Abstract
BACKGROUND:
Pyrosequencing is recognized as a strong technique to analyze the MGMT status of glioblastoma patients. The most commonly used assay, quantifies the methylation levels of CpGs 74 to 78. A more recent CE-marked In Vitro Diagnostic Medical Device (CE-IVD) assay, Therascreen, analyzes CpGs 76–79.
METHODS:
We performed a comparison of these two assays to evaluate the potential impact of this shift in analyzed CpGs. Therascreen analysis was centrally performed for 102 glioblastoma patients, who were part of a prospective multicenter trial.
RESULTS:
A strong correlation was observed for the mean values of the 4 or 5 analyzed CpGs, with lower values recorded using the Therascreen assay, especially for values greater than 20%. When considering a classification in 3 categories (
CONCLUSION:
The results demonstrate that these assays may be used interchangeably.
Introduction
Qiagen currently sells numerous assays for quantitative measurements of MGMT (methylguanine methyltransferase) methylation status using the pyrosequencing technique. Hs_MGMT_01_PM PyroMark CpG assay (ref PM 00149702) analyzes 7 CpGs located upstream of DMR1 and DMR2 regions, where methylation has significantly been correlated with expression [1]. This assay must therefore be avoided for a clinical purpose. Two additional assays (ref 970032 and 972032) quantify the methylation levels of CpGs 74 to 78. They are respectively optimized for use with the PyroMark Q24 and Q96. These two similar assays are currently the most widely used and validated pyrosequencing assays [2]. They will be further referred to as PSQ. In 2011, a fourth assay was launched: the Therascreen MGMT Pyro Kit (Thera). This assay, in contrast to the others, is a CE-marked In Vitro Diagnostic Medical Device (CE-IVD) that meets all requirements of the EC Directive 98/79.
Several countries around the world have adopted the international standard ISO 15189, which specifies requirements for quality and competence in medical laboratories. Among the preferred procedures described in this international standard are those specified in the instructions for use of in vitro medical devices. Furthermore, as the steps of validation into the laboratory are less extensive for CE-IVD assays, laboratories tend to favor this type of assay. Thera appears to be a suitable choice for MGMT testing and some studies have reported strong analytical performances for this assay [3, 4]. However, this assay quantifies the methylation levels of CpGs 76 to 79 instead of CpGs 74 to 78. One of the strengths of PSQ for MGMT testing relies on the several independent studies that are concordant with the threshold levels that discriminate glioblastoma (GBM) patients as being good or poor responders to Temozolomide (TMZ) treatment [2]. As a heterogeneous pattern of methylation can be observed for some tumors, the shift in the CpGs analyzed between the two assays could potentially impact the result (
We have recently performed a prospective dedicated multicenter trial, which allowed us to validate the use of PSQ in a daily practice. For the present study, we analyzed 102 frozen GBM patients from this trial with the Thera assay and compared the results to those obtained previously with the standard PSQ test.
Materials and methods
Patients and samples
Samples were analyzed from patients enrolled in a prospective study dedicated to the validation of two techniques to assess MGMT status [5]. Patients were enrolled for this study between the dates of March 11, 2009 and June 29, 2011 from 8 French centers. Eligible patients had histologically confirmed de novo-glioblastoma, between the ages of 18–70 and presented with no contraindications, as dictated by the Stupp protocol. The protocol was approved by the Rennes medical ethics committee and informed consent was obtained from each patient.
DNA was extracted from 3 primary cell lines (RNS85/96 and 175), which were used as quality controls in each series of tests.
MGMT promoter methylation analysis
DNA extractions from frozen clinical samples and sodium bisulfite treatment were performed at each center according to local procedures. Samples with a histologically estimated tumor cell content below 40% were excluded from the study. The Thera test was centrally performed on any remaining bisulfite treated DNA following completion of the main portion of the project. Thera was performed using the Therascreen MGMT Pyro Kit (ref 971061, Qiagen, France) according to the manufacturer’s instructions. The average percentage of the 4 CpGs tested was considered to calculate the cut-off.
Statistical analysis
Statistical analysis was performed using R statistical software (version 2.13.0,
Results
Study population
Among the 112 samples of patients initially analyzed with PSQ [5], 102 were available for the Thera analysis. The median Progression Free Survival (PFS) for these 102 patients was 9.5 months (8.8–11.2; 95% CI) and the median Overall survival (OS) was 20.6 months (18.7–23.0; 95% CI).
Analysis of intra-laboratory reproducibility of Thera
Each control was evaluated in 6 different series. The mean values were 4% for RNS85 (range: 4–4%), 16% for RNS175 (range: 15–17%) and 33% (range: 32–34%) for RNS96. The reproducibility CVs were 5% for RNS175 and 3% for RNS96. All the results were identical for RNS85, which is close to the limits of quantification previously published for pyrosequencing (4%).
Mean, median and extreme values for each CpG and for the mean of the 4 or 5 CpGs analyzed with the PSQ and Thera assays
Mean, median and extreme values for each CpG and for the mean of the 4 or 5 CpGs analyzed with the PSQ and Thera assays
Comparison of the prognostic impacts when evaluating MGMT promoter methylation with therascreen pyrosequensing (Thera) and “standard” pyrosequencing (PSQ). The previously determined cut-offs of 8% and 12% were tested to determine the associated Hazard ratio (HR) and the level of significance (represented by the
Agreement between the two assays. Number of patients identically classified dichotomizing patients in 3 classes with cut-offs at 8 and 12% (A) or in two classes with cut-offs at 8% (B) or 12% (C)
Comparison between Thera and PSQ. Agreement between the two tests for CpG76, CpG77, CpG78 and the mean of the 4 or 5 CpGs is described using scatter and Bland-Altman plots.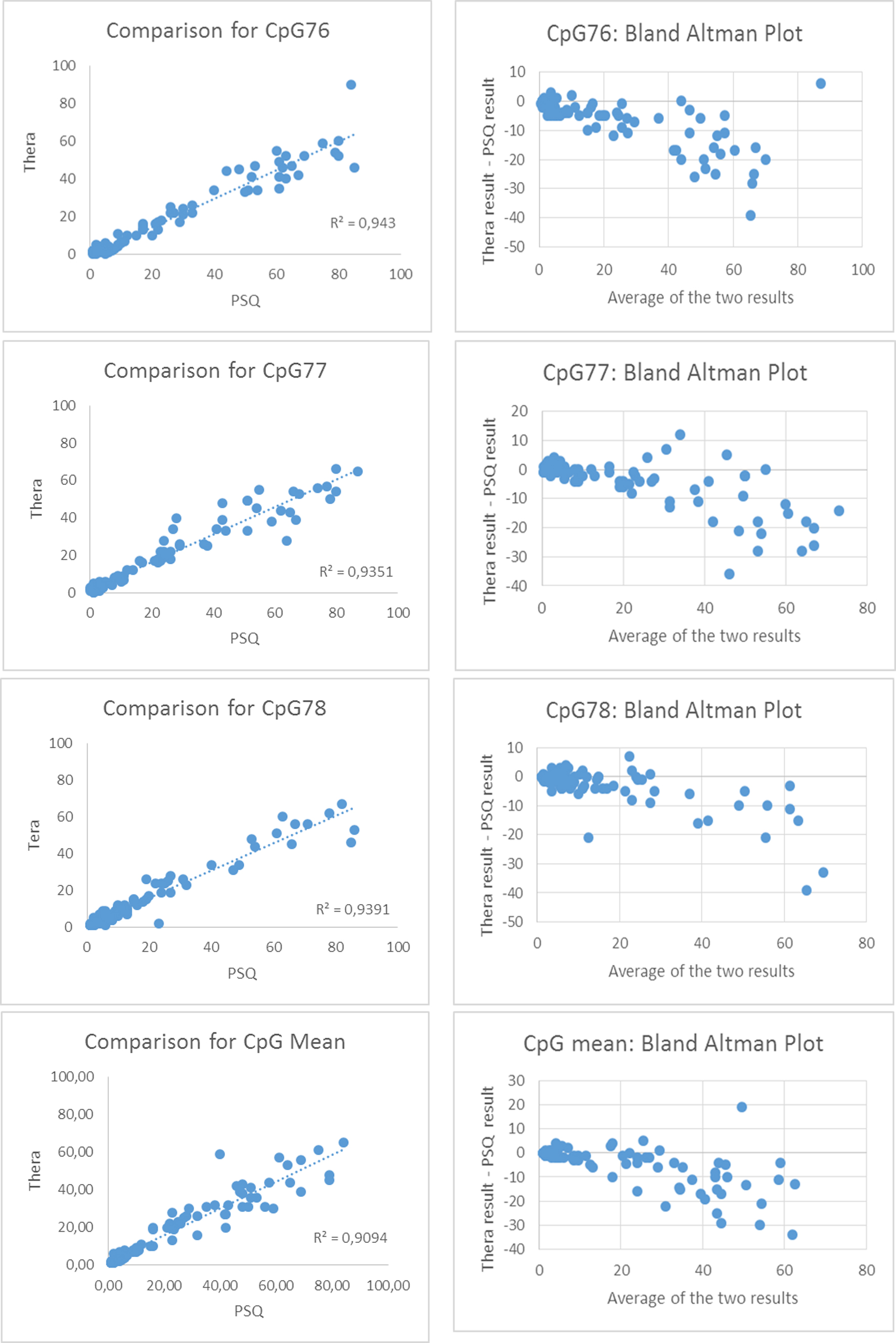
Results for samples classified differently by PSQ and Thera assays. For patients without event occurrence, time to death or time to recurrence are written in italics
Kaplan-Meier analysis of overall survival (OS) according to MGMT promoter methylation status. M: patients with a value above the calculated cut-off and therefore considered as “methylated”; UM: patients with a value below or equal to the calculated cut-off and therefore considered as “unmethylated”.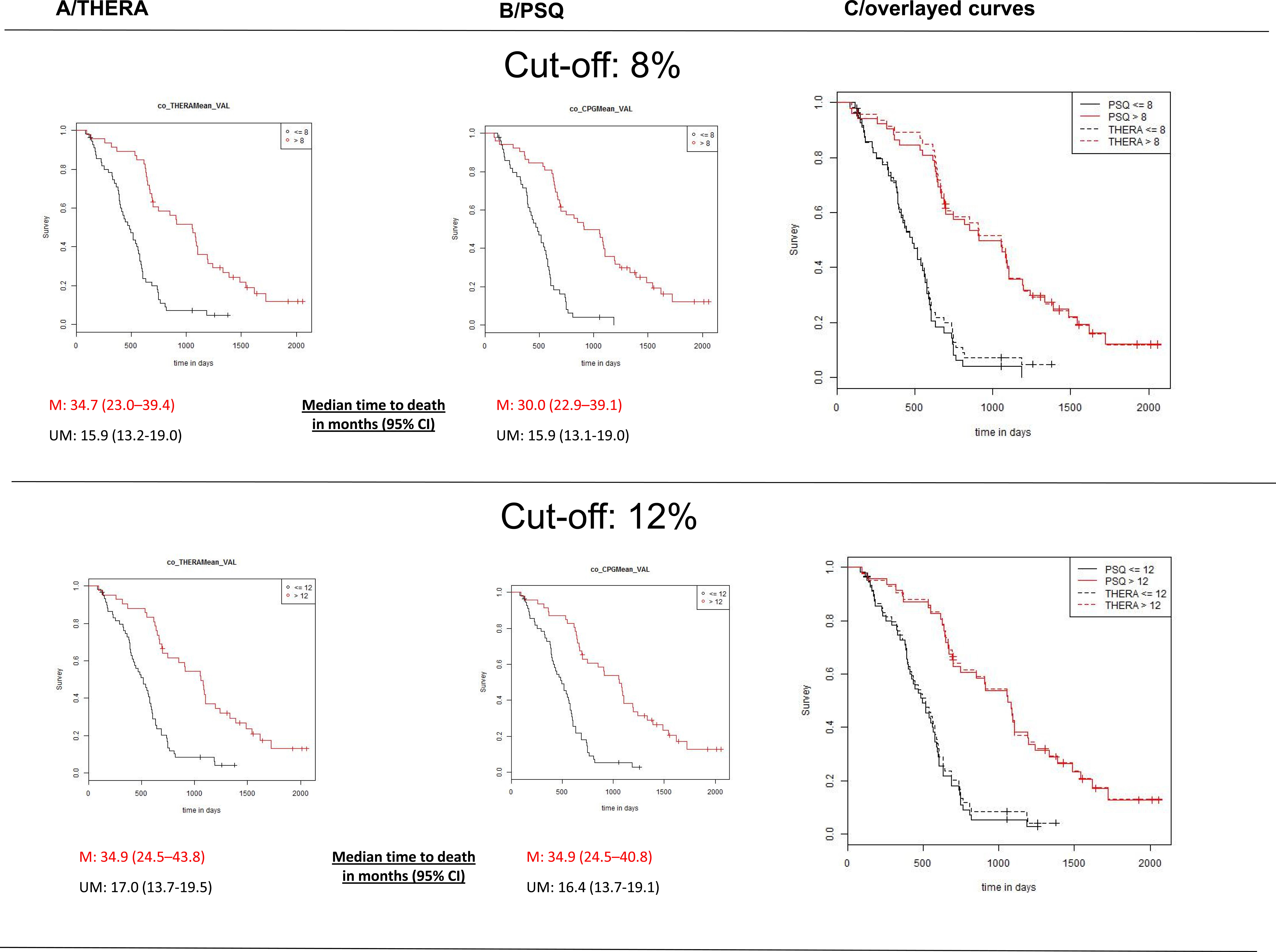
The median percentages of methylation for the studied population were 7% when using Thera (range 1–65%) and 8% when using PSQ (range 1–84%) (Table 1). A strong correlation was observed for CpG76, 77, 78 and CpG mean analyzed using the PSQ and Thera assays. However, almost systematically, lower results were recorded with Thera, especially for values greater than 20% (Fig. 1).
Validation of the pre-defined cut-offs 8% and 12% for Thera
We recently recommended a classification of MGMT
promoter methylation status into three subgroups: “unmethylated” (0–8%), “methylated” (13–100%) and a grey zone for patients with intermediate values (9–12%) [5]. The cut-offs 8 and 12% were tested in this series of patients. The percentages of patients classified as “methylated”, when employing a cut-off of 8% and 12% for PSQ, were 50% and 44% with AUCROC values of 0.69 for OS (Table 2). These data are almost identical to those obtained for the overall population (
Kaplan-Meier survival curves displaying the OS of patients dichotomized according to these cut-offs are presented in Fig. 2. At a methylation cut-off of 8%, median OS were 34.7 months (95% CI 23.0–39.4) versus 15.9 months (95% CI 13.2–19.0) for respectively methylated and unmethylated patients analysed with Thera, with a p value of 1.3 10
Discussion
MGMT promoter methylation is recognized as an effective predictor of response to TMZ for newly diagnosed GBM patients. Among the different techniques to analyze MGMT status, pyrosequencing is regarded as a very robust technique and its clinical utility has been validated in several independent studies [7, 8, 9, 10, 11, 12, 13, 14]. Pyrosequencing provides the percentage of methylated alleles of each CpG site analyzed and generally the average of the different sites is used to classify patients as “methylated” or “unmethylated”. The 5 most commonly analyzed CpGs are CpGs 74 to 78 and commercial kits are available to assess them, allowing reproducible and comparable results from one laboratory to another. The more recently launched Thera kit quantifies the methylation levels of CpGs 76 to 79. We have previously described that methylation can be heterogeneous from one site to the other [15]; analyzing different CpGs could therefore have an impact on the mean result. As a corollary the thresholds optimized for a combination of CpGs may not be optimal for additional combinations.
A comparison of CpGs one by one demonstrated a strong correlation between the results obtained with PSQ and Thera for common CpGs 76, 77 and 78, but with lower results with Thera for values above 15–20%. In our own experience, bisulfite treated DNA is very stable, so the delay of 2–3 months between Thera analysis and bisulfite treatment can’t explain these lower results. The same observation was performed for the mean values. As we propose threshold values of 8 and 12%, this difference has a minor impact for patient’s classification as “methylated” and “unmethylated”. With cut-offs at 12% and 8%, 97% and 95% of patients were identically classified according to the two techniques. When considering a three-class classification, among the 7 patients differently classified, 6 would have been classified as methylated with one technique and as being in the grey zone with the alternate technique. Specific authors have proposed higher cut-offs between 25% and 35% to classify patients [8, 11, 16]. In our study, the higher the cut-off increased, the higher the percentage of discordant cases. For example, values of 25%, 35% and 50% associated with 6%, 9% and 21% of patients having been differently classified within the two techniques.
To validate a clinical cut-off, we tested values of 8 and 12%. Forty-one percent of patients had values
In conclusion, Thera and PSQ may both be used to analyze MGMT status in glioblastomas. Intra-laboratory reproducibility for Thera was good and others have previously reported a high analytical performance of this kit, including inter-laboratory reproducibility [3]. The same cut-offs can be applied for the two kits, although they do not interrogate exactly identical CpGs.
Conflict of interest
The experiments were undertaken with the understanding and written consent of each subject. The study was conformed with the code of ethics of the word medical association.
Footnotes
Acknowledgments
We gratefully thank M. Marty for her technical contribution. The authors acknowledge the Centre de Ressources Biologiques (CRB) Santé of Rennes (BB-0033-00056), the Tumorothèque de Caen – Basse Normandie and the AP-HM Tumor Bank or Marseille (AC 2013-1786) for managing patient samples. Funding was provided by the French Ministry of Health (Support for Costly Cancer Technical Evaluation – STIC – Gov-0478).
