Abstract
Endometrial carcinoma (EC) is a common malignant tumor in gynecology. Its incidence and development are closely associated with the levels of estrogenic and progesterone hormone. Extracellular signal-regulated kinase (ERK) signaling pathway abnormity is associated with a variety of tumors. This study detected estrogen receptor (ER), progesterone receptor (PR), ERK1, and ERK2 expression in EC and analyzed their correlations. A total of 40 EC patients in our hospital were selected as test group, while another 40 healthy volunteers were enrolled as control group. ER, PR, ERK1, and ERK2 expression in EC tissue, para-carcinoma tissue, and normal endometrial tissue were detected by immunohistochemistry and Western blot. The positive rate of ER, PR, ERK1, and ERK2 in the test group was 50%, 40%, 60%, and 65%, respectively, which were significantly higher than those in the control (
Introduction
The incidence and development process of malignant tumor involves multiple genes. A variety of signaling pathways regulate the different stages synergistically. One of the transductions abnormity may cause importantly physiological and pathological chang-es [1, 2, 3]. MAPK signaling pathway is found to play a critical role in the process of tumor cell proliferation, of which the extracellular signal-regulated kinase (ERK) presents the dominant role [4]. Estrogen and progestogen are important hormones in regulating endocrine. Their imbalance can affect the normal state. Endometrial carcinoma (EC) is characterized as hormone-dependence. Long-term high level of estrogen and low level of progestogen may stimulate endometrial hyperplasia, further leading to the development of EC. Estrogen can bind to estrogen receptor (ER), which is located in the nucleus, while progestogen can bind to progestogen receptor (PR). It was reported that ER and PR might be used to evaluate the degree of endometrial malignant hyperplasia. The abnormal levels of ER and PR can lead to the incidence and development of EC [5]. This study tested the expression levels of ER, PR, and ERK in EC patients and analyzed their correlations.
Materials and methods
General information
A total of 40 EC patients in the First People’s Hospital of Jining City were selected as test group with a mean age of 41.2
Inclusion criteria: Menstrual rule; no use of hormonal drugs within three months; no other ovary or uterus benign and malignant lesions; No serious liver and kidney disease.
Exclusion criteria: Endometrial hyperplasia; corpus uteri or cervical inflammation; long-term use of hormone drugs; other gynecological malignant tumors.
This study has been pre-approved by the ethical committee of the First People’s Hospital of Jining City. All subjects have signed the consent forms before recruitment in this study.
Reagents and instruments
ER, PR, ERK1, and ERK2 blocking buffer and rabbit anti mouse secondary antibody were purchased from Keygentec. ER, PR, ERK1, and ERK2 monoclonal antibodies were from Cell Signaling. Anti rabbit IgG-HRP was from Bioworld. Centrifugal machine was from Shanghai Flying Pigeon. Microplate reader was from TECNA. Image analysis system was from Hewlett Packard.
ER and PR expression in endometrial tissue
ER and PR expression in endometrial tissue
ERK1 and ERK2 expression in endometrial tissue
ER, PR, ERK1, and ERK2 protein expression in endometrial cells
The specimen was fixed and embedded before being sectioned at 3
Analysis: Brown granules appeared on cell membrane, cytoplasm, or nucleus were considered as positive. For the slice, negative (-) was defined as
Endometrial cell isolation
The endometrial tissue was cut into pieces under aseptic condition and put into the solution containing penicillin and streptomycin. After washed and digested, the cells were resuspended in DMEM with a number of 10
Western blot
A total of 40
Statistical analysis
The statistical analysis was performed on SPSS17.0 software. The data was depicted as mean
Results
ER and PR expression in endometrial tissue from patients detected by immunohistochemistry
The positive rate of ER and PR in the test group was 50% and 40%, respectively. They were significantly higher than those in the paracarcinoma tissue and control (
ER and PR expression in endometrial tissue (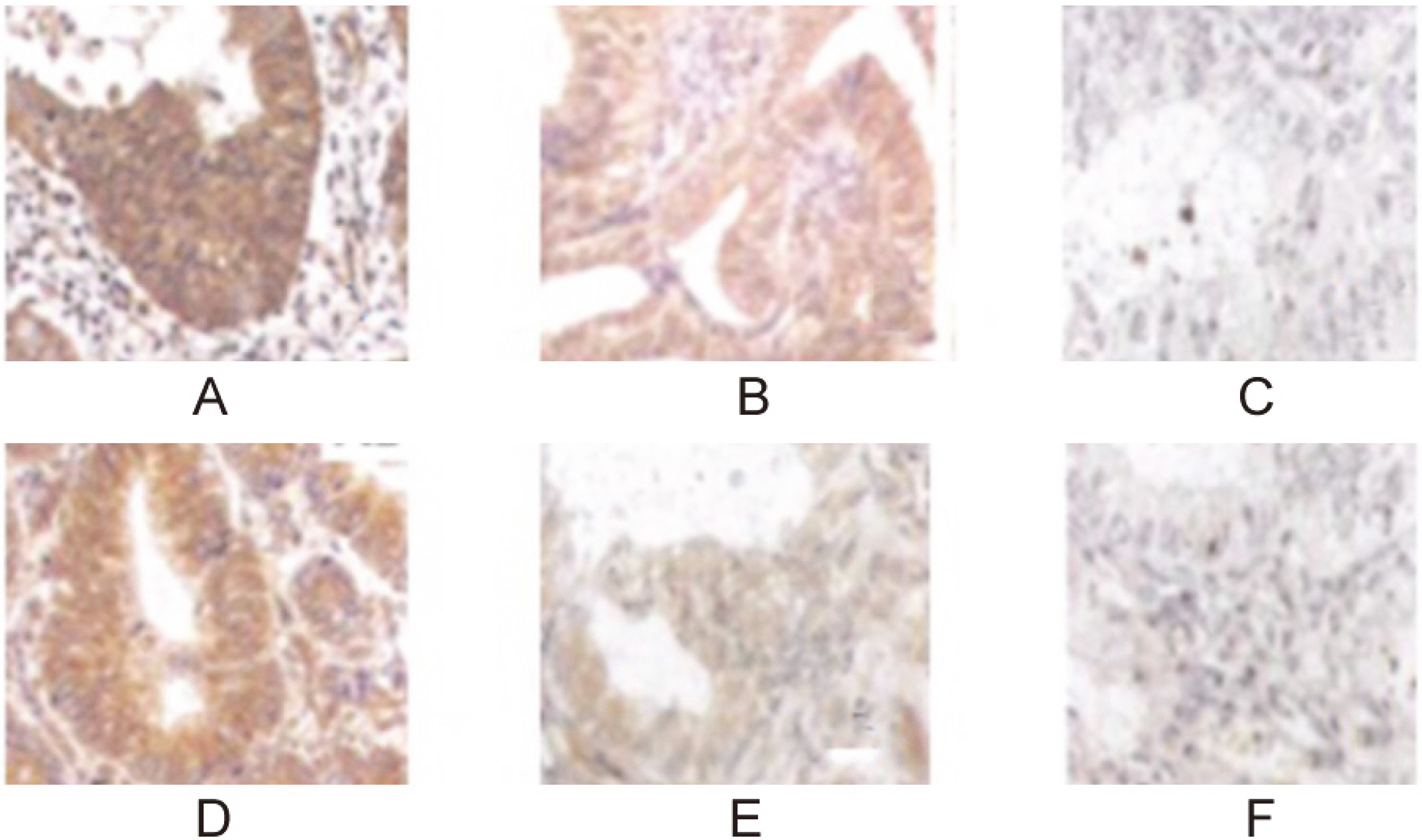
The positive rate of ERK1 and ERK2 in the test group was 60% and 65%, respectively. They were significantly higher than those in the paracarcinoma tissue and control (
ERK1 and ERK2 expression in endometrial tissue (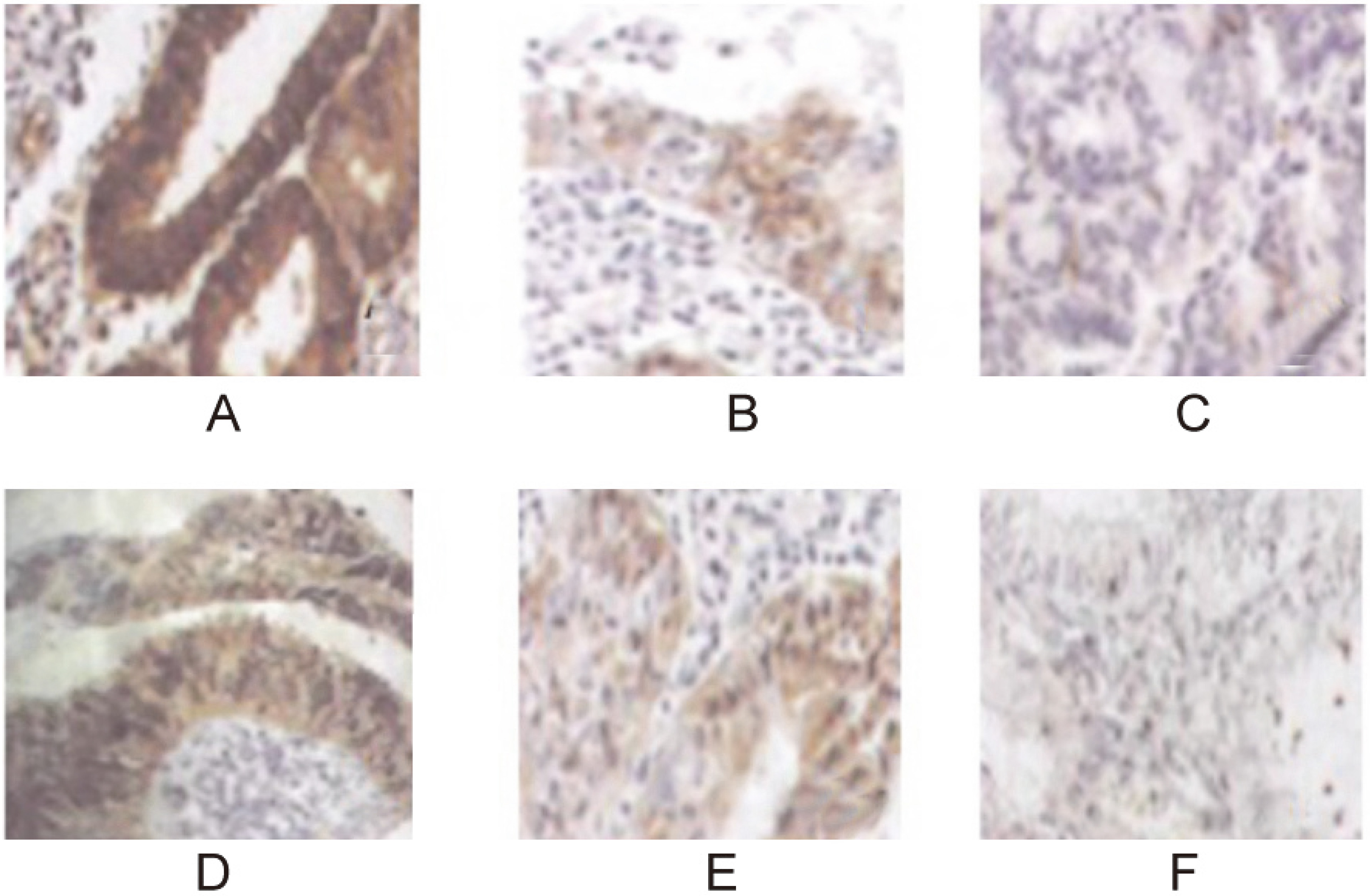
Western blot was applied to test ER, PR, ERK1, and ERK2 protein expressions in endometrial cells. It was found that their levels in test group weree markedly higher than those in the control (
ER, PR, ERK1, and ERK2 protein expression in endometrial cells.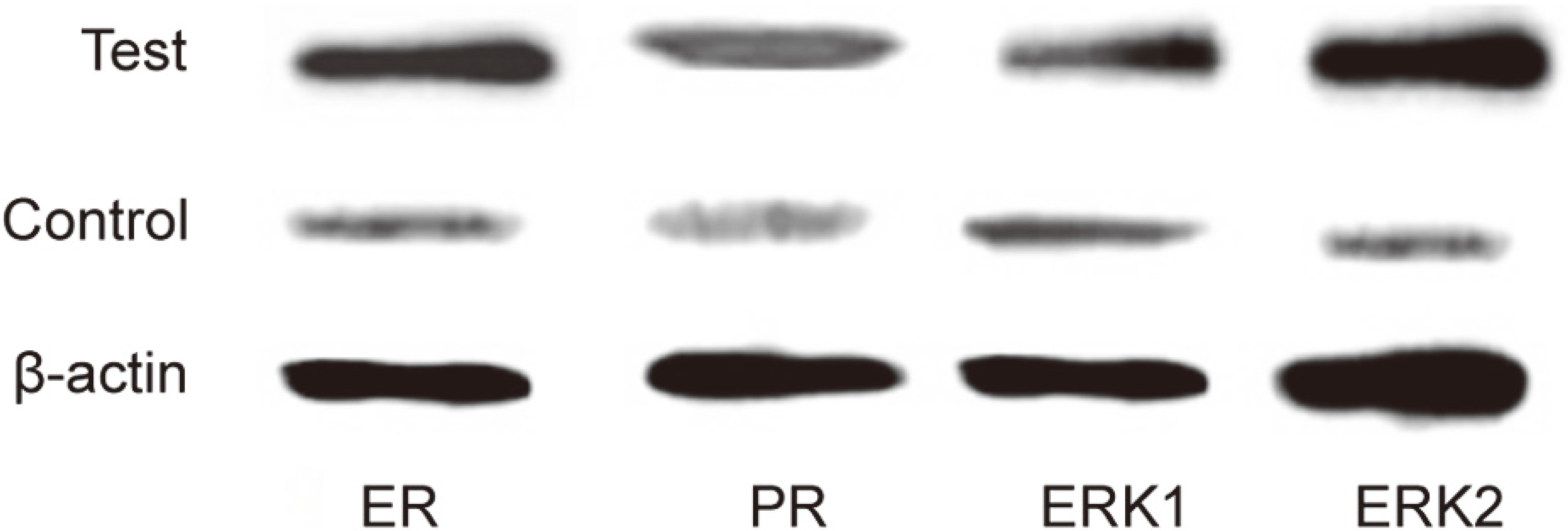
The relationship of ERK1 expression with ER and PR in endometrial carcinoma cells was also analyzed. The positive rate of ERK1 was lower in ER negative EC patients, while it was higher in ER positive EC patients. ERK1 showed a positive correlation with ER (
The relationship of ERK1 expression with ER and PR
The relationship of ERK1 expression with ER and PR
The relationship of ERK2 expression with ER and PR in endometrial carcinoma cells was analyzed. The positive rate of ERK2 was lower in ER negative EC patients, while it was higher in ER positive EC patients. ERK2 exhibited a positive correlation with ER (
The relationship of ERK2 expression with ER and PR
The relationship of ERK2 expression with ER and PR
EC is a common female malignant tumor with younger trend and increasing incidence. As one of the most women malignant tumors only after breast cancer, it seriously impairs women’s health. There are numerous prognosis factors, including clinical stage, vascular involvement, interstitial infiltration, and histological grading. Following the development of molecular biological technology, malignant tumor related factors were gradually identified. It was considered that some abnormal factors, such as oncogene activation and tumor suppressor gene inactivation, can block the signaling transduction [6, 7]. ERK signaling pathway can be activated by a variety of extracellular factors, such as growth factors and transcription factors, to regulate cell growth [8, 9]. This study selected EC patients in our hospital to detect ER, PR, ERK1, and ERK2 expression levels and analyze their correlation.
It was found that ER and PR were highly expressed in EC tissues. Furthermore, the positive rate of ERK1 and ERK2 reached 60% and 65%, respectively, which was significantly higher than those in paracarcinoma tissue and control. It suggested that ERK1 and ERK2 were highly expressed in EC tissue. Moreover, it was showed that ER, PR, ERK1, and ERK2 protein expressions in the EC cells were also significantly upregulated. ER belongs to a nuclear hormone receptor family, while PR belongs to a steroid hormone receptor family [10, 11]. They can regulate EC development. Hormone has the role of stimulating intimal hyperplasia. Low level of the hormone stimulation may increase the activity of glands in atrophic intima, leading to promotion of EC incidence and development [12]. ERK1 and ERK2 signaling pathways have been found to be activated in multiple malignant tumors, such as lung cancer and breast cancer [13]. Yasunari demonstrated that ERK1 and ERK2 were excessively activated in EC independent of KRAS and BRAF, and can affect the prognosis [14, 15].
This study further revealed that ERK1 and ERK2 were positively correlated with ER and PR. It was reported that estrogen participated in mediating ERK activation in a variety of malignant tumors, such as astrocytoma and osteoblastoma [16]. Previous study transfected ER to Hela cells and found ER can activate ERK signaling pathway. The level of estrogen may impact ERK signaling pathway [17]. It was demonstrated that estrogen elevation in breast cancer can stimulate cancer cells proliferation [18]. More than that, as an important signaling pathway in the body, ERK may stimulate cancer cells proliferation that is closely associated with estrogen level in the microenvironment [18, 19, 20]. ERK plays an important role in the regulation of EC incidence and development.
Conclusion
ER, PR, ERK1, and ERK2 were highly expressed in EC patients. ERK1 and ERK2 were positively correlated with ER and PR. Interactions among ER, PR ERK1 and ERK2 might participate in EC incidence and development. However, whether ER and PR regulate ERK1 and ERK2 in EC remains unclear and requires in-depth investigation, which may provide new orientation for EC prognosis and treatment.
Footnotes
Conflict of interest
The authors have no conflict of interest to report.
