Abstract
AIMS AND BACKGROUND:
Squamous cell carcinoma (SCC) is one of the most common skin cancers. Photodynamic therapy (PDT) is a non-invasive treatment for SCC, but it is usually effective only on tumors just under the skin. Resveratrol (Res) is a polyphenolic compound, which is capable of promoting apoptosis of a variety of cancer cells. Res administration is non-invasive and effective on SCC, thus it may be used as an adjuvant for PDT. So far, there is no published study investigating the combination use of PDT with Res to improve clinical outcome of SCC. So in this study, we will examine the effectiveness of combined treatment of PDT and Res as well as its underlying mechanism.
METHODS:
The human HaCaT keratinocytes and human A431 epidermoid carcinoma cells were treated with ALA-PDT or/and Res, and cell proliferation and apoptosis were evaluated by MTT and flow cytometry respectively afterwards. p-ERK, p38, p53 and caspase-3 protein expression was examined by western blot. Then a p38 inhibitor was added to test the involvement of p38 pathway in A431 cells responding to ALA-PDT and Res treatments.
RESULTS:
The results showed that Res could enhance the effect of ALA-PDT on cell proliferation and apoptosis in A431 cells. We also found that the expression of p-ERK, p-p38, p53 and caspase-3 was increased. However, inhibition of p38 pathway attenuated the effect of Res.
CONCLUSION:
Our study demonstrated that Res could enhance the effect of ALA-PDT against skin cancer cells through p38/ MAPK pathway.
Introduction
Cutaneous squamous cell carcinoma (SCC) is a common cancer arising from malignant proliferation of the keratinocytes from the epidermis. Accounting for 20% of all cutaneous malignancies [1], SCC is the second most common non-melanoma skin cancer, next to basal cell carcinoma (BCC). But unlike BCCs which usually have very good prognosis, SCCs tend to metastasize to regional lymph nodes or more distant sites, resulting in significant morbidity or death [2]. Furthermore, SCC incidence was more than doubled in recent years and expected to rise continuously because of aging population [3, 4]. Currently, surgical excision is the primary treatment option to SCC; while radiation therapy and chemotherapy may be considered as adjuvant therapies in select high-risk cases of SCC to provide improved local control.
Resveratrol/ALA-PDT inhibited proliferation in A431 carcinoma cells. Cell proliferation inhibitory rate were determined by MTT method. (A) Res resulted in proliferation inhibition in A431 cells; (B) Res resulted in proliferation inhibition in HaCaT cells; (C) Both Res and ALA-PDT, in single or in combination, could inhibit proliferation in A431 cells. All the results were shown as mean 
Due to recent advances in photosensitizer and laser technologies [5], photodynamic therapy (PDT) becomes a more and more effective treatment method for SCCs [6, 7]. PDT is currently used for treatment of several tumors including skin cancer, lung cancer and colon cancer [6, 8, 9]. Photosensitizer such as ALA preferentially accumulates in hyperproliferating tumor cells and generates reactive oxygen species (ROS) at an appropriate wavelength irradiation, triggering irreversible tumor destruction [10]. Previous studies had found that treatments of HaCaT human epidermal cells and H460 lung cancer cell lines with ALA-PDT were able to induce tumor cell apoptosis through MAPK related signal pathways [11, 12]; and the photocytotoxicities of ALA-PDT to cancer cells were mainly stress-activated. As a new type of cancer treatment, the most prominent advantage of PDT is to accurately target the tumor tissues. Numerous clinical research studies have shown the efficacy of PDT for treating pre-malignant and malignant skin cancer [9]. Compared to surgical treatment, PDT is non-invasive and generally gives a good cosmetic outcome. Additionally, it also has several other advantages, such as fewer adverse effects, short treatment time and lower costs than other treatments [9]. However, PDT has its own limitations. It is less effective on large tumors or tumors deeply under the skin. Thus, resistant or recurrent tumors may appear, and sometimes become more aggressive [13, 14]. Thus, combined treatments with PDT are desired; and resveratrol (Res) is a promising candidate to treat SCC together with PDT because of its anticancer effects [15].
Res is a stilbenoid, a type of natural phenol existing in red grapes, mulberries and other food products. It has been suggested to be effective against stages of carcinogenesis and to have strong effects against skin cancers [16, 17]. Res can act as an antioxidant and antimutagen, mediate anti-inflammatory effects and inhibit cyclooxygenase and hydroperoxidase functions. Moreover, it also inhibited tumorigenesis in a mouse skin cancer model [17]. In our previous study, we found that Res had anti-cancer effect on squamous cell carcinoma A431 xenografts in nude mice, inhibiting cells growth through the MAPK pathway [18]. Thus, we hypothesized that Res might be an effective supplement to improve outcome of PDT as a treatment for SCC.
In the present study, we combined Res administration with ALA-PDT treatment in A431 cell model and found that Res had a strengthening effect on cell proliferation and apoptosis through regulating MAPK pathway.
Reagents and chemicals
The Res was a commercial product from Chengdu Cisco company (98%, HPLC); The fetal calf serum and DMEM culture medium were commercial products from Invitrogen Corporation; ALA, tetrazolium blue (MTT) and p38 inhibitor SB203580 were from Sigma; RIPA lysis buffer was from Beyotime Institute of Biotechnology, Nantong, China. The primary antibody p-ERK, ERK, p-p38, p38 and p53 were purchased from CST; Caspase-3 and
Cell culture and treatment
The human HaCaT keratinocytes and human A431 epidermoid carcinoma cells were cultured in Dulbecco’s Modified Eagle’s Medium (containing 10% fetal bovine serum, 100 U/ml penicillin, 100
Cell proliferation assay
Cell viability was examined by MTT assay. For the experiment, HaCaT cells and A431 cells were seeded in a 96-well plate at a density of 1
Cell apoptosis analysis
The effect of Res or/and ALA-PDT on cell apoptosis was evaluated by flow cytometry, using an FITC Annexin V apoptosis kit as previously described [19]. In brief, cells were washed with cold PBS and resuspended at a concentration of 10
Western blot
Total protein was extracted from the cells with RIPA lysis buffer and the concentration of the protein was determined with Pierce™ BCA Protein Assay Kit. Then protein samples were separated by 12% SDS-polyacrylamide gels and transferred to PVDF membranes. Membranes were blocked in 5% non-fat milk for 1 h at room temperature and subsequently incubated overnight at 4
Statistical analysis
All data are expressed as mean
Results
Res treatment inhibits proliferation of A431 carcinoma cells combined with ALA-PDT therapy
Resveratrol/ALA-PDT promoted apoptosis in A431 carcinoma cells. Cell apoptosis were evaluated by flow cytometry. (A–B) Both Res and ALA-PDT, in single or in combination, could promote apoptosis in A431 cells. All the results were shown as mean 
Resveratrol/ALA-PDT activated MAPK pathway in A431 cells. (A) Both Res and ALA-PDT, in single or in combination, activated MAPK pathway, as indicated by upregulation of p-ERK and p-p38. Apoptosis related p53 and caspase-3 also had an increased protein expression treated by Res and ALA-PDT, in single or in combination; (B) Quantification of the results from panel A. The protein ratios were determined by the relative intensities of the protein bands. All the results were shown as mean 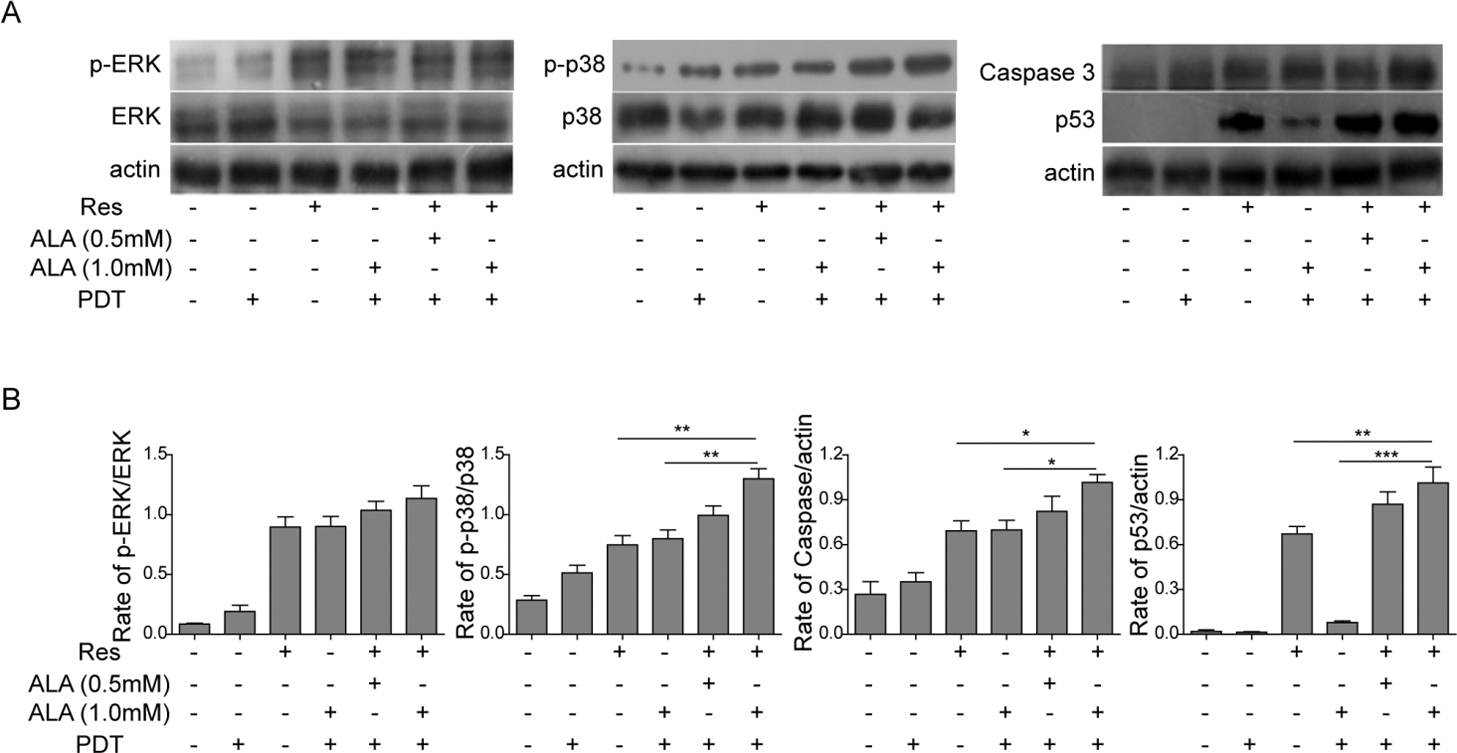
Res has been suggested to have strong effects against skin cancers. In our study, we first used MTT method to evaluate the effects of Res on HaCaT cells and A431 cells proliferation. When we increased Res concentration from 0 to 100 mg/L, we found that A431 cell viability was reduced from 100
p38 inhibitor SB203580 attenuated the effect of Res on A431 cells. (A) SB203580 abated the inhibitory effect of Res and/or ALA-PDT on A431 cells proliferation, as indicated by MTT assay. (B–C) SB203580 protected A431 cells from apoptosis when treated with Res and/or ALA-PDT. All the results were shown as mean 
Targeted cancer cell apoptosis is a research focus for cancer treatment [20]. So, we further evaluated the effectiveness of Res and ALA-PDT combined therapy in terms of A431 cell apoptosis. The apoptotic population was evaluated by using flow cytometry. Our results are consistent with previous cell viability test as shown in Fig. 2. Apoptosis rates of A431 cells treated with Res (39.55
Res activates MAPK pathway in A431 cells
In our study, we have highlighted the role of MAPK pathway in skin carcinogenesis and its modulation by Res and ALA-PDT, both in single and in combination, in A431 cells. We analyzed the expression of MAPK/ ERK and MAPK/p38 by Western-blot to evaluate the effects of Res and ALA-PDT treatment on activation of MAPKs. The results showed that treatment of Res and ALA-PDT independently resulted in phosphorylation of MAPK/ERK and MAPK/p38 in A431 cells (Fig. 3A). Moreover, combination treatment with Res and ALA-PDT resulted in significantly greater phosphorylation of MAPK/ERK and MAPK/p38 in A431 cells.
Besides, we also examined apoptosis related p53 and caspase-3 protein expression in each group. Combination treatment with Res and ALA-PDT also induced higher protein expression of p53 and caspase-3 than Res or ALA-PDT alone (Fig. 3A). These results indicated that Res enhances ALA-PDT resulted A431 cell apoptosis possibly through activating MAPK pathway.
p38 inhibitor SB203580 attenuates the effect of Res on A431 cells
Next, we tested whether suppression of MAPK pathway could block the effect of Res on A431 cells. A p38 inhibitor SB203580 was employed in this study. The results showed that addition of SB203580 (2
Discussion
The occurrence and development of malignant tumors are related to many factors, among which reduced apoptosis and abnormal cell proliferation are the key factors. Res has widely considered as a supplement for cancer treatment [21]. It has shown effects on several cancers, including liver cancer, breast cancer and prostate cancer [22, 23, 24]. In our previous study, we also found that Res was effective in the treatment skin cancer cells [18]. Additionally, researchers [25] found that Res also had anti-proliferation effect and could induced apoptosis in none-cancer cells, while others have found that Res had certain renal and bladder toxicity [26]. One the other hand, PDT is currently in clinical use for treatments of several tumors. The most prominent advantage of PDT is that it can accurately target the tumor tissue, but PDT treatment is mainly effective for shallow skin tumor and recurrence rates are pretty high [9]. Both Res and ALA-PDT treatments have their own limitations. Therefore, we proposed a combined treatment using both Res and ALA-PDT, then examined its effects and potential mechanism in this study.
We first studied the effects of Res and ALA-PDT on the skin squamous carcinoma A431 cells. MTT assay and flow cytometry were employed to detect cell proliferation and apoptosis respectively. The results showed that both Res and ALA-PDT could significantly inhibit the proliferation of A431 cells and promote the apoptosis. Furthermore, the inhibitory effect of Res on HaCaT cells viability was small compared to A431 cells. When the two treatments are used as a combination, the effect is significantly increased compared with single treatment, suggesting Res and ALA-PDT can be used together to treat SCC for a better clinical outcome. Combined treatments with ALA-PDT are common [14], and the popular co-adjuvants used with PDT for SCC include surgery, imiquimod and radiotherapy [27, 28, 29]. Researchers have found that recurrence rates of combined therapy are significantly lower than PDT alone. But so far, no one has used Res together with ALA-PDT. Thus, our results provided insight for a new combined therapy for SCC.
Next, we investigated expressions of apoptosis-related factors ERK, p38, p53, and caspase-3 with the present of Res, ALA-PDT and their combination. We found that significantly higher protein expression of p-ERK, p-p38, p53 and caspase-3, suggesting these proteins might be involved Res and ALA-PDT treatments. ERK and p38 are two important members of the MAPK family. The sustained activation of MAPK/ERK signal pathway is a significant sign of many human tumors, which can lead to the deficiency of cell apoptosis and differentiation and promote cell malignant transformation, abnormal proliferation and tumor growth [30, 31, 32]. But there are contrary opinions in recent years. Zhang et al. found that treatment of ALA-PDT on the skin squamous cell carcinoma SCL-1 cells could induce the phosphorylation of ERK1/2 and cell apoptosis. Another study in non-small lung cancer H460 cells also found that PDT promoted tumor cell apoptosis by activating p38 and JNK pathways [12]. In addition, one of our previous works found that Res could activate MAPK/ERK pathway and initiate p53-dependent apoptosis pathway resulting in growth inhibition of skin squamous cell carcinoma cells [18]. MAPK/p38 pathway is the main pathway involved in the initiation of apoptosis. p38 can induce apoptosis through phosphorylation of p53, enhancing c-myc expression, and is involved in Fas/FasL mediated apoptosis. Studies in gastric cancer and renal cell carcinoma found that activation of p38/MAPK pathway could inhibit tumor cell proliferation and induce apoptosis [33, 34]. When treated with p38 inhibitor SB203580, A431 cells showed decreased cell apoptosis and increased cell viability in all situations of Res and ALA-PDT combination. The results imply that p38 is involved in Res and ALA-PDT induced cancer treatment process.
In conclusion, our study demonstrated that Res could enhance the effect of ALA-PDT against skin cancer cells by inhibiting tumor cell proliferation and promoting tumor cells apoptosis through activation of MAPK pathway, suggesting that Res might act as an adjuvant to enhance the antitumor effect of ALA-PDT.
Footnotes
Acknowledgments
This study was supported by a Natural Science Foundation of Inner Mongolia Autonomous Region, grant No. 2014MS0895.
Conflict of interest
The authors declare no conflicts of interest in this work.
