Abstract
BACKGROUND:
Renal cell carcinoma (RCC) is one of the most malignant genitourinary diseases worldwide. Long noncoding RNAs (lncRNAs) are a class of noncoding RNAs in the human genome that are involved in RCC initiation and progression.
OBJECTIVE:
To investigate the expression of PVT1 in ccRCC and evaluate its correlation with clinicopathologic characteristics and patients’ survival.
METHODS:
Quantitative real-time PCR was performed to examine PVT1 expression in 129 ccRCC tissue samples and matched adjacent normal tissue samples. The relationship of PVT1 expression with clinicopathologic characteristics and clinical outcome was evaluated.
RESULTS:
We identified the lncRNA PVT1, which was upregulated in clear cell renal cell carcinoma (ccRCC) tissues when compared with corresponding controls. Furthermore, PVT1 expression was positively associated with gender, tumor size, pT stage, TNM stage, and Fuhrman grade. Kaplan-Meier survival analysis showed that patients with high PVT1 expression had shorter disease-free survival (DFS) and overall-survival (OS) than those with low PVT1 expression, and multivariate analysis identified PVT1 as an independent prognostic factor in ccRCC.
CONCLUSIONS:
PVT1 may be an oncogene as well as may promote metastasis in ccRCC and could serve as a potential biomarker to predict the prognosis of ccRCC patients.
Introduction
Renal cell carcinoma (RCC) accounts for 3–5% of all malignant diseases in adults and is the tenth most common cancer in women and the seventh most common in men [1]. Surgical resection for clinically localized RCC remains a potentially curative intervention, but the recurrence rates after nephrectomy are 20–40% [2]. For patients with recurrent or metastatic RCC, there are limited treatment options and low response rates to systemic therapy [3]. ccRCC is the most common type of kidney tumor, accounting for 70–80% of all cases [4]. Although numerous genes involved in ccRCC tumorigenesis and metastasis have been identified, the molecular mechanisms underlying these processes are not completely understood [5].
The human genome contains only 20,000 protein-coding genes, representing
The lncRNA plasmacytoma variant translocation 1 (PVT1), which resides in chromosome region 8q24.21, plays an oncogenic role in multiple cancer types and is associated with clinical characteristics such as recurrence and survival in various cancers, including breast cancer, ovarian cancer, colorectal cancer, hepatocellular carcinoma, gastric cancer, esophageal cancer, cervical cancer, lung cancer, prostate cancer, leukemia, and osteosarcoma [18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30]. A pan-cancer analysis suggested that the transcripts of the PVT1 locus were overexpressed in kidney renal cell carcinoma (KIRC) and were associated with worse survival rates and clinical outcomes of KIRC [31]. This suggests that PVT1 could play a crucial role in ccRCC and merits further research. In this study, we investigated the expression levels of PVT1 in ccRCC tissues and evaluated its potential as a prognostic biomarker in ccRCC patients.
Materials and methods
Patients and specimens
CcRCC tissue samples and matched adjacent normal tissue samples were collected from 129 patients who underwent nephrectomies at the Urology Department of Chinese People’s Liberation Army (PLA) General Hospital (Beijing, China) between January 2010 and December 2010. All samples were immediately preserved in liquid nitrogen after resection and then stored at
Total RNA extraction and quantitative real-time PCR
Total RNA was extracted from tissue samples with TRIzol Reagent (ComWin Biotech, Beijing, China) according to the manufacturer’s protocol. The total extracted RNA was then converted into cDNA using the TransScript First-Strand cDNA Synthesis SuperMix (TransGen Biotech, Beijing, China). Real-time PCR was performed with the TransStart Green qPCR SuperMix (TransGen Biotech) using the Applied Biosystems 7500 Detection System (Applied Biosystems, Foster City, CA) and the primers as follows: PVT1 forward 5’-TCTTGGTGCTCTGTGTTC-3’ and reverse 5’-CCGTTATTCTGTCCTTCTCA-3’; PPIA forward 5’-ATGGTCAACCCCACCGTGT-3’ and reverse 5’-TCTGCTGTCTTTGGGACCTTGTC-3’. The relative expression levels, which were normalized to the expression levels of the housekeeping gene PPIA, were calculated by the 2
Statistical analysis
Statistical analysis was performed with SPSS 22.0 software (SPSS Inc., Chicago, IL, USA). Statistical differences between two groups were analyzed by Student’s t-test. The Chi-square test was performed to determine the association between ccRCC clinicopathological characteristics and PVT1 expression. The survival curves were plotted according to the Kaplan-Meier method, and differences were compared with the log-rank test. Cox’s proportional hazards model was used to identify the independent prognostic factors. Results were represented as mean
Expression of PVT1 in ccRCC specimens and matched adjacent non-tumor tissues. (A) PVT1 was highly expressed in ccRCC specimens compared with matched adjacent non-tumor tissues. (B–F) The expression of PVT1 in each category grouped by localized or metastatic, tumor size, pT stage, TNM stage, and Fuhrman grade of ccRCC, respectively. 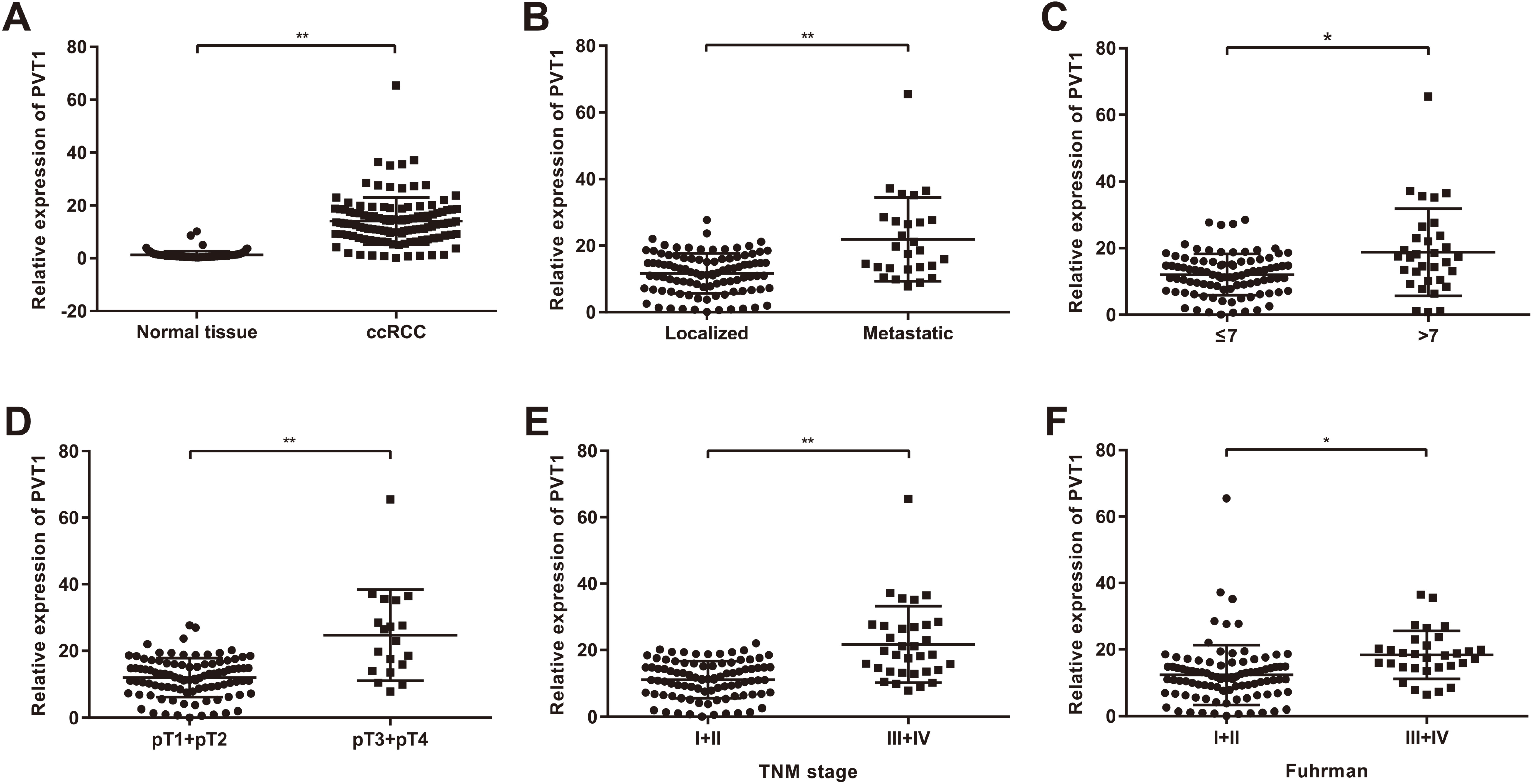
PVT1 is highly expressed in ccRCC tissues
We examined PVT1 expression in pairs of ccRCC and matched adjacent noncancerous tissues. The expression of PVT1 was significantly higher in cancer tissues than in normal tissues (
Relationship between PVT1 expression and clinicopathological features in patients with ccRCC
Relationship between PVT1 expression and clinicopathological features in patients with ccRCC
To evaluate the clinical relevance of PVT1 expression in ccRCC, we examined the correlations between PVT1 expression and clinicopathological characteristics of ccRCC patients (Table 1). Using the mean expression of PVT1 in ccRCC tissues as a cut-off value, the ccRCC patients were categorized into low- or high-expression subgroups. Of the 129 ccRCC patients included in the study, 71 were classified as having low PVT1 expression and 58 were as having high PVT1 expression. PVT1 expression in ccRCC was significantly correlated with gender (
Role of PVT1 in ccRCC patient survival
We then evaluated the prognostic value of PVT1 expression in ccRCC patients. The median follow-up time was 36 months. ccRCC recurred in 27 patients (20.93%), and 16 patients (12.40%) died (all as a result of ccRCC). Disease-free survival (DFS) was significantly shorter in ccRCC patients with high PVT1 expression than those with low PVT1 expression (
Univariate and Multivariate Cox proportional hazard analyses for disease-free survival and overall survival in patients with ccRCC
Univariate and Multivariate Cox proportional hazard analyses for disease-free survival and overall survival in patients with ccRCC
Kaplan-Meier survival curves of patients with ccRCC categorized by PVT1 expression status. (A) Disease-free survival. (B) Overall survival.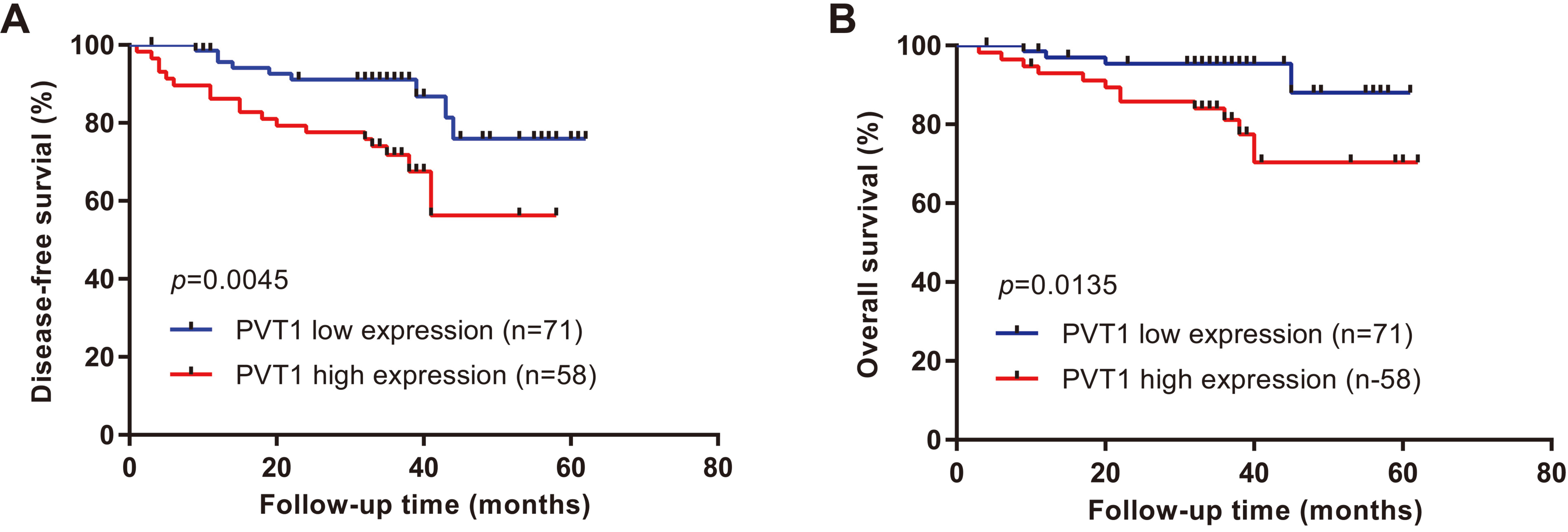
We next carried out univariate analysis of prognostic factors for DFS and OS with Cox’s proportional hazards regression model (Table 2). Larger tumor size (
Disease recurrence takes place in one-third of patients after a partial or radical nephrectomy for RCC, and the median survival for metastatic patients is only about 13 months [33]. Although multiple potential prognostic biomarkers for RCC, such as TNM stage, nuclear grade, and genetic and molecular markers, have been identified in the past two decades to help monitor or predict further progression of RCC and the survival rates of patients, many of them have shown contradictory results and none of them is 100% accurate [34, 35]. In recent years, lncRNAs have attracted mounting attention from researchers, as they have found that lncRNAs are expressed in a more tissue-specific manner than protein-coding genes, and the misexpression of lncRNAs could be profiled to aid in determining cancer prognosis. For example, the HOTAIR expression level is a powerful and independent predictor of eventual metastasis and death in breast cancer by multivariate analysis [15]. SChLAP1 expression is an independent predictor of prostate cancer aggressiveness and predicts biochemical recurrence, clinical progression, and prostate cancer-specific mortality [16, 36]. High MALAT1 expression is highly predictive of a poor prognosis in early-stage non-small cell lung cancer [14].
The PVT1 locus, first discovered in 1984, is located 57 kb downstream of the MYC locus and encodes both long noncoding RNAs and a cluster of six microRNAs including miR-1204, miR-1205, miR-1206, miR-1207-5p, miR1207-3p, and miR-1208 [37, 38]. Previous studies have reported that this locus is a site of translocation in mouse plasmacytomas or human Burkitt’s lymphoma and a site of retroviral insertion in mouse lymphomas [39, 40, 41]. When amplified and overexpressed in ovarian and breast cancers, the PVT1 locus acted independently of the MYC locus to increase proliferation and inhibit apoptosis and had a negative influence on survival [18]. In human pancreatic cancer, functional inactivation of the PVT1 gene led to increased sensitivity to gemcitabine [42]. Following the dramatic advance in RNA-seq technology, it has been gradually realized that the human genome is pervasively transcribed and more and more lncRNAs have been identified [43]. The lncRNA PVT1 is transcribed from the locus mentioned above. In gastric cancer, PVT1 promoted proliferation and invasion by directly binding to the FOXM1 protein, and the high expression of PVT1 predicted a poor prognosis [44]. In hepatocellular carcinoma (HCC), PVT1 promoted cell proliferation, cell cycling, and the acquisition of stem cell-like properties in HCC cells by stabilizing the NOP2 protein, High expression levels of PVT1 were correlated with tumor size and tumor stage and predicted markedly reduced recurrence-free survival and overall survival [20]. In addition, another study also showed that the high expression of PVT1 was correlated with a higher TNM stage and larger tumor size, as well as a shorter OS, and demonstrated that PVT1 affected cell proliferation and apoptosis by PVT1/EZH2/LATS2 interactions in non-small cell lung cancer [24]. Consistent with these results, our clinical data showed that PVT1 was upregulated in ccRCC tissues. High expression of PVT1 was positively associated with unfavorable DFS and OS. Our study indicates that PVT1 may be an oncogene and may promote metastasis in ccRCC and has potential as a prognostic biomarker in ccRCC patients.
Detection of gene expression using RNAs from fresh or frozen tumor samples is one of the best options to study cancer development, progression and prognosis. However, fresh or frozen tissue specimens are not readily used [45]. Archived formalin-fixed paraffin embedded (FFPE) tissues are the most widely used worldwide for tissue storage, and could be kept indefinitely with long-term clinical follow-up. Thus, FFPE represents an invaluable resource for examining RNAs such as lncRNAs [46, 47, 48, 49]. Various methods, including in situ hybridization (ISH), fluorescence in situ hybridization (FISH) and quantitative real-time PCR, have been applied to try to determine RNAs levels in this kind of specimens [50]. For example, in prostate cancer study involving 937 patients, high SChLAP1 expression was with higher risk of lethal progression, as determined using SChLAP1 ISH [51]. A report showed that they developed a novel RNA-FISH assay, which could be used to identify FGFR3-TACC3 fusion transcripts in FFPE human bladder cancer samples [52]. Another study revealed that miR-451 expression was a reliable FFPE tissue biomarker for papillary thyroid carcinoma malignancies by real-time PCR [53]. However, archived FFPE tissues still present many problems in molecular analysis, including nucleic acid degradation, nucleic acid base modifications and cross-linking and contamination with inhibitors of downstream PCR-based application [48, 54, 55]. To the best of our knowledge, there is no study about PVT1 expression in FFPE tissues by PCR or FISH. Only one study performed ISH in tissue microarrays to examine nuclear expression of PVT1 [56]. So it is worthy of further research to confirm whether it is possible to detect the expression levels of PVT1 in FFPE ccRCC samples by PCR or FISH or not.
Over the past decade, with the advancement of knowledge of underlying biology about RCC, targeted agents and/or immune check-point inhibitors were completely or ongoing developed, which revolutionised the treatment of inoperable or metastatic RCC, ending an cytokine-based immunotherapy era [57, 58, 59]. Three classes of targeted therapy have been developed including tyrosine kinase inhibitors (TKIs): sorafenib, sunitinib, pazopanib bevacizumab and axitinib; mammalian target of rapamycin (mTOR) inhibitors: everolimus and temsirolimus; check-point inhibitors: nivolumab [57]. In addition, agents with additional targets apart from VEGFR have been granted, such as cabozantinib and lenvatinib, and several other check-point inhibitors are being tested or studied in various clinical settings or clinical trials, such as CTLA-4-directed antibodies: ipilimumab, PD-1-directed antibodies: pidilizemab, and anti-PD-L1 antibodies: BMS-936559, durvalumab and atezoli- zumab [60, 61]. Meanwhile, much enthusiasm also exists for combination therapy, sequential therapy, adjuvant therapy, and neoadjuvant therapy in RCC, which still need more studies and more evidences for their efficacy and safety [57, 62, 63].
Currently, no clinically validated biomarkers are established to aid in the diagnosis, prediction, prognosis and risk stratification in RCC, although many putative biomarkers have been suggested. For example, Li et al. demonstrated that up-regulation of lncRNA NETA1 by real-time PCR in ccRCC tissues predicted worse 5-year survival rate of ccRCC patients [64]. Zhang et al. showed that RCC patients with higher histone deacetylase 6 (HDAC6) expression detected by real-time PCR, western blot or immunohistochemistry in fresh or paraffin embedded samples had relative poor survival [65]. Voss et al. identified a number of circulating biomarkers, such as AXL and VEGF-A, which was associated with progression-free survival (PFS) of treatment using everolimus and sunitinib in mRCC [66]. Wu et al. reported advanced magnetic resonance imaging (MRI) techniques (perfusion MRI and diffusion-weighted imaging) and radiomics analysis have the strength for serving as diagnostic, therapeutic, and prognostic RCC biomarkers [67]. In addition, gene mutations or methylations, neutrophils to lymphocyte ratio, single-nucleotide polymorphisms (SNPs), and tumor-associated macrophages (TAMs) have also been investigated as RCC biomarkers [68, 69]. Biofluids from patients with RCC, like urine and saliva, could be applied to development of less invasive diagnostic methods and novel disease markers as well [70, 71].
However, the present study has some limitations. First, it was conducted using a relatively small sample size. A large-scale and multi-institutional research study is required in the future. Second, although PVT1 may be an oncogene and may promote metastasis, we have not verified its effect on ccRCC in in vivo and in vitro experiments or explored the underlying mechanism affecting the progression of ccRCC.
Footnotes
Acknowledgments
This work was supported by the National High Technology Research and Development Program (“863” Program) of China (2014AA020607) and the National Nature and Science Grant of China (81402109).
