Abstract
Numerous studies have proved that microRNAs (miRNAs) play crucial roles in the tumorigenesis and progression of gastric cancer (GC). Our study was to investigate the correlation between miR-338-3p expression and clinical features as well as prognosis of GC. A total of 138 GC tissue specimens and adjacent non-cancerous tissues were collected for further analysis, then quantitative PCR method was used to detect the relative miR-338-3p expression. Our study showed that tissue miR-338-3p level was greatly decreased in cancer tissues compared with paired normal tissues. Furthermore, loss of tissue miR-338-3p was positively associated with aggressive clinical characteristics (advanced clinical stage, poorer differentiation and lymph node invasion), shorter overall survival and recurrence free survival. Finally, tissue miR-338-3p expression was confirmed to be an independent prognostic factor for GC. Overall, our findings indicate that miR-338-3p acts as a tumor suppressor in GC and tissue miR-338-3p might serve as a novel prognostic biomarker of GC.
Introduction
Gastric cancer (GC) is one of the most common human malignancy and the second leading cause of cancer-related deaths around the world. In China, the mortality of GC has steadily declined since the early 1990s, but the incidence and mortality rates of this deadly disease remain high [1, 2]. Unfortunately, most GC patients were diagnosed at a late stage with unfavorable clinical outcome [3, 4]. Thus, it is urgent to identify effective markers to improve prognosis prediction of GC and develop new therapies for this disease.
MicroRNAs are a family of small, non-coding RNAs which regulate gene expression through binding to the 3’ untranslated region (UTR) of target genes, leading to degradation and inhibition of translation of target mRNAs [5, 6]. Altered miRNAs expression had been observed in many types of cancer and demonstrated to involve in multiple biological processes [7, 8]. MiRNAs had been identified to function as oncogenes or tumor suppressors in GC. For instance, Wang et al. showed that deletion of miR-211 enhanced GC cell proliferation and invasion by targeting SOX4 [9]. Conversely, inhibition of miR-215 expression suppressed GC cell growth, migration and invasion through up-regulating RUNX1 [10].
MiR-338-3p, which located on chromosome 17q25, is a highly conserved gene [11]. Recent studies have found that miR-338-3p is aberrantly expressed in different tumor types including gastric cancer [12, 13, 14]. In addition, miR-338-3p inhibits the proliferation and migration capacity of gastric cancer cells [12]. Therefore, we hypothesized that miR-338-3p played a tumor suppressive role in gastric cancer and reduced miR-338-3p was associated with poor clinical outcome of gastric cancer. In this study, we aimed to assess tissue miR-338-3p expression in GC patients and analyze its potential prognostic value.
Materials and methods
Patients and tissue samples
This study was authorized by the Ethics Committee of the First Hospital of Jilin University. Written informed consent was collected from all the participants. All samples were handled and made anonymous according to the ethical and legal standards.
The tumor samples and adjacent normal tissues were obtained from 138 patients (89 males, 49 females; 59.2
Correlation between miR-338-3p expression with clinicopathological characteristics in gastric cancer
Correlation between miR-338-3p expression with clinicopathological characteristics in gastric cancer
Total RNA was extracted from tissue specimens by using Trizol reagent (Invitrogen Corp, Carlsbad, CA, USA) in accordance with the manufacturer’s instructions. RNA concentration was measured using the Epoch Multi-volume Spectrophotometer System (BioTek, Winooski, VT, USA). Reverse transcription was carried out using the TaqMan MicroRNA Reverse Transcription Kit (Applied Biosystems, Foster City, CA, USA). Quantitative PCR was performed with the Maxima SYBR Green qPCR Kit (Thermo Scientific, CA, USA) on ABI7500 real-time PCR detection system (Applied Biosystems, Foster City, CA, USA). The relative amount of miR-338-3p expression was normalized using U6 small nuclear RNA (RNU6B) by the 2
Statistical analysis
The difference in miR-338-3p expression level between tumor tissues and paracancerous normal samples was evaluated with Mann-Whitney U-test. A receiver operating characteristic (ROC) curve analysis was used to identify the cut-off point to divide the gastric cancer patients into high and low miR-338-3p expression group. Association of miR-338-3p expression with clinical parameters was analyzed by Chi-square test. Survival curves were calculated by Kaplan-Meier method and compared with log-rank tests. The independent prognostic factors of OS and RFS were evaluated by the multivariate survival analysis using Cox proportional hazards regression model. P-values less than 0.05 were defined statistically significant. All statistical analyses were processed with GraphPad Prism (V. 5.0, La Jolla, CA, USA).
Results
MiR-338-3p was significantly down-regulated in GC tissues
In this study, qRT-PCR was used to measure miR-338-3p levels in 138 paired GC tissues and non-neoplastic tissues. The results showed that tissue miR-338-3p expression was greatly decreased in GC tumor samples in comparison with corresponding normal tissues (
MiR-338-3p expression was significantly down-regulated in GC tissues than adjacent noncancerous tissues.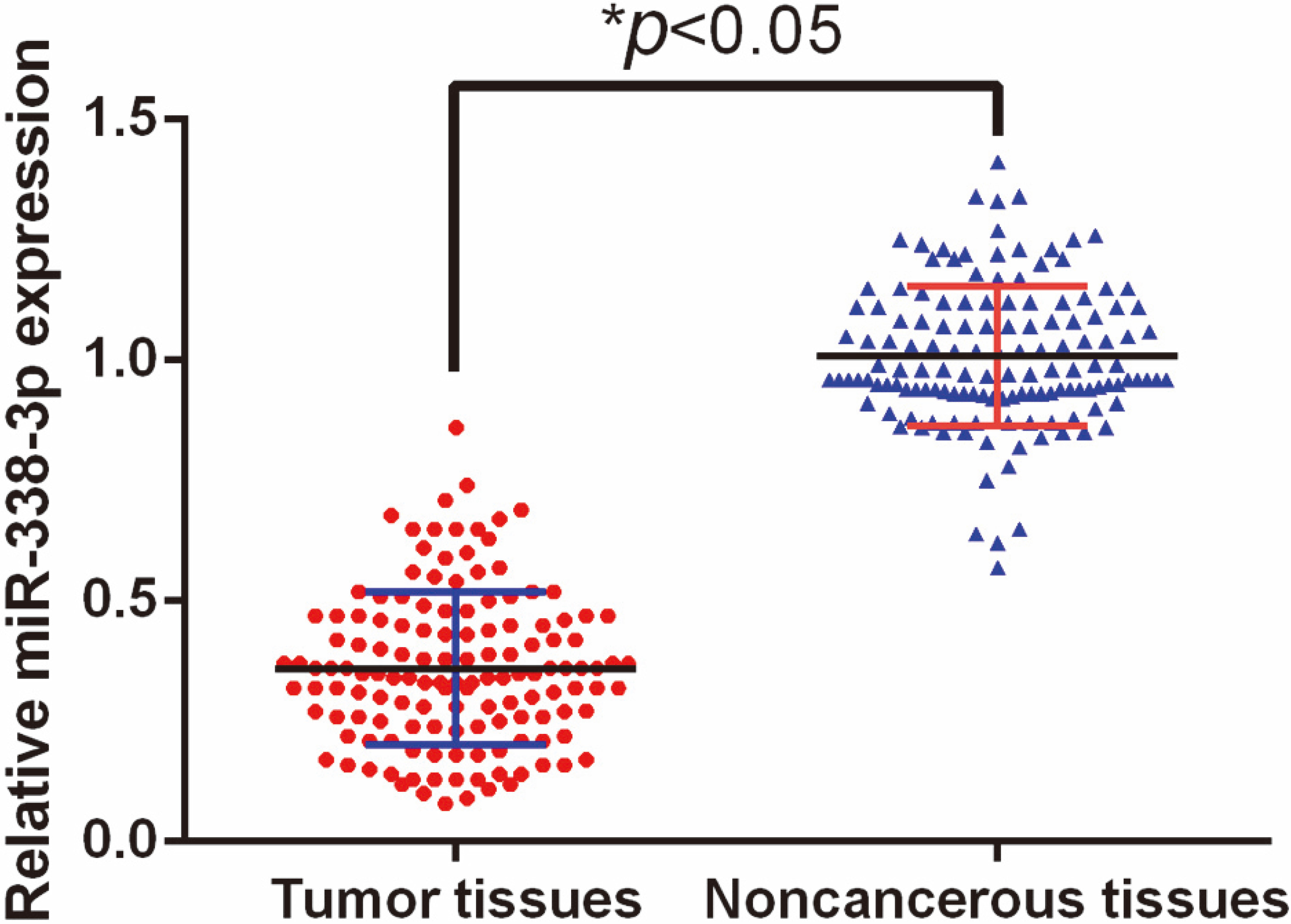
To further analyze the correlation between tissue miR-338-3p expression and clinical variables of the GC patients, all the patients were classified into high expression group (
Of note, we found that tissue miR-338-3p expression in GC patients with lower clinical stage was strongly increased compared to those with higher clinical stage (
Multivariate analysis of overall survival and recurrence free survival in 138 GC patients
Multivariate analysis of overall survival and recurrence free survival in 138 GC patients
A. Low miR-338-3p expression was closely correlated with higher clinical stage. B. Low miR-338-3p expression was closely correlated with poorly differentiated tumors. C. Low miR-338-3p expression was closely correlated with lymph node invasion.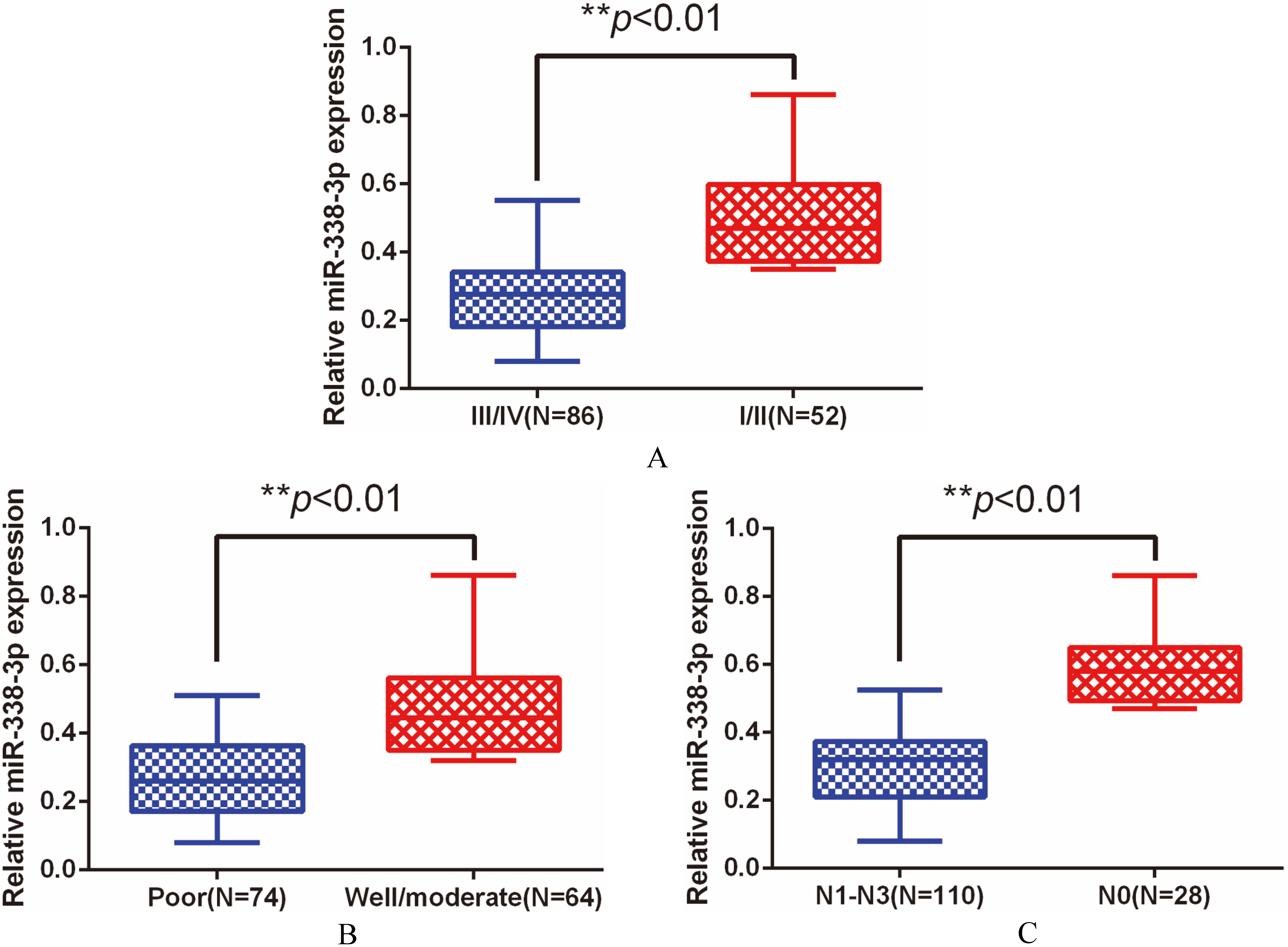
A. Kaplan-Meier curve of overall survival in 138 GC patients. B. Kaplan-Meier curve of recurrence free survival in 138 GC patients.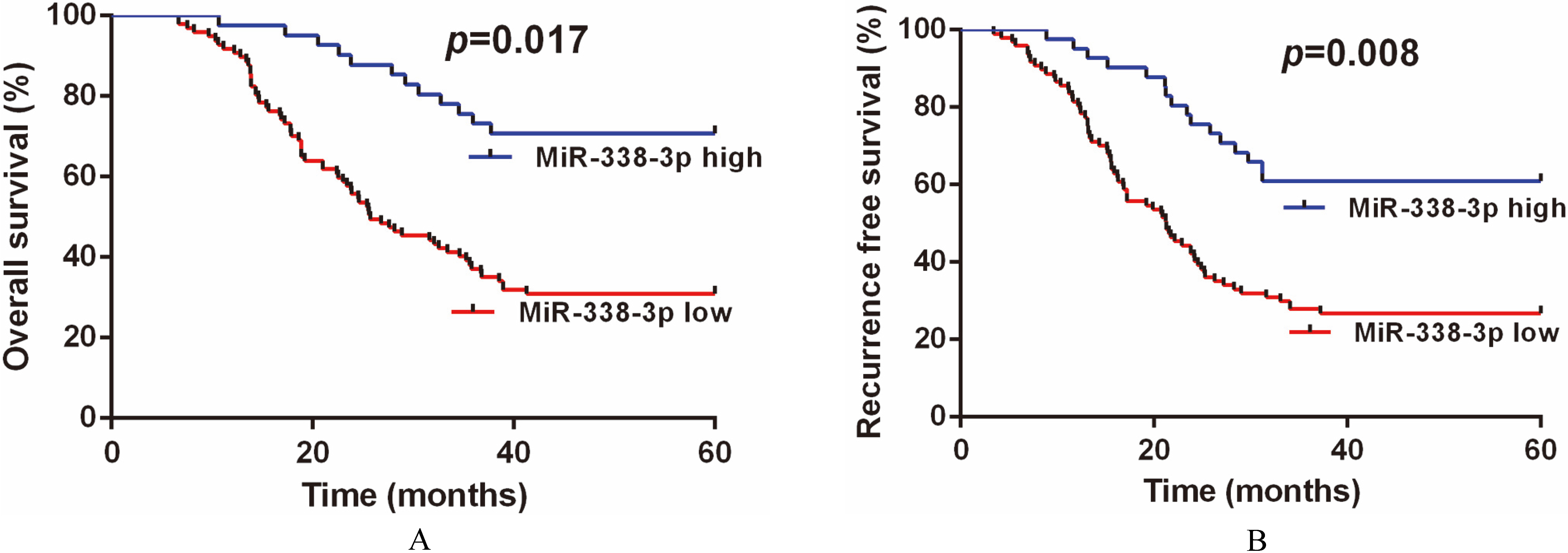
Kaplan-Meier analysis demonstrated that the patients with low tissue miR-338-3p level had worse OS (
Discussion
Emerging evidence revealed that abnormal miRNA expression was associated with tumor development and progression [6, 7, 8]. In the current study, we demonstrated that tissue miR-338-3p expression was significantly decreased in tumor tissues compared with normal tissues. Furthermore, down-regulation of tissue miR-338-3p expression was positively associated with aggressive clinicopathological parameters and shorter OS/RFS. Moreover, tissue miR-338-3p level was identified to be an independent prognostic factor for GC. Conclusively, these results implied that miR-338-3p might act as a tumor suppressor in GC.
The role of miR-338-3p expression in GC had been analyzed by some previous studies. For example, Chen et al. showed that restoration of miR-338-3p expression repressed the oncogenic activities of GC cells in vitro via targeting a disintegrin and metalloproteinase 17 [12]. Moreover, Guo and colleagues demonstrated that enhanced miR-338-3p expression inhibited carcinogenesis of GC cells in vivo by directly silencing P-Rex2a [15]. Similarly, Huang et al. revealed that up-regulation of miR-338-3p suppressed epithelial-mesenchymal transition in GC cells through degrading zinc finger E-box-binding protein 2 [16].
Besides GC, miR-338-3p functioning as a tumor suppressor had also been proved in other types of cancers. In ovarian cancer, Zhang and colleagues showed that miR-338-3p level was under-expressed in both cancer tissues and cell lines. Loss of miR-338-3p promoted cancer cell proliferation [13]. In nonsmall cell lung cancer, overexpression of miR-338-3p restrained cancer cell growth and invasion via regulating ras-related protein 14, ZEB2 or IRS2 [14, 17, 18]. Furthermore, Zhang et al. revealed that miR-338-3p expression was negatively associated with worse clinical outcome [18]. Elevated expression of miR-338-3p inhibited neuroblastoma proliferation, invasion and migration by down-regulating PREX2a [19]. In colorectal carcinoma, up-regulation of miR-338-3p suppressed cancer cell viability by inversely regulating smoothened [20, 21]. In addition, inhibition of tissue miR-338-3p expression was strongly correlated with poorer prognosis in colorectal cancer [22]. In hepatocellular carcinoma, forced miR-338-3p expression greatly reduced cell growth and induced cell apoptosis by targeting HIF-1, FOXP4 or SMO [23, 24, 25]. Additionally, a reduction in miR-338-3p expression was detected in both glioma tissues and cell lines. Ectopic miR-338-3p expression was correlated with lower histological grade [26]. These consisted with our findings. Nevertheless, the limitation of this study was that the number of tissue samples were relatively small. Thus, further analysis will be required using a larger cohort of GC patients.
In summary, our study revealed that decreased tissue miR-338-3p expression was significantly associated with poorer clinical outcome and unfavorable survival. As a result, our data indicated that tissue miR-338-3p might serve as a promising prognostic indicator for GC patients.
