression in SqCC patients (Table 2). The association was also significant if Globo H was calculated as a continuous variable (Fig. 2C and D). There was no significant association between Globo H expression and other driver mutations or genetic alterations (Table 2). For PD-L1, the expression of Globo H was significantly associated with positive PD-L1 expression in the ADC group (Table 2), and this association was still significant when Globo H was calculated as a continuous variable (Fig. 2E). In the SqCC group, there was no significant association between Globo H and PD-L1 expression. Through multi-variate analysis by logistic regression, Globo H expression was significantly associated with stage IA (compared with stage IB), EGFR mutation, and positive PD-L1 expression in the ADC group (Table S1). Visceral pleural invasion was not considered as a variable for logistic regression analysis because it was confounded with the variable of stage IA/IB.
The correlation between Globo H expression and clinical outcomes
The median follow-up time was 74 months (95% CI: 68.4–79.6 months). Kaplan-Meier analysis showed that positive Globo H expression only exhibited a trend towards shorter OS in all stage I NSCLC patients (Fig. 3B,
Kaplan Meier curves to analyze DFS and OS stratified by Globo H expression. (A) DFS of all patients (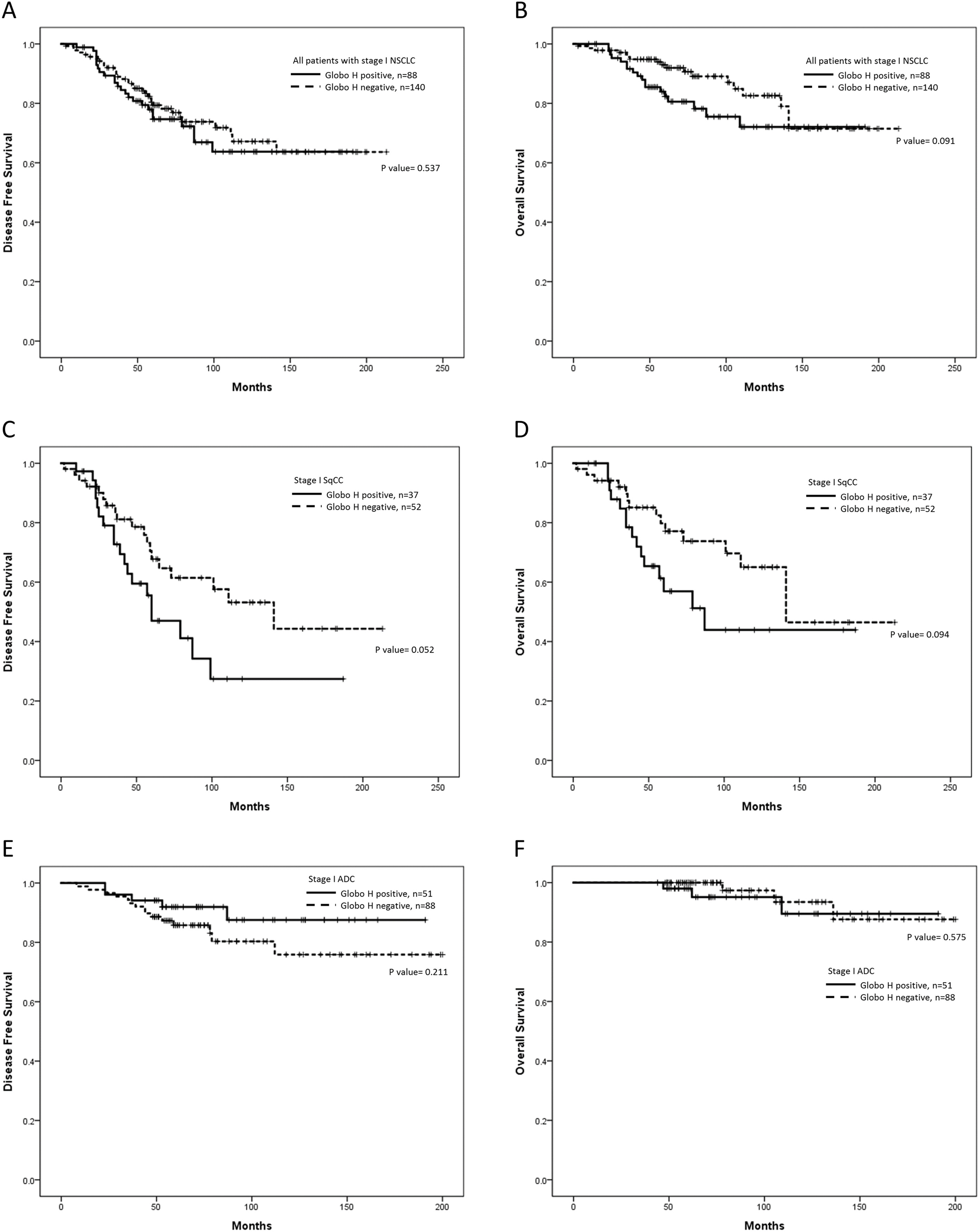
Our study showed that Globo H was overexpressed in 38.6% of stage I NSCLC patients. The proportion was slightly higher in the SqCC group (41.6%) compared to the ADC group (36.7%). Globo H expression was associated with presence of EGFR mutations, and PD-L1 expression in the ADC group, as well as PI3KCA positivity in the SqCC group. The survival analysis did not show significant difference between Globo H positive and Globo H negative patients.
Aberrant glycosylation is involved in several biological processes of lung cancer, such as sialylation on neural cell adhesion molecule 1 (NCAM1) resulting in increased aggressiveness and poor clinical outcomes [31], fucosylation of the EGFR receptor facilitating its dimerization and activation [32], and the dual control of cancer metastasis through the branching conditions of N-acetylglucosamine (GlcNAc) [33, 34]. Our study showed that Globo H expression in lung cancer correlated with EGFR mutation in ADC and PI3KCA expression in SqCC. Currently the interplay between Globo H, EGFR, and PI3KCA is poorly understood. Several studies have investigated the connection between genetic alterations and glycan modification. For example, gangliosides were previously reported to be important modulators of receptor tyrosine kinase (RTK) signal transduction. Depending on the binding structure of the ganglioside, monosialogangliosides (GM1 and GM3) may negatively regulate RTK, while disialogangliosides (GD2, GD3, and GD1a) may cause RTK activation [35]. Like gangliosides, Globo H is also a membrane bound glycosphingolipid synthesized through fucosylation of stage-specific embryonic antigen-3 (SSEA-3) by fucosyltransferases 2 and 1 (FUT2 and FUT1) [36]. Therefore, our findings may pave a road for further studies of the molecular interactions between EGFR, PI3KCA, the activity of fucosyltransferase, and the expression of SSEA-3.
Our findings also showed that Globo H expression was associated with positive PD-L1 expression. As a TACA, the expression of Globo H signals the presence of a transformed cell and triggers immune surveillance and tumor cell killing by the host immune system, mainly through complement-dependent cytotoxicity [37]. However, uptake of Globo H by immune cells was also reported to inhibit cell proliferation and cytokine production, suggesting tumor cell escape from immune attack [38]. Additionally, it has been reported that uptake of Globo H by endothelial cells induced cell migration and tube formation, leading to angiogenesis [39]. Since PD-L1 expression in early stage lung cancer may indicate adaptive resistance, [20, 21] the association of Globo H expression with PD-L1 expression may suggest a more immunogenic microenvironment, which could force escape of tumor cells from the host immune system. Therefore, our findings may open a new direction for further studies to explore the interaction between PD-L1 and Globo H.
Globo H expression is usually linked to an aggressive cancer behavior and poor survival in various neoplasms [6]. However, our data only yielded a trend towards shorter DFS and OS in Globo H positive pulmonary SqCC patients rather than ADC patients. The prognostic impact of Globo H is not significant through multi-variate analysis. These results suggested that the impact of Globo H expression on cancer cell aggressiveness may be less significant in lung cancer than in other cancers.
In conclusion, our data suggested that Globo H is overexpressed in a subset of NSCLC patients, and these findings may help identify a subgroup of patients who may benefit from Globo H-based immunotherapy. Our results also implicate the necessity for further explorations on the molecular interactions between Globo H, EGFR, PI3KCA, and PD-L1.
Footnotes
Acknowledgments
The authors would like to thank Ms. Chih-Hsin Chen for her skillful technical support. This study is funded by National Taiwan University Hospital (NTUH106-S3342 and NTUH106-S3343).
Conflict of interest
The authors declare no conflict of interest.
Supplementary data
The supplementary files are available to download from
