Abstract
BACKGROUND:
Epithelial-mesenchymal transition (EMT) plays a crucial role in circulating tumor cells (CTCs) dissemination and cancer metastasis.
OBJECTIVE:
To investigate the EMT phenotypes of CTCs in hepatocellular carcinoma (HCC) patients and the clinical utility in the early diagnosis of HCC metastasis and progression.
METHODS:
We retrospectively analyzed the count and EMT classification of CTCs detected by the CanPatrol
RESULTS:
CTCs were detected in 95% of the 195 HCC patients with a range of 0–86 CTCs. Total CTCs numbers were correlated with BCLC stages, metastasis and serum AFP levels. The AUC of the ROC curve was 0.861 (95% CI: 0.782–0.940) in discriminating metastatic HCC patients with non-metastatic patients. Epithelial, hybrid and mesenchymal CTCs were found in about 53%, 83% and 57% patients, respectively. The proportion of hybrid and mesenchymal CTCs was associated with ages, BCLC stages, metastasis and AFP levels. Besides, recurrent HCC patients presented higher CTCs count and increased hybrid and mesenchymal CTCs.
CONCLUSIONS:
CTCs count and EMT classification are correlated with clinical stages and metastasis of HCC, suggesting that they may be potential markers for the early diagnosis of HCC metastasis and progression.
Keywords
Introduction
Cancer-related mortality of hepatocellular carcinoma (HCC) ranks second in the world [1] and third in China [2]. Metastasis and recurrence account for most of the lethality, thus posing big challenges for HCC therapies such as hepatectomy, interventional therapy and liver transplantation. Therefore, early detection of metastasis and prevention of recurrence are important for improving the clinical efficacy and patient prognosis. The diagnosis of cancer metastasis mainly relies on imaging examinations including X-ray, Ultrasound, Computed Tomography and Magnetic Resonance Imaging. However, they are not satisfactory in discovering small metastases and micrometastases due to the technological limitations. And there is no reliable metastasis or relapse-specific serum biomarker up to now. Thus it is urgent to find effectual methods for the early diagnosis of HCC metastasis and monitoring of recurrence.
The detection of circulating tumor cells (CTCs) opens new horizons to address this problem. CTCs are tumor cells that released into the peripheral blood from the primary tumors or metastases. Since hematogenous spread is the dominant route of HCC dissemination [3], CTCs could be an active origin of metastasis and recurrence in HCC patients. Previous studies have shown that the presence of CTCs may indicate metastases or early recurrence of HCC in certain conditions [4, 5]. And a higher CTCs number is demonstrated associated with poor prognosis, higher recurrence risks, lower disease-free survival and overall survival after surgery [6, 7, 8, 9]. In addition, as a new pattern of liquid biopsy, CTCs test exhibits outstanding performance compared with traditional tissue biopsy, including easy sample collection, minimal invasiveness and achievable multi-monitoring. These advantages equip CTCs test to be a promising biomarker for HCC diagnosis and prognosis.
Lots of methods have been developed for CTCs isolation and detection [10], basing on physical properties [11, 12] and immunological principles [13, 14]. However, most immunological based methods target the epithelial markers to capture CTCs, such as epithelial cell adhesion molecule (EpCAM) or cytokeratins (CKs). This may result in the bias and inaccuracy of CTCs detection because of epithelial-mesenchymal transition (EMT) [15]. EMT is a vital process of metastasis because it enables epithelial cells to obtain enhanced migrating and invasive potential. CTCs may go through a dynamic process of EMT and MET (mesenchymal-epithelial transition) during their intravasation, dissemination and extravasation. Thus some CTCs may lose epithelial characteristics with decreased or deleted expression of epithelial markers and acquire a mesenchymal phenotype. Expressions of mesenchymal markers have been proved in CTCs from patients with lung, breast and prostate cancer [16, 17]. Therefore, combining the epithelial and mesenchymal markers can improve the sensitivity and accuracy of CTCs detection.
Canpatrol
Here we explored the application of the Canpatrol
Subjects and methods
Patient information
A total of 195 HCC patients from Nanfang Hospital, Southern Medical University (Guangzhou, China) during December 1, 2014 to December 31, 2016 were investigated in this retrospective study. Patients were diagnosed with HCC by histopathological examination, including 99 newly diagnosed patients and 96 post-treatment patients.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards. All patients provided written informed consent and the research protocol was approved by the Ethics Committee of Nanfang Hospital (Guangzhou, China).
CTCs test and clinical data
We analyzed the data of CTCs tests and other clinical pathological characteristics of the 195 HCC patients from the Laboratory Information System (LIS) and Jiahe Electronic Medical Record System. CTCs test was conducted by a newly imported Canpatrol
Statistical analysis
Statistical analyses were performed using SPSS 13.0 (SPSS Inc., Chicago, IL, USA) and GraphPad Prism 5.0 (GraphPad Software, San Diego, CA, USA). Data were presented as median (Range) or median (InterQuartile Range, IQR) for discontinuous variables, and categorical variables were expressed as numbers and percentages. Correlations between CTCs test and clinical characteristics were evaluated using Chi-square test. Mann – Whitney U test was used to compare the differences between the groups. Receiver operating characteristic (ROC) curves and the area under the ROC curves (AUC) were applied to analyze the efficacy of assisted diagnosis of metastasis. All tests were two tailed, and we considered a
Results
Patient characteristics and CTCs test
The clinical characteristics of the 195 HCC patients were shown in Table 1. We investigated 163 male and 32 female patients with median age of 50 years (Range 25–85 years). Among them, 123 patients had cirrhosis and 146 patients presented positive results of HbsAg. The distributions and percentages of other pathological parameters (tumor size, BCLC stage and metastasis) and serum tumor markers (AFP and CEA) were also described in Table 1.
Clinical characteristics and CTCs test of 195 HCC patients
Clinical characteristics and CTCs test of 195 HCC patients
Correlations between CTCs detection and clinical pathological characteristics in pre-treated HCC patients
CTCs EMT types were classified by fluorescence-labeled probes targeting epithelial and mesenchymal markers. (a) Epithelial type CTCs (E type). (b) Hybrid type CTCs (H type). (c) Mesenchymal type CTCs (M type). (d) Leukocyte as negative control.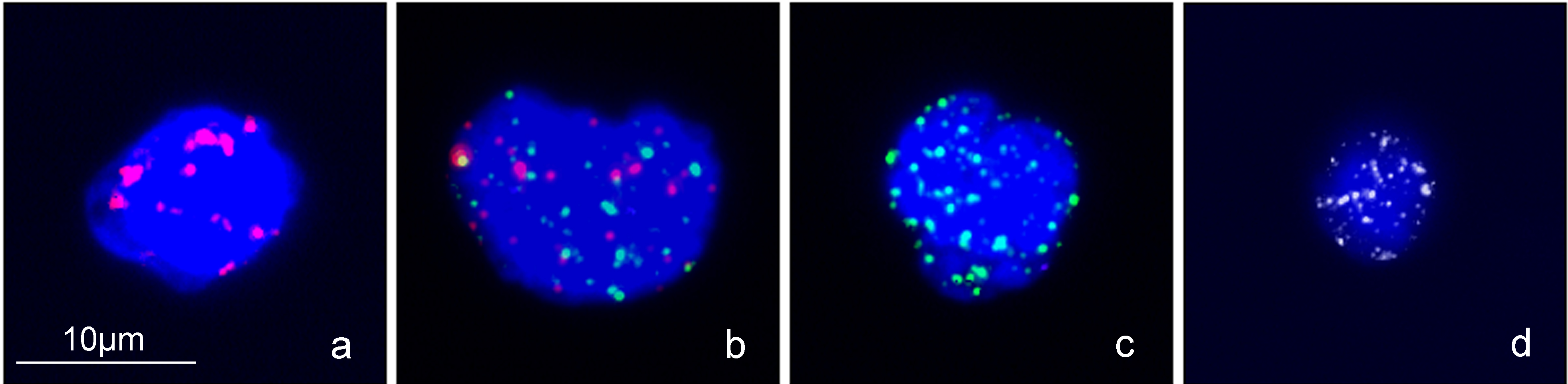
Correlations between CTCs count and clinical pathological characteristics in pre-treated HCC patients. (A) Correlation between CTCs count and BCLC stage. (B) Correlation between CTCs count and metastatic status. (C) Correlation between CTCs count and serum levels of AFP. (D) ROC curve for CTCs count to discriminate metastatic HCC patients from non-metastatic patients.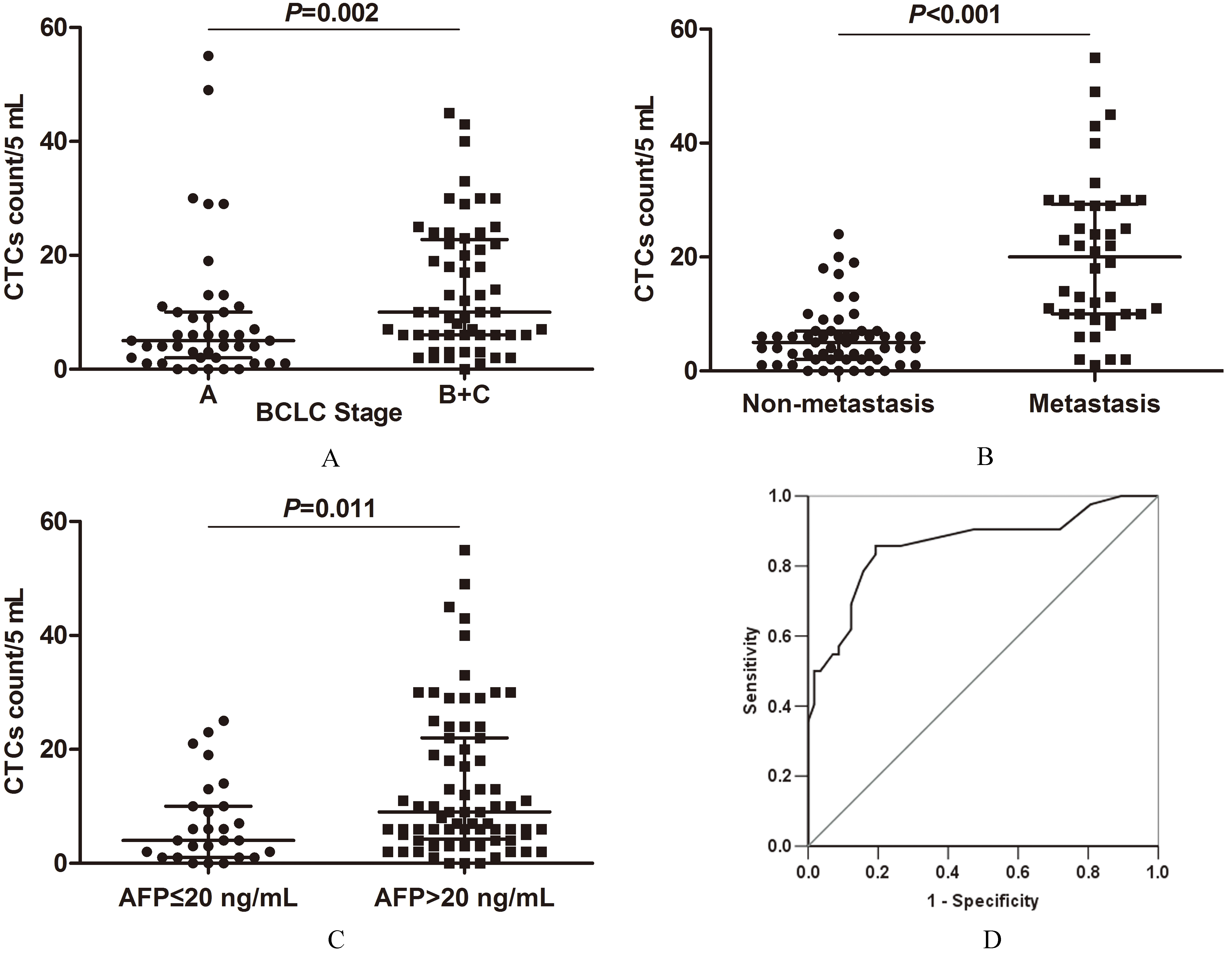
CTCs were detected (
Among the 195 HCC patients, 99 patients were newly diagnosed and blood samples for CTCs test were collected before any surgical or radiochemical therapy. We evaluated the CTCs test results in these pre-treated patients, and CTCs were positive (
CTCs count was closely associated with BCLC stage (
The median CTCs number in patients with BCLC-B/C stage HCC was 10 cells/5 mL (IQR 6–22.75), higher than that in patients with BCLC-A stage HCC (5 cells/5 mL, IQR 2–10) (
CTCs EMT phenotypes correlate with BCLC stages and metastasis of pre-treated HCC patients
Correlations between CTCs EMT phenotypes and clinical pathological characteristics in CTCs positive pre-treated HCC patients. (A) Correlation between CTCs classification and age. (B) Correlation between CTCs classification and BCLC stage. (C) Correlation between CTCs classification and metastatic status. (D) Correlation between CTCs classification and serum AFP levels.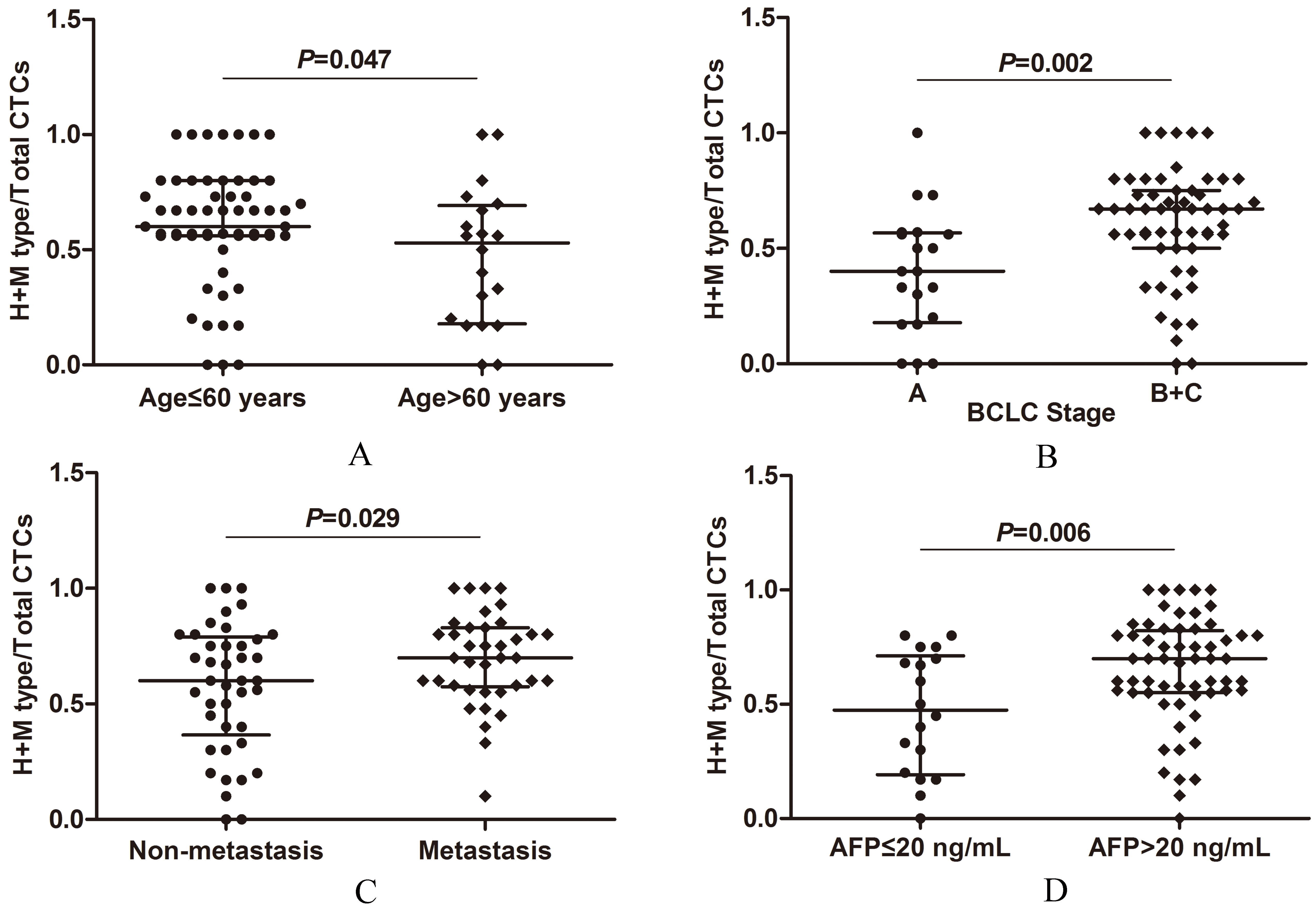
CTCs counts and EMT classification results in 16 HCC patients experienced recurrence. (A) CTCs numbers of different EMT types. (B) Percentages of different EMT types of CTCs. (C) Proportions of CTCs expressing mesenchymal markers (H 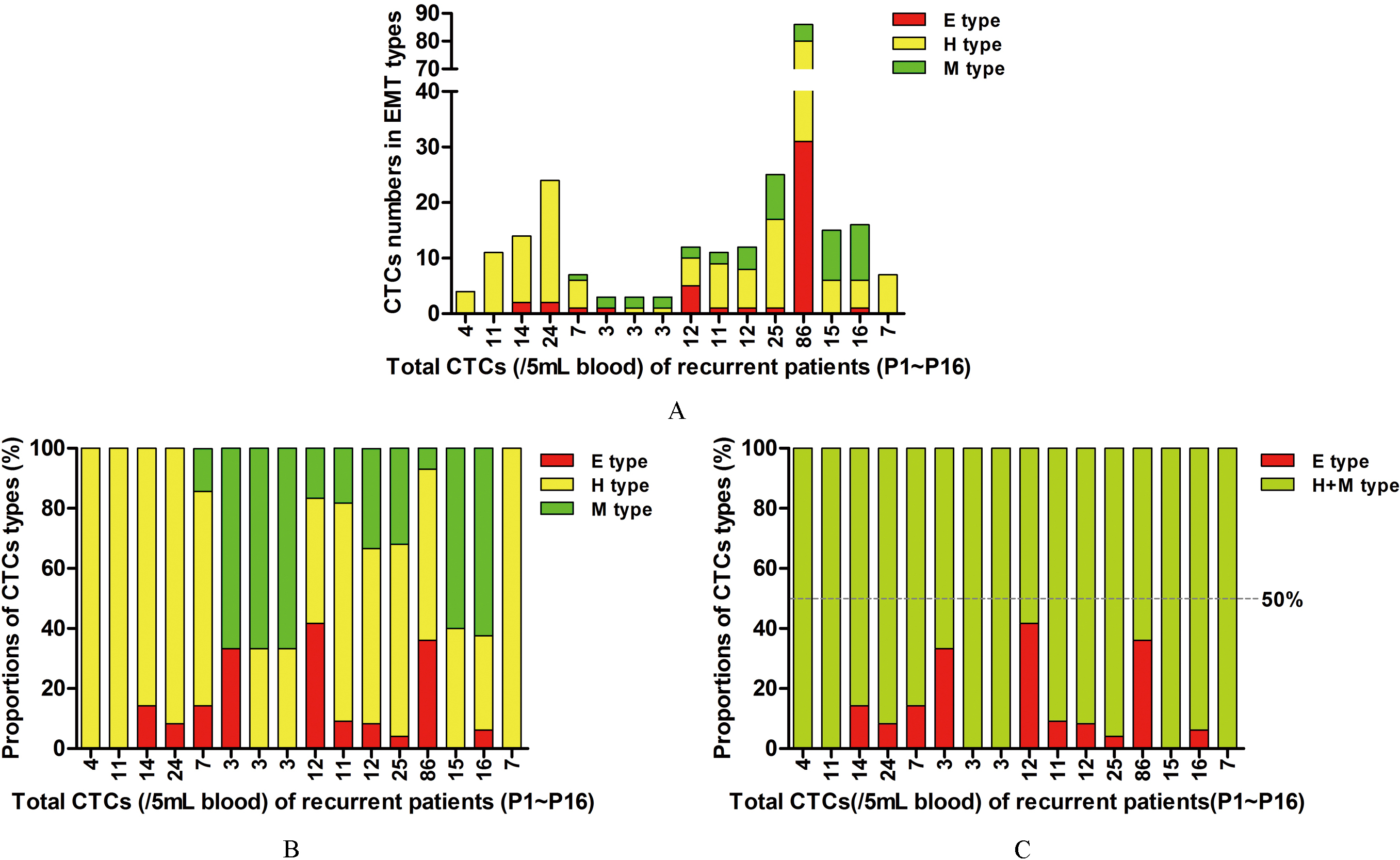
Subsequently, we analyzed the EMT phenotypes of isolated CTCs in the 79 HCC patients with positive CTCs results (
CTCs EMT phenotypes were closely associated with age (
In addition, we compared the proportions of H
In the 16 patients with recurrent HCC, CTCs numbers were
Discussion
EMT is dynamic and reversible, thus the CTCs may consist of heterogeneous subgroups expressing epithelial markers, mesenchymal markers, or simultaneously expressing two types of markers. Studies have proved the hybrid and mesenchymal phenotype of CTCs by the subsequent identifications. Among CTCs isolated by cell size-based methods, the proportion of Vimentin
CTCs detection results in 16 hepatocellular carcinoma patients experienced recurrence
The CanPatrol
CTCs are regarded as potential seeds of the metastatic dissemination of malignant tumors [23]. Some studies have demonstrated the clinical relevance of CTCs in patients with HCC and other cancers [24, 25, 26]. CTCs count could be a biomarker in evaluating the response to different therapies and predicting recurrence of HCC [5, 27, 28]. In line with the previous reports, we found that CTCs count was closely associated with BCLC stage, metastasis and serum AFP levels in pre-treated HCC patients. The higher positive rates and increased CTCs numbers were correlated with advanced BCLC stages, higher metastatic tendency and elevated serum levels of AFP. There was no significant correlation between CTCs numbers and other clinical characteristics, including gender, age, cirrhosis, HbsAg, tumor size and serum CEA levels. When using the CTCs count to discriminate metastatic HCC patients with non-metastatic patients, the AUC of the ROC curves was 0.861 (95% CI: 0.782–0.940), and the sensitivity and specificity were 85.7% and 80.7%. The metastasis might be more likely to have occurred if the CTCs count was over 7.5 cells/5 mL. These results demonstrated total CTCs number could be an assistant diagnostic marker for cancer metastasis in HCC patients, thus making up for the shortcomings of the image examinations.
CTCs expressing mesenchymal markers were thought to have stronger invasive and metastatic ability than epithelial CTCs. Li et al.’s study proved that CTCs co-expressed Twist and Vimentin was highly correlated with portal vein tumor thrombus, and the detection of both Twist and Vimentin in CTCs could predict HCC metastasis more accurately [29]. Min et al. [30] also reported that the expression of Snail in CTCs was associated with extra-hepatic metastasis of HCC. Accordingly, we calculated the index on proportions of H
The classification of BCLC stage is based on the evolutionary course of tumor progression and liver disease, including tumor size, number, and assessment of liver function scores [31]. It is commonly used in clinical practice because it helps to predict life expectancy and choose specific treatment mode of HCC patients. The co-expression of Twist and Vimentin in CTCs was proved highly correlated with the portal vein tumor thrombus, TNM classification and tumor size of HCC [29]. In this study, both the CTCs count and EMT phenotypes were closely associated with advanced stages of HCC. Remarkably, we noticed that some of the patients in the early stage of BCLC (stage A) had relatively higher proportions of H and M type CTCs. Large tumor size was reported to be correlated with prognosis and recurrent metastasis of HCC. Studies proved that giant tumor size over 10 cm was an independent risk factor of elevated AFP level [32], ascites after hepatectomy [33] and 5-year survival [34]. In our study, relatively higher CTCs count along with higher H and M type CTCs proportions in some patients with tumor size smaller than 5 cm was also observed. These results suggest that there may be a tendency for HCC to metastasize at early stage when the tumor volume is small. Therefore, the combination of CTCs count and EMT phenotypes may complement the analysis of patients with metastatic status and BCLC staging.
Serum AFP is the most widely used serological marker for the diagnosis and management of HCC currently. Although there is heterogeneity in the levels of AFP in HCC patients, the clinicians consider the primary tumor size and serum AFP levels as important risk factors of recurrence. Wong et al. [35]reported that in conjunction with the serum AFP test, sequential quantification of serum AFP mRNA could predict clinical metastasis/recurrence in 56% of HCC patients during a 4-year follow-up. Schulze and the colleagues showed evidence for a significant correlation between the detection of epithelial CTCs and AFP values in HCC patients [7]. Li’s group isolated the asialoglycoprotein receptors (ASGPRs) – positive CTCs from the blood of HCC patients, but the immunofluorescence staining presented that the expression of Vimentin or Twist in CTCs had no relationship with serum AFP levels [29]. Our results indicated that both the higher CTCs counts and increased H
Age is a noticeable characteristic of HCC in clinics, because patients with HCC are known to consist of heterogeneous populations [36]. Compared with middle-aged group (50 to 79 years) and elderly group (
In conclusion, we retrospectively analyzed the results of CTCs test detected by the CanPatrol
Conflict of interest
The authors declare that they have no conflict of interest.
Footnotes
Acknowledgments
This work was supported by the Natural Science Foundation of Guangdong Province, China (Grant No. 2015A030313247) and the Science and Technology Planning Project of Guangdong Province, China (Grant No. 2016A010105006).
Supplementary materials
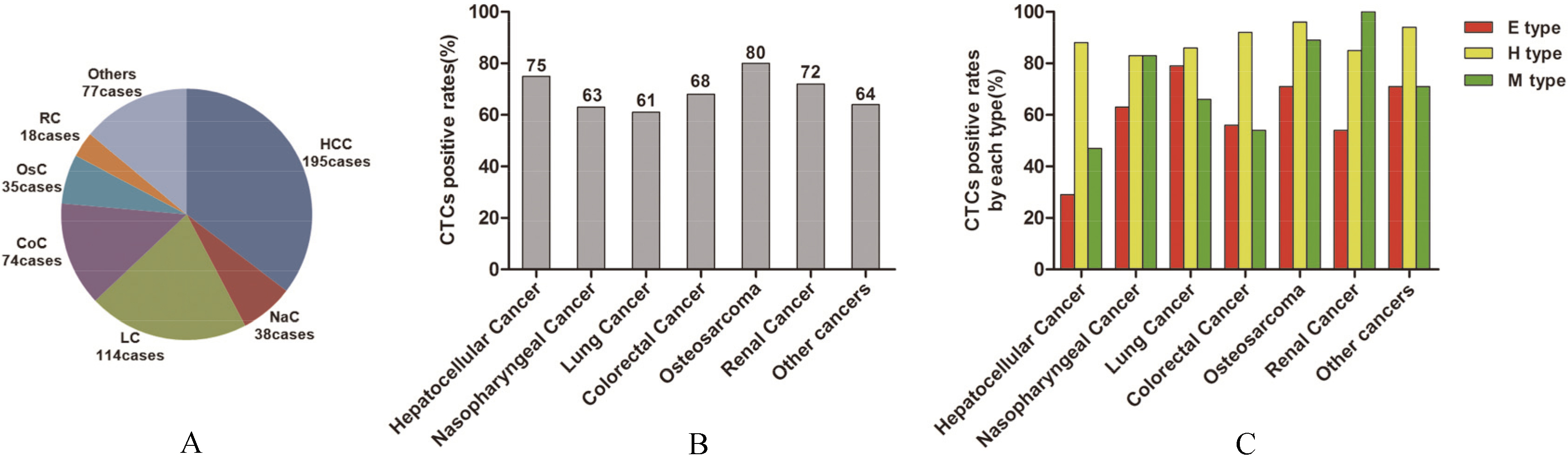
CTCs count and EMT classification in 551 patients with common cancers
Cancer type
CTCs+/Total
Median (range)/5 mL
CTCs+ patients (
(rates)
Total patients
CTCs+ patients
E type CTCs+
H type CTCs+
M type CTCs+
Hepatocellular cancer
146/195
(75%)
6
(0–86)
10
(3–86)
42/146
(29%)
128/146
(88%)
69/146
(47%)
Nasopharyngeal cancer
24/38
(63%)
4
(0–34)
7
(3–34)
15/24
(63%)
20/24
(83%)
20/24
(83%)
Lung cancer
70/114
(61%)
7.5
(0–67)
7.5
(3–67)
55/70
(79%)
60/70
(86%)
46/70
(66%)
Colorectal cancer
50/74
(68%)
4
(0–58)
6
(3–58)
28/50
(56%)
46/50
(92%)
27/50
(54%)
Osteosarcoma
28/35
(80%)
6
(0–35)
10
(3–35)
20/28
(71%)
27/28
(96%)
25/28
(89%)
Renal cancer
13/18
(72%)
4
(0–54)
7
(3–54)
7/13
(54%)
11/13
(85%)
13/13
(100%)
Other cancers
49/77
(64%)
4
(0–51)
5
(3–51)
35/49
(71%)
46/49
(94%)
35/49
(71%)
Total
380/551
(69%)
–
–
202/380
(53%)
338/380
(89%)
235/380
(62%)
CTCs count and EMT classification in 551 patients with common cancers. (A) The distribution of patients with different cancers. (B) CTCs positive rates in different cancers. (C) EMT phenotypes of CTCs in different cancers.