Abstract
OBJECTIVE:
To explore the correlation of PD-1/PD-L1 polymorphisms and their expressions with clinicopathologic features and prognosis of ovarian cancer.
METHODS:
A total of 164 patients with ovarian cancer were enrolled as case group and 170 healthy women as control group. We conducted quantitative reverse transcription-PCR (qRT-PCR) to determine PD-1/PD-L1 expressions in peripheral blood mononuclear cells (PBMCs). Polymerase chain reaction-restriction fragment length polymorphism (PCR-RFLP) and allele-specific amplification were used to detect PD-1 rs2227982 C>T and PD-L1 rs4143815 C>G.
RESULTS:
PD-1 rs2227982 C>T and PD-L1 rs4143815 C>G polymorphisms increased the risk for ovarian cancer. PD-1 rs2227982 C>T was associated with FIGO stage and differentiation grade, while PD-L1 rs4143815 C>G was correlated with histological type and differentiation grade. Besides, PD-1/PD-L1 expressions were positively correlated in PBMCs of patients with ovarian cancer to be associated with differentiation grade. Compared with wild homozygous patients, PD-1/PD-L1 expressions were significantly decreased in PBMCs of ovarian cancer patients carried with the mutant genotypes of rs2227982 C>T and rs4143815 C>G. The PFS and OS in ovarian cancer patients with wild homozygous genotype of rs2227982 C>T and rs4143815 C>G were significantly higher than those with mutant genotypes, which were significantly lower in patients with low expressions of PD-1/PD-L1 than those with high expressions. Univariate COX regression analysis identified FIGO staging, differentiation grade, rs2227982 C>T, rs4143815 C>G and expressions of PD-1/PD-L1 as the prognostic factors, and multivariate COX regression analysis demonstrated that high FIGO stage and low expressions of PD-1/PD-L1 were independent risk factors for the prognosis of ovarian cancer.
CONCLUSION:
PD-1 rs2227982 C>T and PD-L1 rs4143815 C>G polymorphisms increased the risk of ovarian cancer, leading to a poor prognosis, associated with low expressions of PD-1 and PD-L1. While high PD-1 and PD-L1 expressions are indicators of a favorable prognosis in ovarian cancer.
Introduction
Ovarian cancer, as one of the three most common malignant tumors in gynecology, ranks top two in morbidity and top one in mortality respectively from gynecological malignancies [1]. Statistically, about 238,700 individuals were diagnosed with ovarian cancer, and 151,900 patients were died of ovarian cancer in 2012 [2]. Unfortunately, owing to the unspecific symptoms and a lack of early adequate screening method, ovarian cancer is usually diagnosed at advanced clinical stage [3]. Currently, a variety of factors are known to involve in ovarian cancer, with the continuous research and the development of technique, to influence its clinical prognosis, such as age at diagnosis, fertility, family history, mental, environmental, and dietary factors [3, 4], as well as the genetic susceptibility [5], which may possibly result in gene expression difference in individuals, and thereby playing an important in tumor survival [6], including in ovarian cancer [7].Programmed cell death-1, also named as PD-1 or CD279, a newly found gene related to immune escape, is located on chromosome 2q37.3 and encodes a 50
Comparison of baseline characteristics between case group and control group
Comparison of baseline characteristics between case group and control group
Note:
Therefore, this study enrolled 164 patients with OC and 170 healthy women by conducting quantitative real-time polymerase chain reaction (qRT-PCR) to determine PD-1/PD-L1 expressions in peripheral blood mononuclear cell (PBMC) as well as polymerase chain reaction-restriction fragment length polymorphism (PCR-RFLP) and allele-specific amplification to detect PD-1 rs2227982 and PD-L1 rs4143815, to further analyze the associations of PD-1/PD-L1 expression and its genetic polymorphism with clinicopathological features and prognosis of ovarian cancer.
Ethics statement
Following the Declaration of Helsinki [26], this study has been obtained informed consents in written form from all participants and approved by the ethics committee of the First People’s Hospital of Jingzhou City.
Study subjects
A total of 164 women with ovarian cancer aged 38
Quantitative reverse transcription-PCR (qRT-PCR)
The peripheral venous blood was collected from fasting patients using 3 ml ethylenediaminetetraacetic acid, and PBMCs were isolated by Ficoll gradient centrifugation. Following protocols of manufacturer, TRI (Life Technologies, Invitrogen, Thessaloniki, Greece) was used to isolate the total RNA. Then, with random 6-mer oligonucleotide primer (50 pmol/mL) (Roche, USA) and M-MLV reverse transcriptase (Invitrogen), total RNA of 1 mg was reversely transcribed into complementary DNA (cDNA), according to the manufacturer’s introduction. In the automated thermocycler RotorGene 6000 (Corbett Life Science, Sydney, Australia), SYBR-Green PCR Supermix (Invitrogen, UK) was used to determine PD-1/PDCD1 (encodes PD-1) and PD-L1/PDCD1LG1 (encodes PD-L1) at the mRNA level. The
Primers and PCR conditions for the amplification of the analyzed genes
Primers and PCR conditions for the amplification of the analyzed genes
The whole blood was taken to extract genomic DNA using Universal Genomic DNA Extraction Kit Ver. 3.0 (TaKaRa, Japan). With amplification in a T-Gradient Thermoblock PCR System (Biometra, Germany), the genotypes of PD-1 rs2227982 polymorphism was detected by PCR-RFLP method. The total of 50
Follow-up
The follow-up started from discharge time after systemic treatment till December 2016. During follow-up, patients who died of other causes, or were lost to follow-up or were survivors till the end of research, were recorded as censoring, and patients lost to follow-up were treated according to the last statistical time. The follow-up was carried out via outpatient service, telephone or medical record. The survival time was calculated as month. The OS was the time starting from date of diagnose with ovarian cancer till the date of death caused by any reason. The PFS was the time beginning from the date of diagnose with ovarian cancer till the date of disease progression and death.
Statistical analysis
All data were analyzed using SPSS 21.0 software (SPSS Inc., Chicago, IL, USA). Represented as mean
Results
Association of PD-1 rs2227982 C>T and PD-L1 rs4143815 C>G polymorphisms with the risk of ovarian cancer and its clinicopathologic features
Association of PD-1 rs2227982 C>T and PD-L1 rs4143815 C>G polymorphisms with the risk of ovarian cancer
Association of PD-1 rs2227982 C>T and PD-L1 rs4143815 C>G polymorphisms with the risk of ovarian cancer
Association of PD-1 rs2227982 C>T and PD-L1 rs4143815 C>G polymorphisms with clinicopathological features
The genotype frequencies of PD-1 rs2227982 C>T and PD-L1 rs4143815 C>G in the control group were in accordance with Hardy-Weinberg equilibrium (
The qRT-PCR results demonstrated that the relative mRNA expression levels of PD-1 and PD-L1 were significantly increased in PBMCs of patients with ovarian cancer as compared with the control group (both
Correlation of PD-1/PD-L1 expressions with clinicopathologic features in PBMCs of patients with ovarian cancer
Correlation of PD-1/PD-L1 expressions with clinicopathologic features in PBMCs of patients with ovarian cancer
Notes:
Expressions of PD-1 and PD-L1 in PBMCs of patients with ovarian cancer. Notes: A, Expressions of PD-1 and PD-L1 in PBMCs in ovarian cancer patients and controls by qRT-PCR. *
Expressions of PD-1 and PD-L1 in PBMCs of ovarian cancer patients with different genotypes of PD-1 rs2227982 C>T and PD-L1 rs4143815 C>G Notes: A, Expressions of PD-1 and PD-L1 in PBMCs of ovarian cancer patients with different genotypes of PD-1 rs2227982 C>T; B, Expressions of PD-1 and PD-L1 in PBMCs of ovarian cancer patients with different genotypes of PD-L1 rs4143815 C>G; 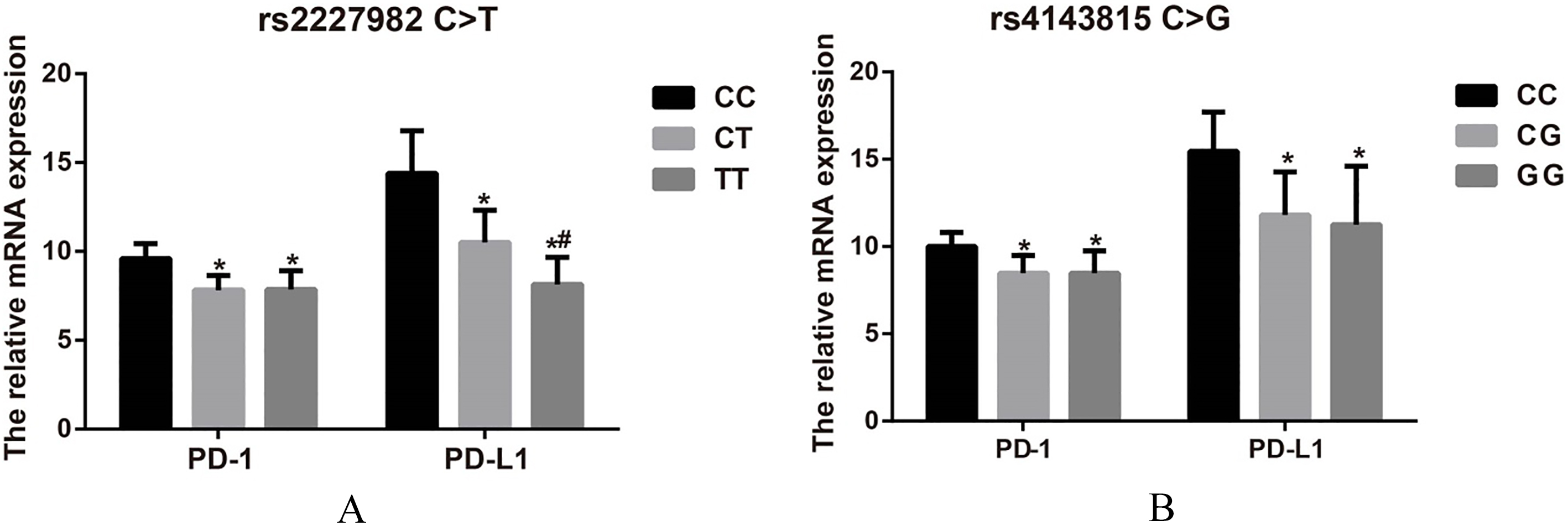
Effects of PD-1/PD-L1 polymorphisms and their expressions on the prognosis of patients with ovarian cancer. Notes: A-B, Effects of PD-1 rs2227982 C>T (A) and PD-L1 rs4143815 C>G (B) on PFS of patients with ovarian cancer; C-D, Effects of PD-1 rs2227982 C>T (C) and PD-L1 rs4143815 C>G (D) on OS of patients with ovarian cancer; E-F, Effects of PD-1 (E) and PD-L1 (F) expressions on PFS of patients with ovarian cancer; G-H , Effects of PD-1 (G) and PD-L1 (H) expressions on OS of patients with ovarian cancer.
As revealed in Fig. 2, when compared with wild homozygote carriers, both PD-1 and PD-L1 expressions were significantly decreased in PBMCs of ovarian cancer patients who were the mutant carriers of PD-1 rs2227982 C>T and PD-L1 rs4143815 C>G (all
The ovarian cancer patients were followed up for 5 years, and the loss-to-follow-up rate was 4.88% (8/164). Kaplan-Meier survival curve showed that significant differences in PFS and OS were found in patients with ovarian cancer in different genotypes of PD-1 rs2227982 C>T and PD-L1 rs4143815 C>G (all
Risk factors for prognosis of patients with ovarian cancer by Cox regression model
Risk factors for prognosis of patients with ovarian cancer by Cox regression model
Notes: FIGO, Federation of Gynecology and Obstetrics; values in bold font is to underscore statistical significance (
In this study, one of the most striking results was PD-1 rs2227982 C>T can affect the PD-1 expression in PBMCs to be involved in the occurrence and development of ovarian cancer. Currently, there still exists controversy about the role of PD-1 rs2227982 C>T in diseases, taken an example by Piskin et al., the frequency of T alleles in the PD-1 rs2227982 polymorphism was higher in control group than patients with subacute sclerosing panencephalitis (SSPE) [28], suggesting a protective role of T allele in SSPE. Dong W and his group carried out a meta-analysis stating that no significant association was observed between rs2227982 polymorphism and cancer risks in all genetic models and alleles [19]. Nevertheless, our results showed that the T allele within the PD-1 rs2227982 C>T significantly increased the risk for ovarian cancer, and such carriers of ovarian cancer patients had a higher FIGO stage but a lower differentiation grade. Consistently, both Qian and Yang teams demonstrated that the single nucleotide polymorphism (SNP) rs2227982 of the PD-1 gene may confer susceptibility to the development of ankylosing spondylitis, and the carriage of the TT genotype can enhance the thoracolumbar kyphosis severity in ankylosing spondylitis patients [29, 30]. And patients with dilated cardiomyopathy presented higher frequencies of TT genotype and T allele of rs2227982, as revealed by Yang et al. [31], indicating T allele of rs2227982 C>T was a risk factor for ankylosing spondylitis, dilated cardiomyopathy, as well as ovarian cancer and other diseases, despite of different mechanisms in different diseases. To our knowledge, gene mutation can affect diseases progression via altering its transcriptional level, for example, the NF-
Furthermore, another important finding was that patients carrying PD-L1 rs4143815 C>G C/G, G/G and CG+GG genotypes and G allele were in increased risk for ovarian cancer, and this SNP was significantly correlated with histological type and differentiation grade. Multiple researchers have found that the PD-L1 rs4143815 G allele carriers had an obvious elevation with the risk of numbers of diseases as compared with C allele carriers, such as gastric cancer [23] and Type I diabetes [33], which were consistent with our results, possibly because G allele in the SNP rs4143815 of PD-L1 can reduce luciferase activity via influencing miRNA binding, exhibiting a reduced PD-L1 expression [34, 35]. What’s more, Kataoka et al. reported structural variations disrupted PD-L1 3’-UTR and thereby resulted in abnormal PD-L1 expression, which capacitates immune evasion in various cancers [36]. Therefore, we conducted qRT-PCR to determine the transcription level of PD-L1 and found that PD-L1 expression was significantly decreased in PBMCs of patients carried with rs4143815 mutant genotype, indicating that PD-L1 rs4143815 C>G can potentially regulate PD-L1 expression in ovarian cancer.
PD-1/PD-L signaling pathway is one of the most important pathways inhibiting the tumor immune response, and one previous study indicated that PD-L1 expression on tumor cells or antigen-presenting cells (APCs) in the tumor microenvironment could promote tumor growth and induce the apoptosis of T cells of PD-1 with antitumor activity, further leading to poor-prognosis and therapy-resistant, such as esophagus cancer [37] and renal cell carcinoma [38]. Of note, in spite of their well-known immune-modulatory function, the high expression of PD-1/PD-L also suggested a good prognosis in pancreatic cancer [14], thymic cancer [39], and malignant melanoma [40], and there is a possible explanation that PD-1 and PD-L1 is in compensatory up-regulation in a microenvironment which may threaten tumors by means of an active immune response [16] Moreover, this study also revealed that PFS and OS in ovarian cancer patients with high PD-1 and PD-L1 expression were significantly higher than those patients with low expression, and high FIGO stage and low expressions of PD-1 and PD-L1 were independent risk factors for the prognosis of patients with ovarian cancer. As illustrated by Darb-Esfahani et al., high PD-1 and PD-L1 expression levels are indeed indicators of a good prognosis in ovarian cancer [16]. but PD-L1 expression was found to exert a negative prognostic effect in ovarian cancer cells in Hamanishi et al. article [17], which might be partially associated with only 40% of serous carcinomas cases investigated in this study, while our study enrolled 65% patients with serous ovarian cancer. Besides, PFS and OS were significantly higher in patients carrying wild homozygote genotypes of PD-1 rs2227982 C>T and PD-L1 rs4143815 C>G than mutant genotype carriers. Considering the above, we reasonably speculated that PD-1/PD-L1 mutation may lead to aberrant gene expression, and consequently influencing the prognosis of patients with ovarian cancer.
Taken together, PD-1 rs2227982 C>T and PD-L1 rs4143815 C>G mutation increased the risk of ovarian cancer, which was associated with down-regulated PD-1/PD-L1 expression in PBMCs. Moreover, patients carried with the wild homozygous genotype of rs2227982 C>T and rs4143815 C>G had significantly higher PFS and OS than mutant genotype carriers, and higher expressions of PD-1 and PD-L1 indicated a better prognosis with higher PFS and OS. However, PD-1/PD-L1 expressions in cancer cells and tumor-infiltrating lymphocytes (TILs) as well as other SNPs in PD-1/PD-L1 gene (especially rs2297136 and rs2282055) were not determined due to the limited time and fees, which needs to be further explored in the following experiments.
Footnotes
Acknowledgments
The authors appreciate the reviewers for their useful comments in this paper.
Conflict of interest
The authors declare no conflicts of interest.
