Abstract
OBJECTIVE:
Emerging evidence has suggested that circulating microRNAs (miRNAs) in body fluids have novel diagnostic and prognostic significance for patients with malignant diseases. The lack of useful biomarkers is a crucial problem of osteosarcoma (OS); Previous study has reported that miR-542-3p was significantly upregulated in osteosarcoma tissues and miR-542-3p may be as an oncogene in osteosarcoma pathogenesis. In our study, we investigated the circulating miR-542-3p and its clinical relevance in osteosarcoma.
METHODS:
Serum MiR-542-3p levels were determined by quantitative real-time PCR assays (qRT-PCR) in 76 patients with OS and 76 healthy volunteers. Patient survival analyses were performed by Kaplan-Meier analyses and Cox regression models. All statistical tests were two-sided.
RESULTS:
It was observed that the serum levels of miR-542-3p was significantly higher in patients with OS compared with the control groups (
CONCLUSIONS:
The results showed that serum miR-542-3p levels could serve as a non-invasive blood biomarker for tumor monitoring and prognostic prediction in osteosarcoma patients.
Introduction
Osteosarcoma (OS) generally localizes to the metaphysis of long bones and is the most common bone tumor in children and adolescents, comprising 2.4% of all malignancies in pediatric patients [1, 2]. It is also a major cause of malignancy-related death in adolescence, due to its rapid progression and high metastatic potential [2, 3]. At present, the detection of the primary tumor or tumor relapse has generally relied on imaging methods such as X-ray, computed tomography (CT), positron emission tomography (PET)-CT, magnetic resonance imaging (MRI), and scintigraphy. The main treatment strategies for OS consist of tumor excision, chemotherapy, and radiotherapy, and the five-year survival rate of OS patients is approximately 60%–70% [4, 5]. However, a large proportion of OS patients respond poorly to combined therapy and their risk of local relapse and distant metastasis is relatively high [6]. The unfavorable prognosis of OS patients is partially due to current poor strategies for early diagnosis and evaluation of prognosis [7]. Thus, development of methodologies for real-time monitoring of drug response and early detection of recurrence or metastasis will further improve patient prognosis. Currently, less complex monitoring using patient blood has not been developed. Alkaline phosphatase (ALP), a known serum-based tumor marker of osteosarcoma, sometimes provides false positives since ALP is generally elevated in children and affected by organ damage. Thus, the development of highly sensitive, specific, and minimally invasive biomarkers that can be used to detect and monitor tumor burden of tumors is the most important challenge for osteosarcoma management.
MicroRNAs are a group of single-stranded, endogenous and non-coding RNAs that are generally 18–24 nucleotides in length [8]. These molecules can modulate gene expression by binding to the 3’-untranslated regions (3’-UTR) of mRNAs [9, 10] and they participate in various biological and pathological processes, including development, apoptosis, cell differentiation, cell proliferation, and inflammation [11, 12, 13]. Upsregulation of microRNAs, including miR-30a-5p, miR-33a, miR-9 and miR-218 has previously been reported in OS [14, 15, 16, 17].
Tumor cells have recently been demonstrated to secrete miRNAs into the circulation [18]. Despite the presence of RNase activity in human blood, data have demonstrated that serum miRNAs remain stable under protection by exosomes or argonaute 2 [19, 20]. Analysis of circulating miRNA levels in patient blood presents a novel approach for diagnostic cancer screening or monitoring.
Recently, a group has reported the association of miR-542-3p downexpression in favorable histology, MYCN nonamplified neuroblastoma samples [21, 22]. Li et al. has only found that miR-542-3p was overexpressed in OS, providing assistance in understanding the development of osteosarcoma and aid in the development of strategies for the diagnosis and treatment of osteosarcoma [23]. However, they did not study the further relation between miR-542-3p expression and clinical significance in OS.
In this study, we detected miR-542-3p level in osteosarcoma patient’s serum and evaluated whether serum miR-542-3p could monitor and assess prognosis in patients with OS.
Patients and methods
Patients
The study was approved by the Ethical Committee of people’s Hospital of Linyi, and informed consent was obtained from all enrolled participants. From January 2008 to December 2012, primary OS patients administered in our department were screened for eligibility for inclusion in the study. Patients meeting the following criteria were excluded: metastatic OS, previous malignant tumor in other organs or systems, history of cardiovascular or cerebral vascular disease, poor general physical condition, previous chemotherapy, radiotherapy or surgery, and unwillingness to participate. All enrolled patients received neoadjuvant chemotherapy, tumor resection, and adjuvant therapy according to NCCN (National Comprehensive Cancer Network) clinical practice guidelines on OS, and patients were followed-up via outpatient visit, telephone, and letter. Patients were asked to undergo physical examination and radiography every 3 months, and computed tomography or magnetic resonance imaging when necessary. Progression-free survival was calculated from the date of surgery to recurrence or metastasis, and overall survival was calculated from the date of surgery to that of death. An age- and gender-matched healthy control group was also recruited.
Cox proportional regression analysis of the association between miR-542-3p expression levels in plasma and progression-free survival of osteosarcoma patients
Cox proportional regression analysis of the association between miR-542-3p expression levels in plasma and progression-free survival of osteosarcoma patients
Peripheral venous blood samples (5 ml) were collected into EDTA anticoagulation tubes, before any treatment was administered, and centrifuged at 4,000
A Rotor gene 6000 real-time PCR machine (Qiagen, Germany) was used to conduct qPCR, and 20
Statistical analyses
MedCalc 13.0 (MedCalc Software bvba, Ost-end, Belgium) and SPSS 16.0 (SPSS, Chicago, IL, USA) were used for statistical analyses. One way analysis of variance and Student’s t tests were used for evaluation of differences between groups, as appropriate. Receiver operating characteristic (ROC) curve analysis was conducted to evaluate the efficacy of miR-542-3p for the differential diagnosis of OS and healthy controls. The association between miR-542-3p expression and survival was assessed by Logrank test and Cox proportional hazard regression analysis. Age, gender, tumor location, tumor stage, histologic grade, and miR-542-3p expression level were included in the multivariate analysis. The mean value of miR-542-3p expression in the OS group was set as the cutoff point for differentiation of patients with high or low miR-542-3p expression.
Cox proportional regression analysis of the association between miR-542-3p expression in plasma and overall survival of osteosarcoma patients
Cox proportional regression analysis of the association between miR-542-3p expression in plasma and overall survival of osteosarcoma patients
Kaplan-Meier analysis of the association between serum miR-542-3p level and (A) Progression-free survival, and (B) Overall survival of osteosarcoma patients. Osteosarcoma patients with elevated serum miR-542-3p level have shorter progression-free and overall survival.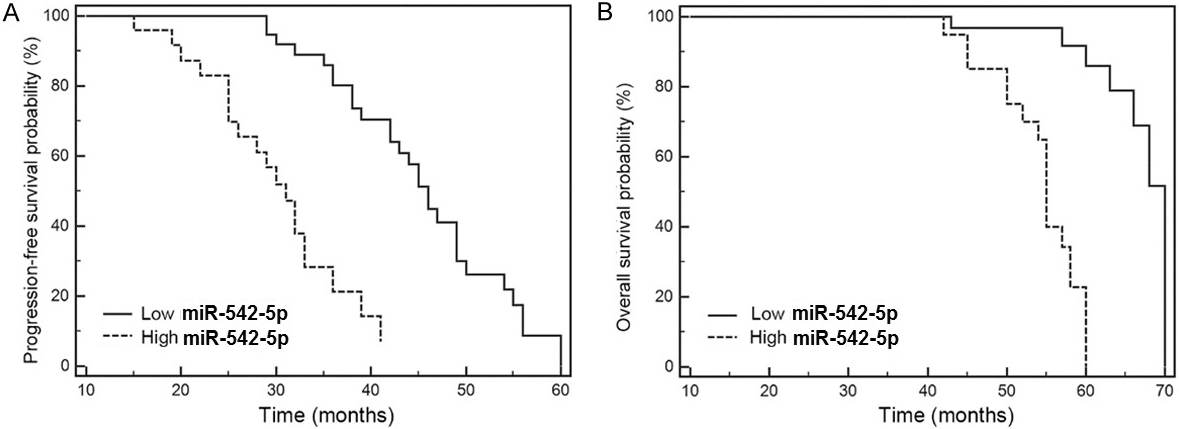
A total of 76 OS patients and 76 healthy volunteers were enrolled in the study. The expression level of miR-542-3p in the OS groups (0.87
In survival analysis, patients with miR-542-3p levels
Discussion
The present study found that miR-542-3p was up-regulated in the plasma of OS patients, and its overexpression was positively associated with advanced tumor stage. Moreover, miR-542-3p could identify OS patients from healthy volunteers with high specificity (77.8%) and 93.6% sensitivity. Further, survival analysis indicated that elevated miR-542-3p was a risk factor for both progression-free and overall survival.
Previous studies have reported that miR-542-3p are down-regulated and act as tumor suppressors in neuroblastoma [24] and esophageal cancer [25]. However, miR-542-3p was upregulated in OS cells, and overexpression of miR-542–3p can enhance the cell proliferation and migration ability of U2OS cells [20]. Therefore, the function of miR-542-3p in malignant tumors may depend on the cellular context.
In our study, we found that plasma miR-542-3p expression was increased in patients with advanced tumor stage. Further, ROC analysis indicated that miR-542-3p could identify OS with high specificity; however, the sensitivity of this test was relatively low, which is difficult to explain, hence further work is required to explore this issue, since such a low sensitivity value could limit the value of miR-542-3p as a biomarker for OS screening. Additionally, the value of miR-542-3p in prognosis assessment has previously been explored in studies of various other tumors. Our study provides the first report that OS patients with elevated plasma miR-542-3p expression have shorter progression-free and overall survival, indicating that screening of miR-542-3p levels could allow identification of OS patients with a high risk of progression and recurrence, facilitating optimization of treatment strategies. How miR-542-3p affects the development and progression of OS remains a matter of debate.
Although the results of the present study are encouraging, several limitations should be considered. First, the sample size of the study was small, and larger scale investigations are required to verify the results. Second, only OS patients without metastasis were enrolled, as the purpose of the study was to explore the efficacy of miR-542-3p in diagnostic screening. The present study showed that miR-542-3p had low sensitivity as a diagnostic biomarker; however, it performed well with tumor stage. Whether miR-542-3p will function adequately in prognostic assessment of OS with metastasis remains to be determined. Third, although patients were treated according to the NCCN practice guidelines for OS, there were differences in the detailed treatment strategies used for individual patients. This factor was not taken into account in our survival analyses, and may reduce the credibility of the study to a limited extent. Fourth, all participants in this study were from a single center, hence whether they are representative of the whole population remains to be determined.
In conclusion, miR-542-3p is up-regulated in OS and acts as a risk factor for progression-free and overall survival. Serum miR-542-3p levels could serve as a non-invasive blood biomarker for tumor monitoring and prognostic prediction in osteosarcoma patients.
