Abstract
OBJECTIVE:
To investigate the improvement of cytotoxicity of autologous CIKs from patients with breast cancer to MCF-7 cells by suppressed PD-1 expression.
METHODS:
The Lentiviral Vector/PD-1 carrying the gene that can suppressed PD-1 was transferred to CIK cells from patients with breast cancer to inhibit PD-1 expression. The PD-1 protein were detected by RT-PCR and Western blot. The positive PD-1 of CIKs and PD-L1 of MCF-7 cells were detected by FCM, and cytotoxicity of CIKs to MCF-7 was assayed by CCK-8.
RESULTS:
The PD-1 positive CIKs with Lentiviral Vector/PD-1 transferred from patients with breast cancer were 16.02%, 14.36% and 14.64% at 14
CONCLUSION:
MCF-7 cells have a strong PD-L1 expression at its surface, and inhibition of PD-1 expression can improve the cytotoxicity of CIK cells.
Abbreviations
CIK, cytokine-induced killer;
MCF-7, Michigan Cancer Foundation
(MCF-7 cells);
PD-1, programmed cell death-1;
CCK-8, Cell Counting Kit-8.
Introduction
Cytokine induced killer (CIK) is a kind of CD3/ CD56 double positive cells activated by a variety of cytokines in vitro. In China clinical trial research has been extensively performed, however the curative effect is not clear and definite [1, 2, 3, 4]. The early stage of our research showed that programmed cell death-1 (PD-1) presented high expression on activated CIK cells [5]. PD-1 belongs to immunoglobulin CD28 super family. Programmed cell death-Ligand 1 (PD-L1) (B7-H1) is a ligand of PD-1 and has high expression on malignant tumor cells such as breast cancer, gastric cancer and pancreatic cancer [6, 7, 8]. One possible reason of the poor therapeutic effects of CIK cells in clinical application is when PD-1 combines with its ligands the dephosphorylation of downstream effector molecules is induced, which results in negative regulating effects that induces the apoptosis of T cells [9]. In this study, we have collected and cultured autologous CIK cells from umbilical cord blood and breast cancer patients, the constructed Lentiviral Vector/PD-1 recombinant plasmids was used to inhibit PD-1 expression in CIK cells. The killing effect of the blank control group, empty vector group CIK cells and Lentiviral Vector/PD-1 CIK cells group on MCF-7 breast cancer cells were observed.
Materials and methods
General information
The six breast cancer patients who have accepted CIK cells transplantation in the 105 Hospital of PLA, average age is 46.5 ranging from 35 to 63, and histological type is invasive ductal carcinoma. Autologous blood collection from patients have been approved by the hospital ethics committee, signed informed consents and registered in Chinese clinical laboratory research center (ChiCTR-ONRC-13002983).
Reagents and materials
Lentiviral Vector/PD-1 of PD-1 recombinant plasmids is provided by Doctor Yueshi and Min Zhou from Colton Ltd. The breast cancer MCF-7 cells are purchased from Cell resource center of Shanghai Institute of Life Sciences, Chinese Academy of Sciences. The Lipofectamine 2000 transfection reagent is produced by Invitrogen Company. The Reverse transcription Kit and the SYBR real time quantitative Kit are purchased from Takara Company. The PD-l and PD-Ll antibody are purchased from Abcam Company. Lymphocyte separation solution (Ficoll) (Tianjin Haoyang Biological Technology Ltd); 1640 complete culture medium (Hyclone Company); Fetal bovine serum (Hangzhou Sijiqing Company); Cell counting Kit (CCK-8) (Japan Dojindo Company); Apoptosis Kit (Japan Dojindo Company); IL-2, INF-
The isolation, purification and cultivation of CIK cells
Ficoll two step separation method was applied to isolate mononuclear cells which were cultivated in incubator at 37
CIK cells at 14th day of culture and CIK cells after transfection withLentiviral Vector/PD-1/GFP.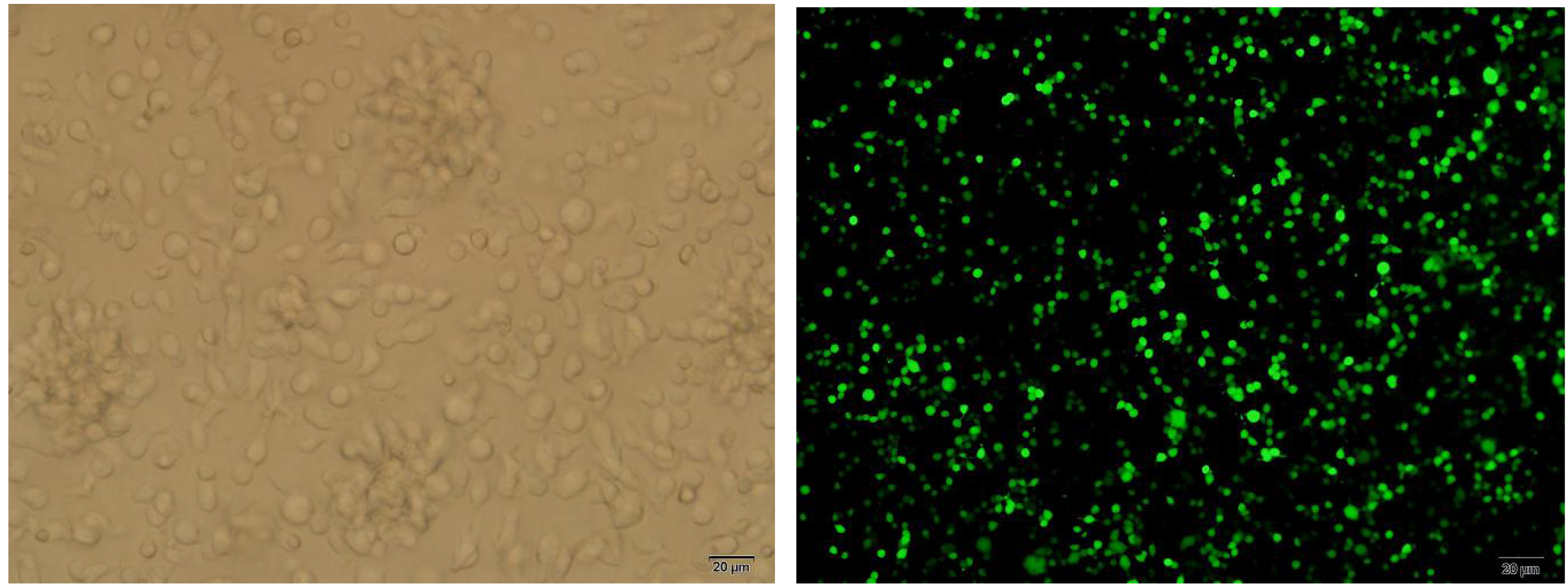
Virus culture medium which contained Lentiviral Vector/PD-1/GFP recombinant plasmids and T cell culture medium (RPMI1640
Cell identification and PD-1, PD-L1 measurement with flow cytometry
CIK cells at 7th, 14th and 21st day of culture were collected to detect the number of CD3
RT-PCR method to detect the expression of PD-1 gene
Chloroform, ethanol, ethanol were gradually added to extract the total RNA of each group according to the RNAios PLUS protocol. Real time quantitative polymerase chain reaction (PCR) was performed according to the standard protocol of reverse transcription kit. PD-1 gene primers include upstream primer 5
Detection of PD-1 protein expression with western blotting
CIK cells were collected and washed two times with cold phosphate buffer saline (PBS) to extract total protein by pyrolysis solution. Protein concentration was determined by bicinchonininc acid (BCA) protein assay kit. With the quality of 50
The expression rate and inhibition efficiency of PD-1 in each CIK cells group were detected by flow cytometry
The expression rate and inhibition efficiency of PD-1 in each CIK cells group were detected by flow cytometry
A: PD-1 high expression of CIK cell at 28th day; B: plasmid significantly inhibited the PD-1 expression; C: Isotype control chart, FS and SS gating and Isotype control with the same type of PD-L1 antibody; D: MCF-7 cells high expressed PD-L1. 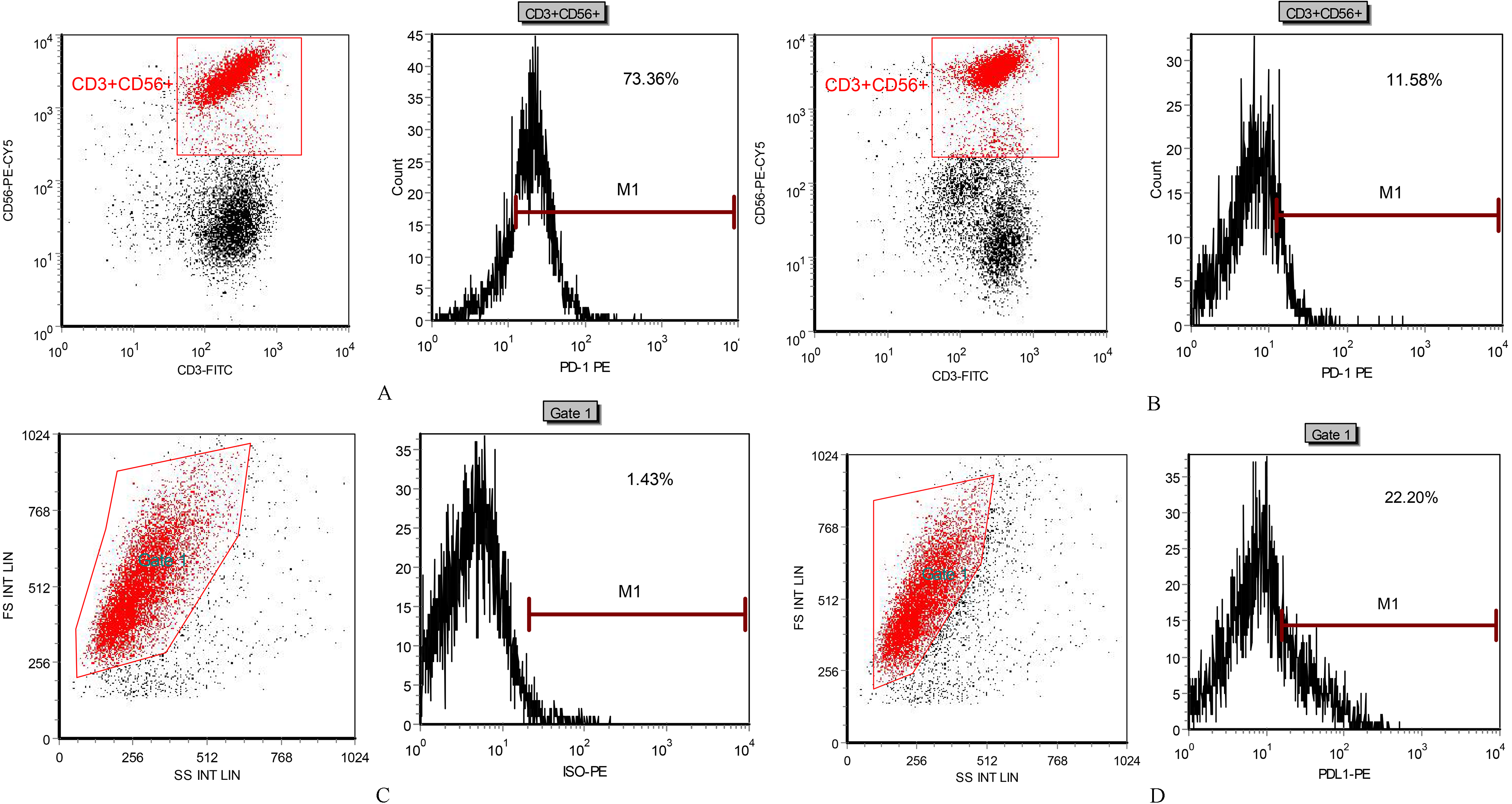
Cytotoxicity was detected by CCK8 assay 1
Statistical analysis
The experimental data was processed by SPSS13.0 software and expressed by
Results
PD-1 recombinant plasmid was successfully transfected into CIK cells
Transfection status of CIK cells were observed under an immunofluorescence microscope on the 7th, 14th, 21st and 28th day of culture. As shown as in the picture, cell colonies with irregular shape were significantly increased, which 80% of the cells showed green fluorescence (Fig. 1).
The expression rate, inhibition efficiency of PD-1 in each CIK cells group and PDL-1 of MCF-7 cells were detected by flow cytometry
Flow cytometry detection showed that as the extension of culture time, the expression rate of PD-1 increased gradually; PD-1 expression on CIK cells was significantly inhibited by Lentiviral Vector/PD-1 plasmids; high expression of PD-L1 was observed on MCF-7 cells (Table 1 and Fig. 2).
The killing effect of the two groups of CIK cells on MCF-7 cells
The killing effect of the two groups of CIK cells on MCF-7 cells
The expression of PD-1 gene was detected by RT-PCR method.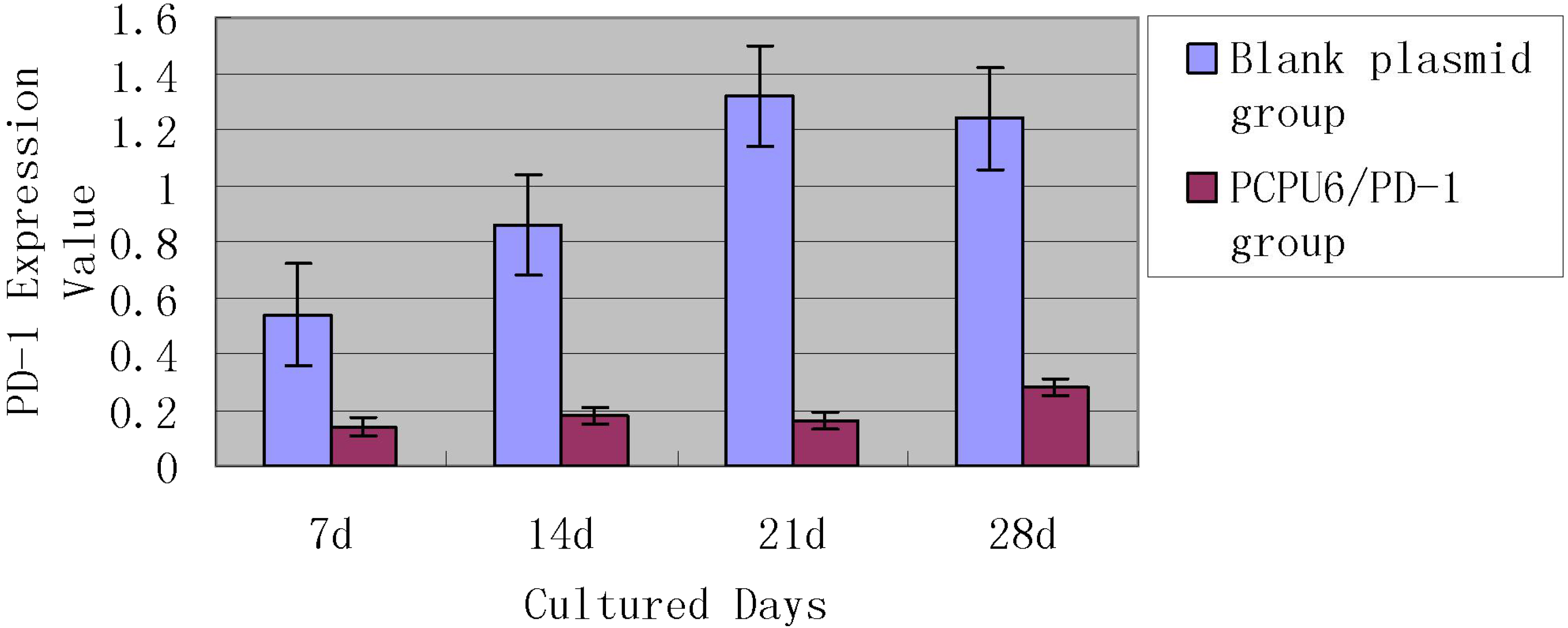
After the plasmid was transfected for 48 hours, the cells were collected and the total RNA was extracted and reverse transcribed, then RT-PCR was performed to detect the level of PD-1 mRNA. The expression of PD-1 in the blank control group was set as 1, and the results showed that relative expression of PD-1 mRNA in CIK cells which transfected with Lentiviral Vector/null was 1.08
The result of Western blotting presented that there was no obvious difference of PD-1 expression between Lentiviral/null group and the control group, but the PD-1 protein expression in Lentiviral/PD-1 group was significantly decreased (Fig. 4).
A: Lentiviral Vector/null group; B: CIK 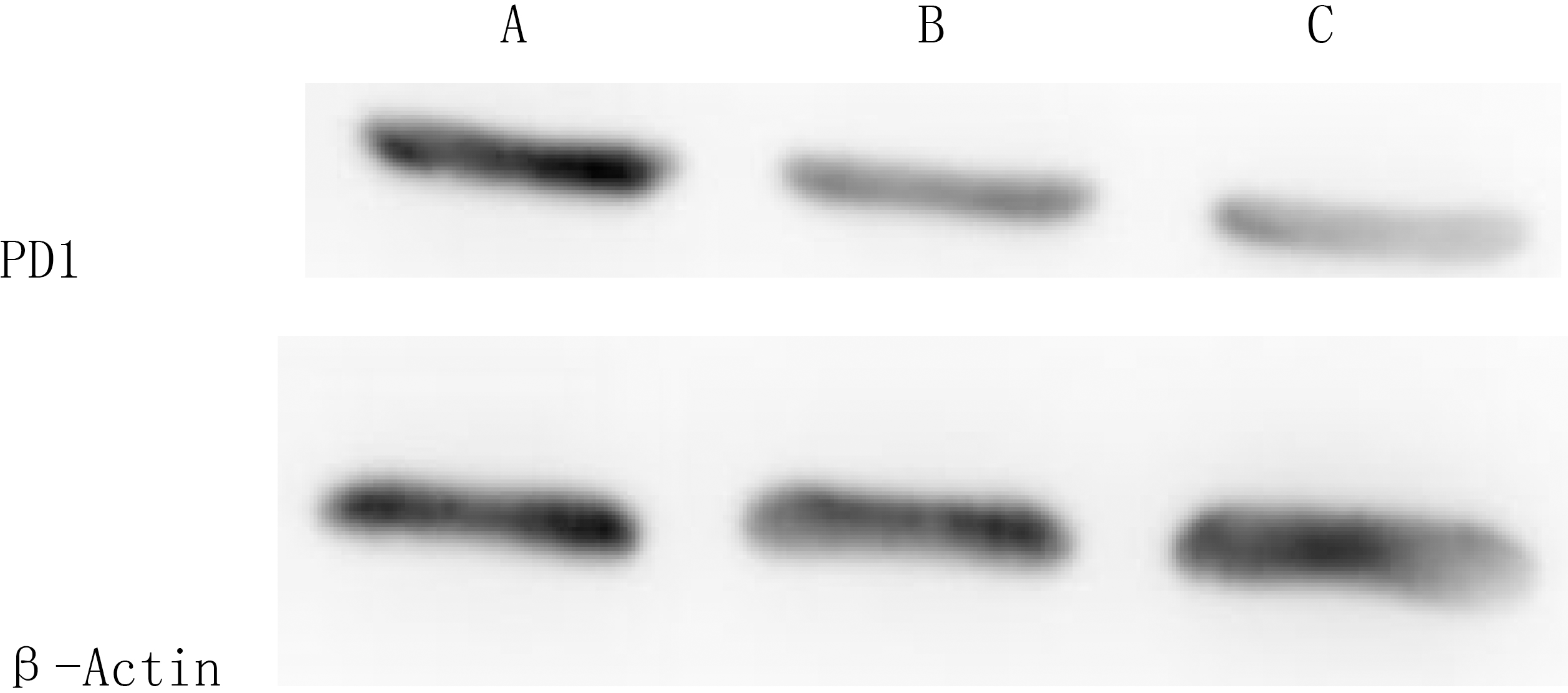
Compared with the two groups, the killing rates of transfected CIK cells with Lentiviral Vector/PD-1 plasmid on 7th, 14th, 21st and 28th day of culture towards MCF-7 cells were significantly higher than that of the blank plasmid group, as shown in Table 2.
Discussion
T lymphocytes play crucial roles in human immune surveillance, killing tumor cells and immunoregulation, and also PD-1 is highly expressed on the surface of activated T lymphocytes [10]. CIK cells are a group of special activated T lymphocytes that making characters of a mixture of natural killer (NK) and T – cell-like phenotype. In order to enhance the maturation and proliferation of CIK cells, cytokines, such as IL-2 and PHA, were added to the culture system. The combination of PHA and IL-2 increased the killing activity of T cells, as well as promoted the expression of PD-1 on the cell surface [11]. PD-1 can inhibit T cell proliferation and promote T cell apoptosis by interacting with ligand PD-L1, PD-L2 [12, 13]. The up-regulated expression of PD-L1 has been demonstrated on cell surface of certain tumor types, also the activation of PD-1/PD-L1 pathway is an important mechanism of tumor immune escape [14, 15], which may be one of the reasons why CIK cell therapy was less effective in certain types of tumors. As binding to the corresponding ligands is requisite for the activation of these immune checkpoints, monoclonal antibodies were selected to block the binding, thereby breaking tumor immune suppression [16, 17]. A series of study targeting on anti-PD-1/PD-L1 monoclonal antibodies have been processed to clinical trials, and the I-phase clinical trial showed that anti-PD-1/PD-L1 monoclonal antibodies had definite effect on advanced malignancies [18]. It has also been shown that the high expressed PD-L1 ligand in breast cancer inhibited the killing effect of CD8
Although higher expression rate of PD-L1 was demonstrated in ER-/PR-breast cancer [22], 60% to 70% of breast cancer patients in China exhibited positive estrogen or progesterone receptors [23]. PEMBRO monoclonal antibody in I-b phase study, KEYNOTE-028 trial, showed that the total remission rate (ORR) of ER
To explore the effect of inhibiting PD-1 expression on CIK cell surface on killing ER
However the relationship between PD-L1 expression on CIK cells and CIK therapeutic efficacy is fairly complicated. In the clinical trial of CIK cell therapy in hepatocellular carcinoma performed by Chen CL suggested that In the CIK treatment group, higher PD-L1 expression was observed in patients who exhibited long-term survival benefit. Survival analysis showed that patients with
As shown as in the ASCO
Conclusions
For the breast cancer, PD-L1 is an independent prognostic factor [30], and our results also suggest that individualized blockade therapy targeting on it will be an important direction for the future clinical therapy.
Conflict of interest
We declare that we have no financial and personal relationships with other people or organizations that can inappropriately influence our work, there is no professional or other personal interest of any nature or kind in any product, service and/or company that could be construed as influencing the position presented in, or the review of the manuscript entitled.
Footnotes
Acknowledgments
Project supported by the Science and Technology Foundation of Nanjing Military Region (No. 15MS048).
