Abstract
BACKGROUND:
The study was aimed at investigating the role of PD98059 on impairing the cisplatin-resistance of ovarian cancer cells and figuring out the potential mechanism.
MATERIAL AND METHODS:
Treated with low dose of cisplatin (DDP), DDP-resistant ovarian cancer cells were built and named as SKOV-3/DDP. The cell viabilities of ovarian cancer cell line SKOV-3 and SKOV-3/DDP were detected using MTT assay. Wound healing assay and flow cytometry were performed to detect the migratory ability and cell cycle variation of the two cells and assess the sensibility to DDP in the two cell lines. However, cotreated with DDP and PD98059, cell viability, migration and cell cycle of SKOV-3/DDP were determined again. The DDP-resistance varied a lot and the potential mechanism was studied via western blot assay.
RESULTS:
Both treated with DDP, SKOV-3/DDP showed an intense resistance than SKOV-3 including stronger cell viability, larger migration area and less G1/G0 arrest, which confirmed the successfully established DDP-resistant cell line. The phosphorylation of ERK and the activation of epithelial mesenchymal transition (EMT) process contributes to the enhanced resistance. PD98059, a MEK inhibitor, suppresses the ERK pathway and the EMT process of SKOV-3/DDP. Co-treated by DDP and PD98059, cell proliferation and migratory area decreased, meantime more cell were arrested in G0/G1 phase compared to simple treatment of DDP or PD98059.
CONCLUSION:
PD98059 efficiently impairs the DDP-resistance of ovarian cancer cells via downregulating the ERK pathway and the EMT process.
Introduction
Ovarian cancer is one of the most deadly female cancer diseases, with 45.6% 5-year survival [1, 2]. The absence of specific symptoms and lack of reliable early diagnostic methods has resulted in the diagnosis of 70% of patients at an advanced stage [3]. The incipient symptoms of ovarian cancer are obscure and most patients received diagnosis until entering the advanced stage [4, 5, 6]. Surgical treatment and platinum-based chemotherapy are major therapeutic strategies for ovarian cancers [7]. Unfortunately, these therapeutic methods seem to become less effective with the progression of the cancer. Moreover, the morbidity of the ovarian cancer also remains a higher level due to lack of reliable predictive biomarker, ovarian cancer cells metastasis and the resistance to chemotherapy [8, 9].
Cisplatin (DDP), a platinum-based drug, is one of the most valid agents of solid tumors. Platinum-based drugs can cause deadly DNA damage and usually exert severe cytotoxic effect on malignant cells by inducing cell apoptosis. However, DDP resistance has been serious obstruction to the recovery of patients with ovarian cancer [10]. In response to DDP, multiple signaling pathways were activated and contributed to DDP-resistance [11].
ERK pathway is a part of the classic MAPK (mito- gen-activated protein kinase) signaling pathway, regulating cell activities [12]. Steinmetz et al. have reported that the activation of ERK pathway will induce tumor genesis and resistance to chemotherapeutic drugs, such as cisplatin and doxorubicin [13, 14]. ERK pathway is increasingly activated in DDP-resistant cells, and it may promote tumor cell survival in ovarian cancer [15, 16]. Epithelial-mesenchymal-transition (EMT) plays an important role in tumor metastasis and physiological reaction to injury [17]. During EMT process, the phenotype and function of the cells will change, which mainly embody in the conversion from epithelial to mesenchymal, a loss of cellular polarization, acquisition of mobility and invasion capacity [18]. Compelling evidence suggests that repressing the EMT process could inhibit the proliferation, migration and invasion of ovarian cancer cells [19]. The work of Haslehurst et al. strongly supported that EMT has an important effect on DDP resistance in ovarian cancer [20].
PD98059 (MEK1 Inhibitor) has been shown to act both in vitro and in vivo as a highly selective inhibitor of MEK1 activation and the MAP kinase cascade [3, 21]. Combination treatments with PD98059 and another anti-tumor reagent were widely reported to enhance the cell cycle arrest and cell apoptosis [21, 22]. In our research, we combined PD98059 and DDP for promoting the inhibitory effects in DDP-resistant ovarian cancer cells.
Our experimental data supported that the activation of ERK pathway and EMT process evaluated the DDP resistance in ovarian cancer cell line SKOV-3/DDP. As an inhibitor of MEK, PD98059 impairs the DDP resistance in SKOV-3/DDP cells, which could offer a novel therapeutic strategy for overcoming DDP resistance.
Materials and methods
Cell lines and cell culture
The ovarian cancer cell line SK-OV-3 (derived from ascites of ovarian tumor) was bought from ATCC, and the DDP-resistant ovarian cancer cell line SKOV-3/DDP are built by our lab, applying the concentration of 4 mg/ml DDP stimulate to the ovarian cancer cell line SK-OV-3 repetitively. RPMI 1640 (Sigma, St. Louis, MO, USA) culture medium with 10% fetal bovine serum (FBS), 100 U/ml penicillin and 100 g/ml streptomycin (Invitrogen, Gaithersburg, MD, USA) were utilized to incubate the cells. All the cells were stored in an incubator containing 5% CO
MTT assay
3-(4, 5-dimethyl-2-thiazolyl)-2, 5-diphenyl-2H-tetr- azolium bromide (MTT) assay was utilized to examine cell viability. After treatment, SKOV-3 cells and SKOV-3/DDP cells were collected in logarithmic growth phase, and different concentrations of DDP (0.00
Wound healing assay
SKOV-3 cells and SKOV-3/DDP cells were seeded into two 6-well plates (1
Western blot assay
Protein concentrations were determined by bicinchoninic acid (BCA) protein assay kit (Beyotime Institute of Biotechnology, Jiangsu, China) and proteins with 30–100
Flow cytometry (FCM) detection
FCM was employed to detect the cell cycle and apoptosis rate. We adopted Annexin V-FITC/PI dual-fluorescence apoptosis detection method to assess the apoptosis of ovarian cancer cells. The cells were placed into 12-well plates and cultured with 5
Statistical analysis
All data were analyzed using SPSS 21.0 (IBM Corp., Armonk, NY, USA). The results are represented as the means
Results
SKOV-3/DDP cells were more drug-resistant than SKOV-3 cells
Treated with different concentrations of DDP for 24 h, SKOV-3 and SKOV-3/DDP cells were collected. Cell viabilities were examined with MTT assay and DDP did have a dose-dependent and time-dependent response for cell growth. However, after the treatment of DDP at same concentration during same time, the growth inhibition rate in SKOV-3 group was significantly higher than that in SKOV-3/DDP group (
(A–B) SKOV-3 cells and SKOV-3/DDP cells were detected by MTT assay at different time. Growth inhibition rate of the SKOV-3 cells and SKOV-3/DDP cells at different concentration of DDP was measured. Growth inhibition rate went up as the concentration increased. Higher inhibition rate was discovered in SKOV-3 cells compared with SKOV-3/DDP cells. Significant difference was detected between two groups, which confirmed the drug resistance of SKOV-3/DDP cells. (C–D) Proliferation was detected by flow cytometry. DDP could promote the apoptosis of SKOV-3 cells, confirming the drug resistance of SKOV-3/DDP cells. (E–F) Cell cycle was detected with flow cytometry. Cells were arrested in G0-G1 phase and G2/M phase in SKOV-3 cells, indicating that DDP blocked the proliferation of SKOV-3 cells, proving the drug resistance of SKOV-3/DDP cells (both 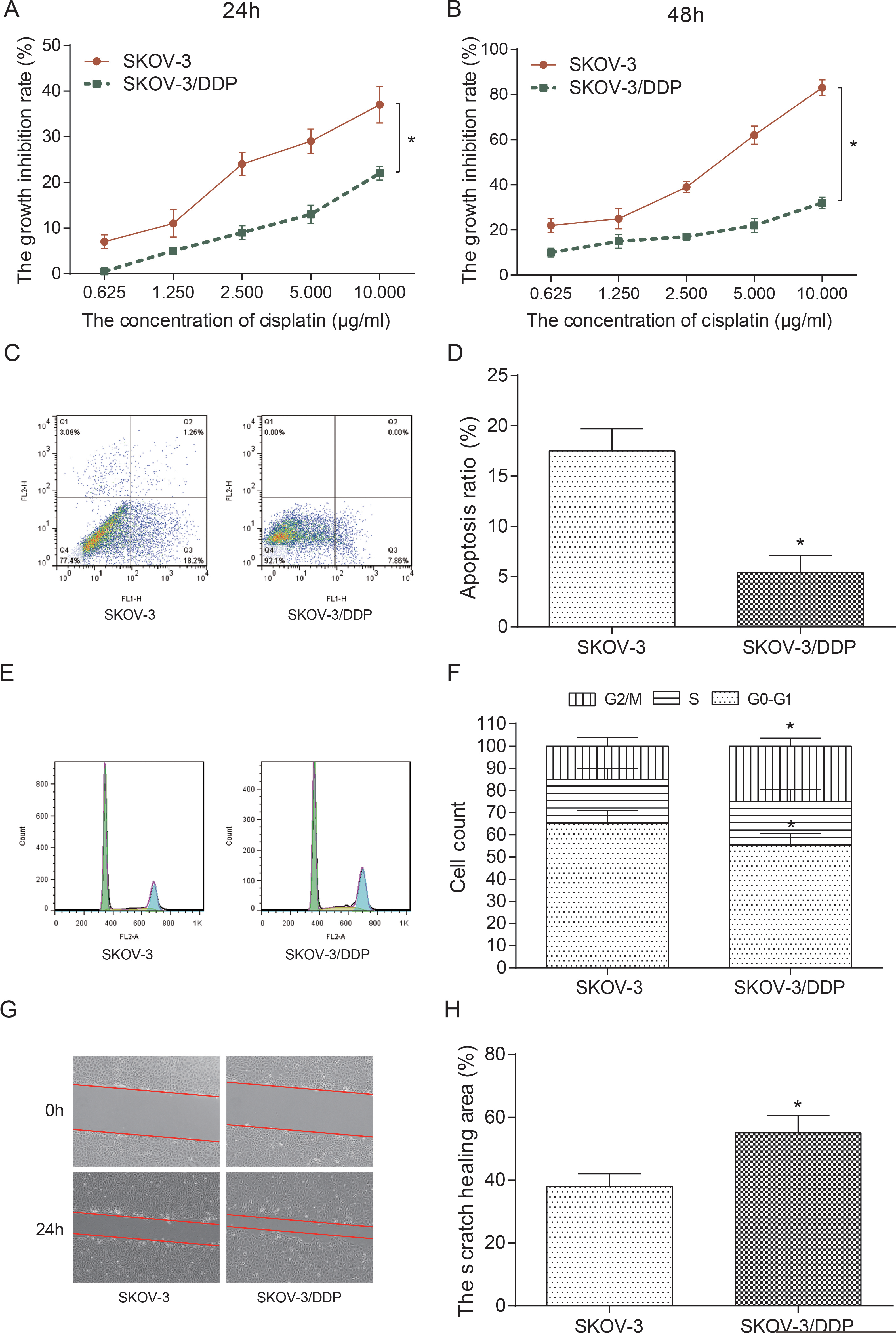
The expression level of ERK pathway- and EMT-related proteins were detected by western blot assay. (A) The expression of ERK pathway- and EMT-related proteins in SKOV-3/DDP cells and SKOV-3 cells before drug treatment. The expression of Vimentin in SKOV-3/DDP cells was higher. E-cadherin, MEK and p-ERK in SKOV-3/DDP cells presented a higher expression in comparison with SKOV-3 cells. (B) The expression level of ERK pathway- and EMT-related proteins in SKOV-3/DDP cells after drug treatment. The expression level of Vimentin increased, the expression levels MEK and pERK were decreased while the expression of E-cadherin displayed no significant difference in DDP group compared with the control group. The expression level of E-cadherin increased and the expression of Vimentin, MEK and pERK were decreased in PD98059 group. The expression level of E-cadherin was increased and the expression of Vimentin, MEK and pERK decreased in the group of combined the PD98059 and DDP. *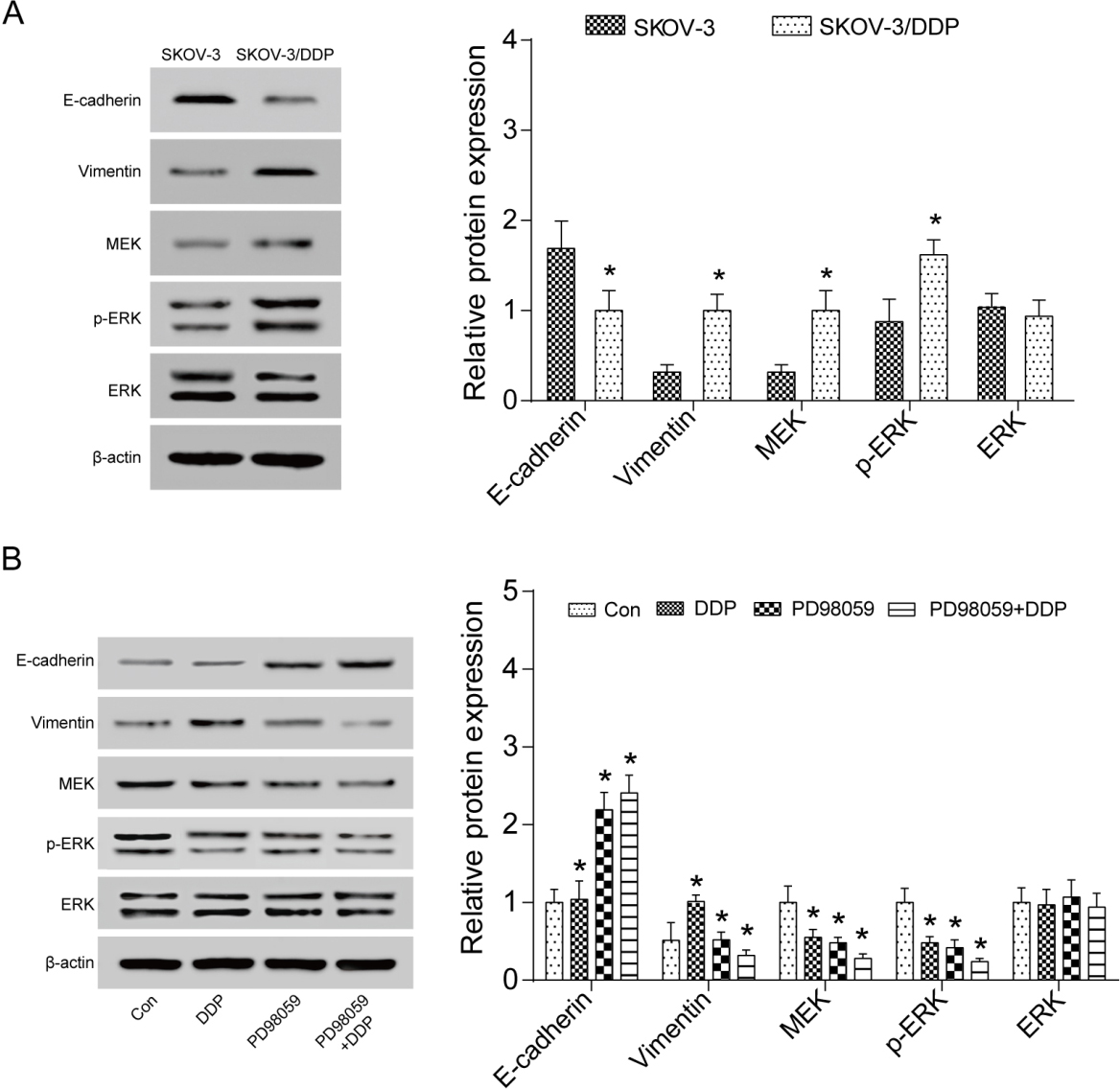
Expression level of related proteins in ERK pathway and EMT process were detected by western blot in SKOV-3 cells and SKOV-3/DDP cells. In the course of EMT, E-cadherin is the biomarker of epithelial cells and Vimentin is the biomarker of mesenchymal cells. MEK was the upstream protein in ERK pathway; p-ERK was the activated protein in ERK pathway. The expression of E-cadherin in SKOV-3/DDP cells was significantly lower. However, Vimentin, MEK and phosphorylation of ERK (p-ERK) in SKOV-3/DDP cells presented a considerably higher expression in comparison with SKOV-3 cells, suggesting an enhanced ERK activation and an EMT process (all
(A–B) Cell apoptosis was confirmed by flow cytometry. Apoptosis rate of DDP group, PD98059 group and DDP 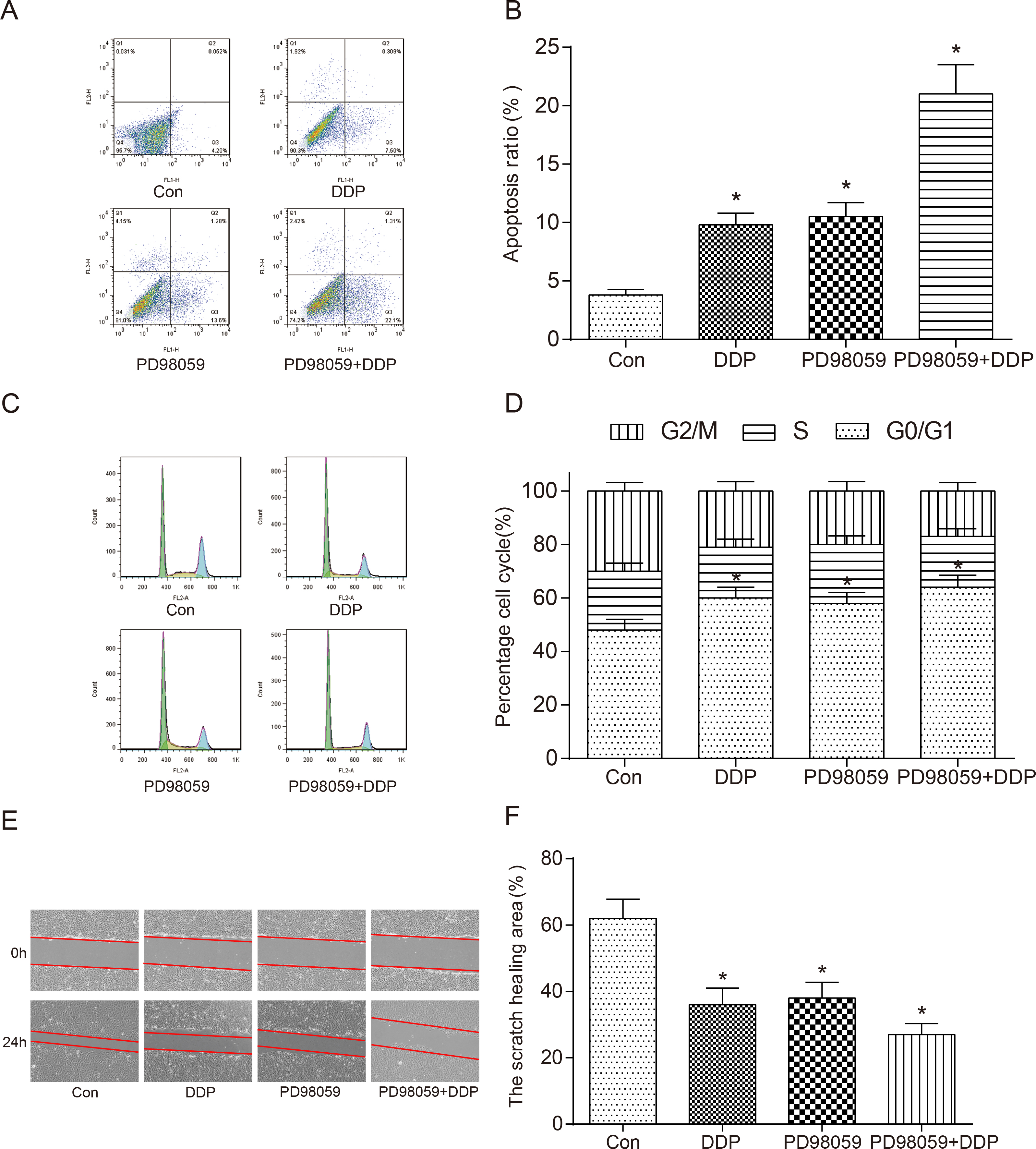
After being treated with DDP, PD98059 or DDP
Cell apoptosis rate was detected in four groups, and that in DDP group, PD98059 group and DDP
Discussion
Ovarian cancer is one of the most lethal malignant gynecological tumors. The 5-year survival in patients with advanced ovarian cancer is less than 30% because of the lack of effective biomarkers for diagnosis, prognosis, and personalized treatment [23]. Although surgical treatment and chemotherapy of ovarian cancer have improved in recent years, the prognosis of ovarian cancer remains poor. The current standard of treatment for primary ovarian cancer is the combination of optimal cytoreductive surgery and platinum-based chemotherapy, but the platinum resistance contributes to a high rate of relapse [24, 25]. Cisplatin is commonly used for ovarian cancer treatment, but the relevant molecular and cellular mechanism for cisplatin resistance remains unclear. Hence, it is critical to figure out the additional mechanism and overcome the cisplatin resistance, which can increase the total prognosis and improve the living qualities of female patients.
In this research, we first successfully obtained DDP-resistant SKOV-3 cells and named it SKOV-3/DDP. Activated ERK pathway and EMT process were found in SKOV-3/DDP cells. ERK pathway is a part of the MAPK pathway, which is associated with cell cycle arrest, DNA repair, cell survival and apoptosis. Large amount of evidence has shown that ERK1/2 activation can influence apoptosis, cell survival and DDP resistance [26, 27, 28]. A recent study on ovarian cancer reported that DDP could induce EMT process, which can be partially inhibited by a selective MEK inhibitor, which was also reported to suppress ERK2 activation [29]. EMT process has been reported extensively to enhance the chemotherapy tolerance including DDP resistance in different kinds of cancers [20, 30, 31]. Kipps et al. and Yuan et al. also demonstrated the determinant role that EMT plays in ovarian cancer metastasis and invasion [32, 33].
To block the ERK pathway and weaken the EMT process, we combined DDP and PD98059 to treat the SKOV-3/DDP cells. Though DDP treatment has few effects on cell viability, migratory ability, cell cycle and apoptosis rate, cotreatment efficiently enhanced the inhibition and impaired the DDP resistance. PD98059 has been reported to trigger the G1 arrest and promote cell apoptosis of prostate cancer cells, leukemic cells and lung cancer cells [21, 34, 35]. Thus, it’s a novel strategy to utilize PD98059 in ovarian cancer cells to enhance the inhibitory effect of DDP, and the enhancement is efficient enough for further performing the in vivo research.
Conclusion
Here, in this research, we first established the DDP-resistant cells, SKOV-3/DDP. The resistance is ascribed to activated ERK phosphorylation and EMT process. Hence, we select PD98059, a MEK inhibitor, to inhibit the ERK activation during the DDP treatment. As a result, PD98059 efficiently enhanced the sensibility of DDP in SKOV-3/DDP cells. The application of the strategy in clinics could be challenging at present, thus further in vivo experiments are essential. We firmly believe it could be clinical valuable for DDP-resistant female patients.
Footnotes
Conflict of interest
None declared.
