Abstract
BACKGROUND AND OBJECTIVE:
Studies in developing animals have demonstrated that when anesthetic agents, such as propofol, are early administered in life, it can lead to neuronal cell death and learning disabilities. However, the mechanisms causing these effects remains unknown. A recent report found that propofol could significantly upregulat miR-206 expression in the human ASCs. miR-206 could also induce apoptosis in human malignant cancers. Therefore, in this study, we hypothesized that propofol induces neurotoxicity in human embryonic stem cells (hESCs).
METHODS:
hESCs were exposed to propofol (50
RESULTS:
hESCs exposed to propofol showed a significant increase in TUNEL positive cells and cleaved caspase-3 expression, followed by the upregulation of miR-206 and PUMA expression. Targeting PUMA inhibits propofol-induced cell apoptosis; miR-206 knockdown decreased propofol-induced cell apoptosis, cleaved caspase-3 and PUMA expression.
CONCLUSIONS:
Propofol induce
Keywords
Introduction
Propofol is an intravenous agent, which is commonly used for induction and maintenance of anesthesia, procedural and critical care sedation in children. Propofol has a neuroprotective role in pathogenic situations. However, it also has neurotoxic effect in children, raising the concerns in the developing brain with its use and the possible neurocognitive decline as a result [1]. The data from animals also showed the neurotoxic effect of propofol.
Cattano et al. has showed that 1/4 dose for surgical anesthesia required could induce neuroapoptosis in infant rodents [2]. Creeley et al. has reported that apoptosis of neurons was significantly increased after exposure to propofol anesthesia for 5 hours in the developing non-human primate brain [3]; the mechanism by which propofol neurotoxicity occurs remains unknown. Pearn et al., recently reported that the neurotoxicity of propofol was mediated by p75 Neurotrophin receptor activation [4].
The mechanism by which propofol-induced neurotoxicity is unclear. Although no direct evidence available in a human model, much of the research in the neurodegenerative field was recently performed in animal models. In addition, due to the ethical factors, the study of neurotoxicity of propofol for children is impractical [5, 6]; the only information available to humans is a limited number of epidemiological studies. Therefore, the neurological effects of propofol on young children remain uncertain.
Human embryonic stem cells (hESCs) are pluripotent cells derived from the inner cell mass of human blastocysts [7]. hESCs could differentiate into any cell type and make them a potentially powerful model of human pathophysiology and physiology. Therefore, neurons derived from hESCs are a valuable model to directly study the effects of anesthetics on immature, human-derived neurons.
MicroRNAs are emerging as robust players of gene regulation. Numerous miRNAs have been implicated in a variety of cellular processes including differentiation, cell proliferation, apoptosis, embryonic development, stress response, stem cell renewal and metabolism [8, 9, 10, 11, 12]. MicroRNAs have also been implicated to play important roles neurological diseases and neurotoxicity [13]. However, the effect of miRs on anesthetic-induced neurotoxicity has yet to be studied.
One miR, miR-206, has been shown to be associated with neurotransmitter regulation in developing neuronal cells [14]. In a healthy brain, miR-206 can only be detected at very low levels, which may be important for proper neural system development. However, introducing miR-206 into the oligodendrocyte cell line CG-4 causes a decrease in TPPP/p25 expression, resulting in inhibition of oligodendrocyte differentiation [15].
A recent report found that propofol anesthesia affects miRNA expression in rat liver [16]. Kim et al. has showed that exposure of adipocyte-derived adult stem cells to propofol increased miR-206 expression [17]. Wang et al. has reported that overexpression of miR-206 promotes neural cells apoptosis and low expression of miR-206 inhibits cell apoptosis [18]. We hypothesized that the miR-206 plays a role in the increased cell death observed in the hESCs following propofol exposure.
Previous study has reported propofol, at clinically relevant concentrations, attenuated cell death through both autophagic and apoptotic mechanisms in the rat hippocampus after a cerebral I/R insult [19].
Li et al. has reported that propofol inhibits H2O2-induced injury in cardiac H9c2 cells via decreasing NF-
Materials and methods
hESCs culture
hESCs used were from lines H1 between 55–70 passages. The cells were obtained from Wicell under MTA to UCLA Stem Cell Core Facility. hESCs were grown on irradiated mouse embryonic fibroblast feeder cells in hESC feeder-dependent medium. The hESCs were routinely passaged once per week by using dispase and collagenase type IV (Gibco, part of Invitrogen, Carlsbad, CA, USA). Cells were maintained in a 37
Differentiation of neurons from hESCs
hESCs were incubated in neural induction medium for 24 h. Then the cells were dissociated into small clumps by mechanical scraping. Small clumps were cultured in NMM supplemented with 10 ng/ml glial cell line-derived neurotrophic factor, 10 ng/ml brain-derived neurotrophic factor (BDNF, R&D Systems, USA), 10 ng/ml insulin-like growth factor 1 (IGF1, Peprotech, USA), 500
miR-206 and PUMA siRNA transient transfection
The human miR-206 inhibitor (anti-miR-206) and inhibitor control (control) were acquired from RiboBio Co., Ltd (Shanghai, China). PUMA siRNA and control siRNA were purchased from Santa Cruz (Shanghai, China); Anti-miR-206 or PUMA siRNA or control siRNA were transiently transfected into hESCs cells for 48 h at concentration of 100 nM using Lipofectamine 2000 reagent (Invitrogen) according to the manufacturer’s procedure.
Propofol exposure
The dose of propofol used clinically typically ranges from about 1–10
Quantification of miR-206 expression
Total miRNA was isolated through the mirVana miRNA isolation kit (Ambion). Quantitative expression studies of miR206 transcripts were performed using TaqMan miRNA Assays (Applied Biosystem). The cDNA was obtained using the High Capacity RNA-to-cDNA Kit (Applied Biosystem). cDNA was amplified using specific primer for miR-206 (Applied Biosystem). qRT-PCR was performed using an Applied Biosystems 7900HT Fast RT-PCR system. The expression level of miR-206 was calculated according to the 2-
Western blot analysis
Western blotting was carried out as described previously [25]. The primary antibodies used were mouse anti-PUMA, rabbit anti-cleaved caspase-3, and mouse anti-GAPDH. The horseradish peroxidase-conjugated secondary antibody (1:5000; Santa Cruz Biotechnology) and chemiluminescence Supersignal (Pierce) was used to detect protein. Protein loading was normalized for GAPDH signal. Band intensity was quantified versus a control sample (considered as 1).
Propofol increases the number of TUNEL-positive cells. Imaging of cell apoptosis determined by TUNEL assay. Nuclei of apoptosis was identified as TUNEL positive (green fluorescent) and total nuclei by DAPI counters taining (blue fluorescent). Scale bar represents 50 
miR-206 is required for the apoptotic activity of propofol hESC-derived neurons were transiently transfected with anti-miR-206 or control miR or/and treated with 50 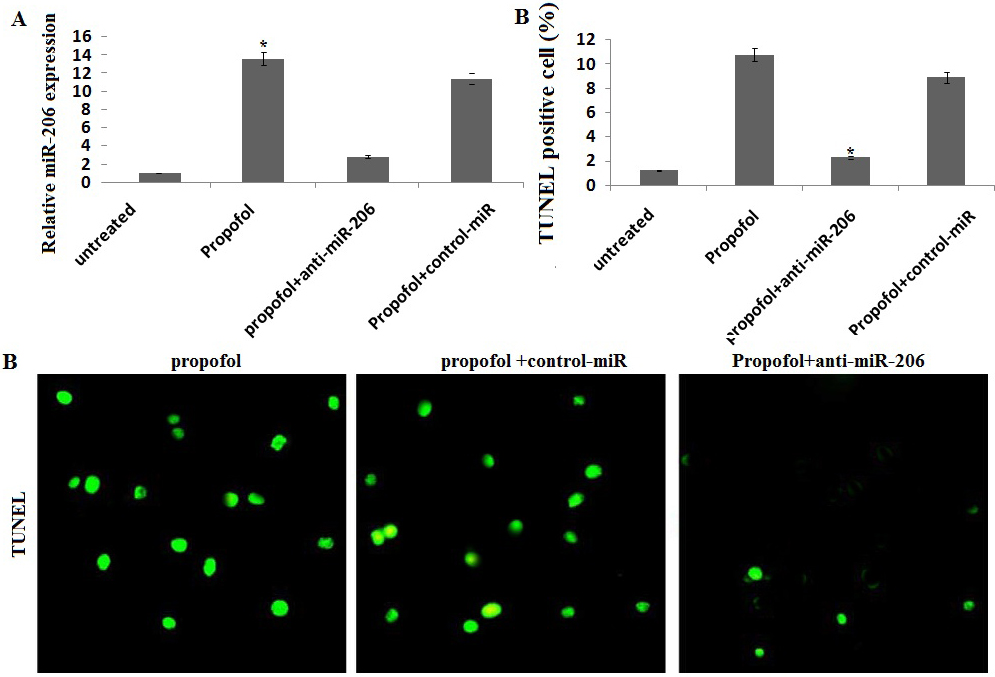
TUNEL staining was performed using an in situ cell death detection kit (Roche, Guangzhou, China) as described previously [26]. TUNEL positive nuclei were quantified blinded for 5 fields per slide using a fluorescence microscope (Zeiss Axioplan2 fluorescence microscope).
Statistical analysis
The data were expressed as the means
Results
Propofol induces cell death in hESC-derived neurons
The TUNEL-positive cells was 0.82%
Propofol upregulates miR-206-dependent apoptosis in hESC-derived neurons
We first investigated the effects of propofol on miR-206 expression of hESC-derived neurons. Exposure to 50
miR-206-dependent induction of PUMA by propofol
We then determined whether propofol-induced miR-206-dependent Puma expression. Exposure to 50
Caspases family plays an important role to regulate cell apoptosis, the mechanisms of which involved in the upstream initiators and downstream effectors. For example, caspase-3 is a main caspase effector. Therefore, we detected the effect of propofol on activity of caspase-3. As seen in Fig. 3, propofol significantly increased the caspase-3 activity by western blot assay. However, transient miR-206 knockdow inhibited propofol-induced caspase-3 upregulation and activation (Fig. 3).
Propofol directly mediates miR-206/Puma induction. Cells were transfected with anti-miR-206 and/or treated with 50 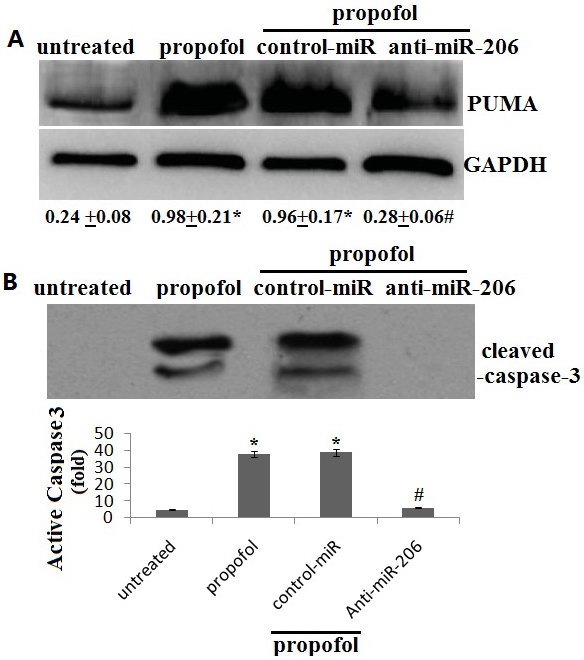
PUMA mediates propofol-induced apoptosis. Cells were transfected with PUMA siRNA and/or treated with 50 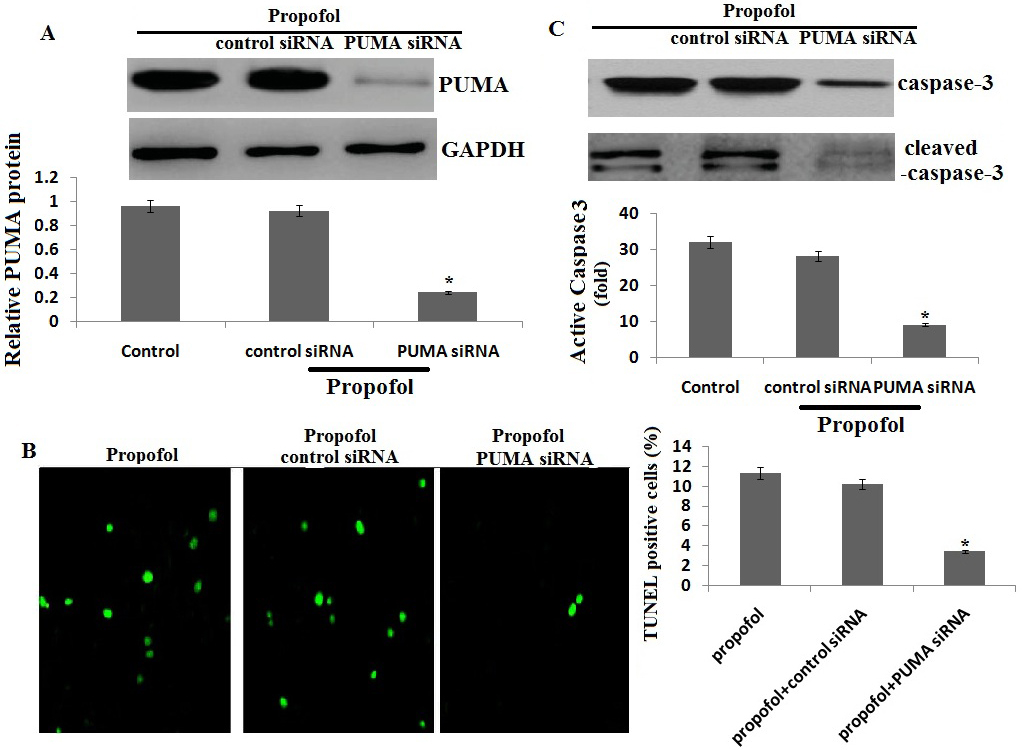
We then investigate the role of PUMA in propofol-induced apoptosis. Exposure to 50
Discussion
Studies have shown that exposure of immature animals to anesthetic drugs, such as N-methyl d-aspartate receptor-antagonist properties (e.g. propofol, pentobarbital, isoflurane, diazepam, halothane, and) or gamma-aminobutyric acid-mimetic receptor-agonist could induce apoptotic degeneration of neurons in various brain regions [27, 28]. Anesthetics can lead to learning disabilities later in life in rodent models when administered early in life [28]. In addition, propofol could induce neuroapoptosis when administered for 5–6 hours in rodent and primate models [5]. Kahraman et al. has recently reported that exposure of 5 and 50
The mechanisms by which propofol induce neurotoxicity are still unknown. Studies have found that propofol could significantly alter the expression patterns of a group of miRNAs in immature rat astrocytes and rat primary embryonic neural stem cells, and the propofol-regulated miRNAs are specifically associated with cell apoptosis [30, 31]. However, the specific mechanism of these effects require further investigation. In our study, miR-206 was significantly upregulated in hESCs cells when exposure to 50
p53-upregulated modulator of apoptosis (PUMA) was identified as a BH3-only Bcl-2 family protein that has an essential role in apoptosis induced by a variety of stimuli [32]. The role of miRNAs in regulating the expression of PUMA and regulation of cell survival is well-established [33, 34]. In the present study, we found that miR-206, PUMA and cleaved caspase-3 were significantly upregulated following exposure to 50
In conclusion, Propofol induce
Conflict of interest
No potential conflicts of interest were disclosed.
