Abstract
Objectives
MicroRNA-206 (miR-206) is downregulated in many human malignancies, which correlates with tumour progression. This study characterized the contribution of miR-206 to the initiation and progression of human breast cancer.
Methods
Consecutive primary breast cancer patients who received radical resection were enrolled. Breast cancer tissue samples were obtained during surgery. MiR-206 levels in matched pairs of cancer tissue and normal adjacent tissue (NAT) samples were examined using quantitative reverse transcription–polymerase chain reaction. The relationship between miR-206 levels and clinicopathological characteristics and overall survival was also investigated.
Results
In 128 patients with breast cancer, miR-206 was downregulated in 119 (93%) tumour tissue compared with their matched NAT samples. Decreased miR-206 was significantly associated with advanced clinical stage and lymph node metastasis. Univariate and multivariate Cox proportional hazard regression analysis revealed that a low miR-206 level was an unfavourable prognostic factor for overall survival, in patients with breast cancer.
Conclusions
This study indicated that miR-206 may be a good candidate as a novel prognostic indicator in breast cancer patients.
Introduction
Breast cancer is one of the leading causes of cancer-related death and is a common leading malignancy among women worldwide. 1 Although many treatment advances have resulted from improving clinical instruments and methods, metastasis still leads to cancer mortality and poor prognosis. 2 Understanding the signalling pathways involved in breast carcinogenesis is important for the development of more effective therapies than those currently available.
MicroRNAs (miRNAs) are small (20–24 nucleotide) noncoding RNA gene products that post-transcriptionally modulate the products of gene expression by negatively regulating the stability or translational efficiency of their target mRNAs. 3 The importance of miRNAs in cancer was first recognized when miRNA genes were found to be specifically deleted in leukaemia. 4 Subsequently, miRNAs have been shown to be differentially expressed in many cancers 5 and are now widely believed to play an essential role in many malignancies, acting as either tumour suppressors or oncogenes. 6 Studies on breast cancer have analysed various miRNAs that might influence disease development and progression.7,8
One tumour-suppressive miRNA is miR-206. Using high-throughput technology such as miRNA oligonucleotide arrays and quantitative real–time polymerase chain reaction (PCR) for validation, research has shown aberrant miR-206 levels in human malignancies such as lung cancer, 9 rhabdomyosarcoma, 10 and endometrioid adenocarcinoma. 11 Downregulation of miR-206 has also been shown in breast cancer cells, 12 although its role in disease progression and overall survival of patients with breast cancer is still unclear.
The present study assessed miR-206 levels in patients who underwent surgery for primary breast cancer by disease stage, tumour grade, histology type and nodal involvement. The association between miR-206 and overall survival in these patients was also investigated.
Patients and methods
Study population
Consecutive primary breast cancer patients who underwent radical resection at The Second Hospital of Shandong University, Jinan, China, between January and October 2006, were consecutively enrolled in this prospective study. Eligible patients were required to fulfil the following criteria: age 18–75 years; performance status of 0 or 1 based on the Eastern Cooperative Oncology Group (ECOG) criteria; 13 adequate haematological, hepatic, renal and cardiac function. Patients were excluded if they had previous or secondary malignancy, were pregnant, had uncontrolled and serious infection and/or had previously undergone radiation therapy, chemotherapy, or immunotherapy.
The diagnosis and histological grade of each case were independently confirmed by two pathologists based on World Health Organization tumour classification. 14 The tumour grading systems combine nuclear grade, tubule formation and mitotic rate. In general, each element is given a score of one to three (one being the best and three the worst) and the score of all three components are added together to give the overall grade. Grade I (low grade) is given to well-differentiated tumours that all form tubules and have a low mitotic rate; grade III (high grade) indicates poorly differentiated tumours with few or no tubules and high mitotic rates. The clinical stage was classified according to the American Joint Committee on Cancer (AJCC) tumour–node–metastasis (TNM) classification system. 15 This study was approved by the Medical Ethics Committee of The Second Hospital of Shandong University and written informed consent was obtained from all patients.
RNA extraction and quantitative real-time RT–PCR
Fresh samples of tumour tissue from all cases and paired normal adjacent tissue (NAT, >2 cm from cancer tissue) were obtained during elective radical resection surgery. Fresh specimens were stored at 4℃ for 24 h in RNAlater® (Ambion Inc [Life Technologies], Foster City, CA, USA), then at −80℃ in liquid nitrogen, until further use.
Total RNA from 50 mg breast cancer and NAT biopsies was isolated using TRIzol® reagent (Invitrogen, Carlsbad, CA, USA) according to the manufacturer’s instructions. RNA quality and quantity were assessed using a Bioanalyzer 2100® system (Agilent Technologies, Santa Clara, CA, USA). miR-206 and RNA U6 (as an internal control) specific cDNA were synthesized from total RNA using gene-specific primers, according to the TaqMan® miRNA assay protocol (Applied Biosystems, Foster City, CA, USA). Reverse transcription (RT) reactions contained 10 ng total RNA, 50 nmol/l stem-loop RT primer, 1 × RT buffer, 0.25 mmol/l each deoxynucleotide triphosphate (dNTP), 3.33 units/μl MultiScribe™ (Invitrogen) reverse transcriptase and 0.25 units/μl RNase inhibitor in a total volume of 7.5 μl (Applied Biosystems). Reactions were incubated in a 96-well plate for 30 min at 16℃, followed by 30 min at 42℃ and 5 min at 85℃, then held at 4℃.
Real-time polymerase chain reaction (PCR) was performed using the 7500® real-time PCR system (Applied Biosystems). Reactions were carried out in 96-well plates in a total volume of 10 μl (0.67 μl RT products, 1 × TaqMan® Universal PCR master mix, and 1 μl TaqMan® miRNA assay primer and probe mix). The miR-206 PCR primers were: sense: 5′-GGAATGTAAGGAAGTGTG-3′; antisense: 5′-GAGCAGGCTGGAGAA-3′. The cycling programme involved preliminary denaturation at 95℃ for 10 min, followed by 45 cycles of denaturation at 95℃ for 16 s and annealing/elongation at 60℃ for 10 min. All samples were processed in triplicate.
The threshold cycle (C
Immunohistochemical analysis of OR and PR levels
Protein levels for the oestrogen receptor (OR) and progesterone receptor (PR) were evaluated by immunohistochemistry using the mouse antihuman OR (clone 1D5) and PR (clone PGR-1A6) monoclonal antibodies, respectively (both from DAKO, Glostrup, Denmark). All immunohistochemistry procedures were performed according to the manufacturer’s instructions and as described previously. 17 Only tissues with distinct nuclear staining for OR and PR in >10% of the tumour cells were recorded as positive. 17
Follow-up
Survival time was calculated from the date of surgery to date of death or last follow-up.
Statistical analyses
Statistical analyses were performed using the SPSS® statistical software package, version 17.0 (SPSS Inc., Chicago, IL, USA) for Windows®. Values are expressed as mean ± SD. Differences in miR-206 levels between breast cancer tissue and paired NAT samples were compared using a paired Student’s t-test. MiR-206 levels between groups (inter-group differences) were compared using an unpaired Student’s t-test. The correlation between miR-206 levels and overall survival was analysed by Spearman’s correlation coefficient. The joint effect of covariables was examined using the Cox proportional hazards regression model. All tests were two-tailed, and the significance level was set at P < 0.05.
Results
A total of 128 patients with breast cancer was enrolled. The median age of these patients at diagnosis was 49 years (range, 26–73 years). Among patients, 111 (87%) had ductal carcinoma, five (4%) had lobular carcinoma, and 12 (9%) had other forms of breast cancer. In terms of histological grade, 32 (25%) patients were grade I, 71 (55%) were grade II, and 35 (20%) were grade III. Lymph node involvement was found in 79 (62%) patients. At the time of surgery, 13 (10%) patients were classified as TNM stage I, 78 (61%) as stage II, and 37 (29%) as stage III.
MicroRNA-206 was downregulated in 119 (93%) breast cancer tissue compared with matched NAT samples. The mean ± SD Δ C T value of miR-206 in breast cancer tissue samples was 13.15 ± 1.73 compared with 9.28 ± 0.85 in NAT samples (P < 0.001).
Levels of micro RNA-206 (miR-206) in tumour tissue samples, stratified by clinicopathological features, from 128 patients with breast cancer.

Data presented as mean ± SD.
Δ C T = C TmiR-206 − C TU6.
Unpaired Student’s t-test.
OR, oestrogen receptor; PR, progestin receptor; NS, not statistically significant (P ≥ 0.05).
At the time of scheduled analyses (December 2012), 47 (37%) patients were alive; 81 (63%) had died. The median follow-up time was 76 months. There was a significant correlation between miR-206 level (mean ± SD Δ
C
Univariate and multivariate Cox regression analyses of the relationship between tumour tissue micro RNA-206 (miR-206) levels and clinicopathological features, in tumour samples from 128 patients with breast cancer.
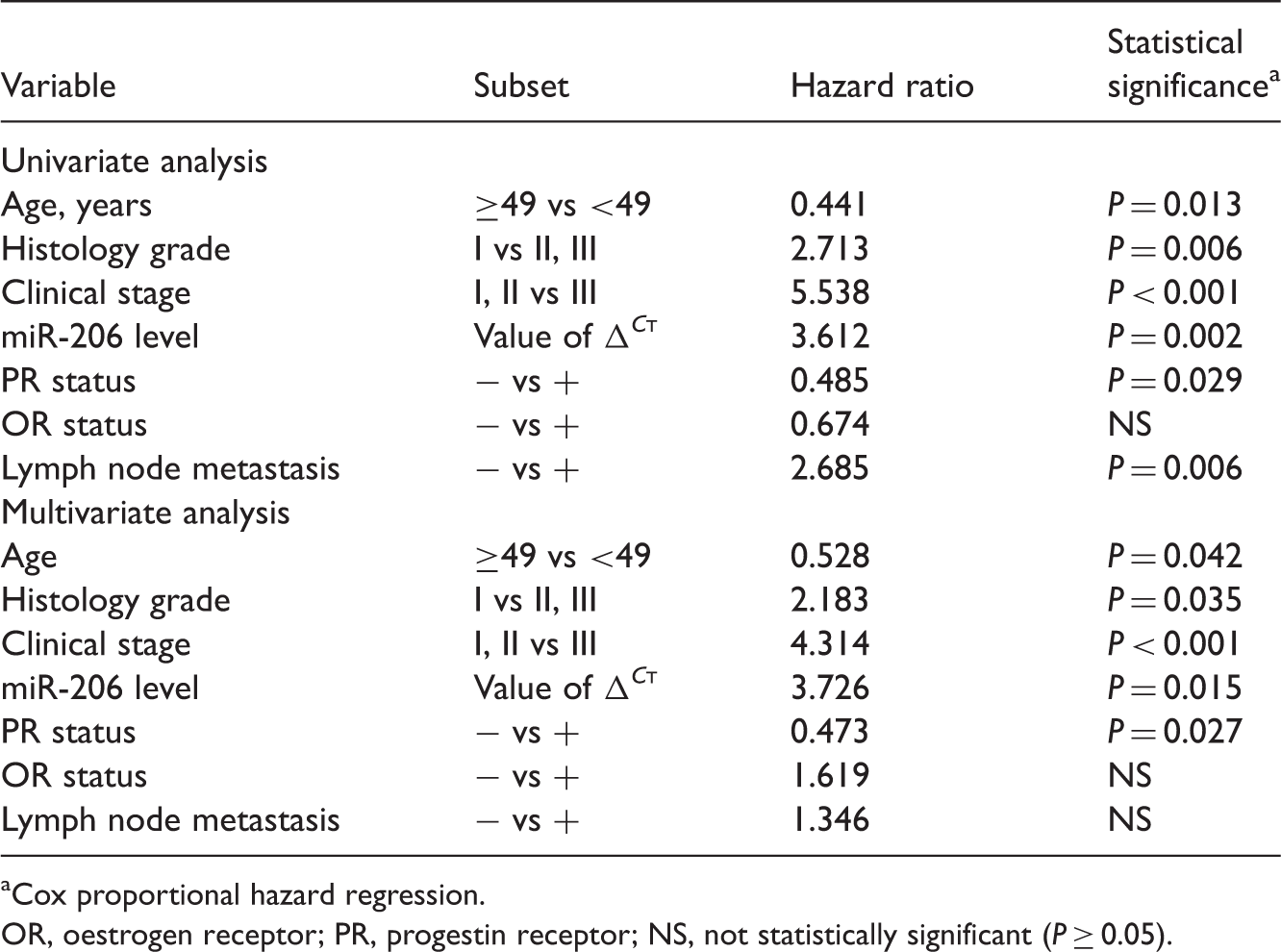
Cox proportional hazard regression.
OR, oestrogen receptor; PR, progestin receptor; NS, not statistically significant (P ≥ 0.05).
Discussion
Currently, it is estimated that one in eight women will develop breast cancer. 18 Although progress has been made in the diagnosis and treatment of breast cancer there is still room for improvement, particularly in cancer therapeutics. The miRNAs are involved in many biological processes including cell-cycle regulation, cell growth, apoptosis, cell differentiation and stress response. 19 Additionally, miRNAs can modulate both oncogenic or tumour suppressor pathways. 20 A better understanding of the role of miRNAs may lead to the discovery of new therapies in cancer, including breast cancer, and research suggests that miRNAs play a critical role in breast cancer, in particular. 21 miRNAs have been implicated in breast-cancer cell proliferation, invasion, angiogenesis and metastasis. 22
To improve understanding of its role in human breast cancer, the present study examined levels of miR-206 in matched fresh-frozen breast cancer tissue and NAT samples. MiR-206 was significantly downregulated in cancer tissue compared with NAT, and this downregulation was significantly associated with advanced clinical stage, lymph node metastasis and poor prognosis. This suggested that miR-206 downregulation may be acquired in the course of tumour progression and, in particular, during the acquisition of metastatic potential. Additionally, survival rates were significantly worse in patients with low miR-206 levels, independent of clinical stage, PR status, histological grade and age, suggesting that it could serve as a prognostic marker for survival.
Located on chromosome 6p12.2, miR-206 functions as a tumour suppressor, inhibiting cell growth and inducing apoptosis in rhabdomyosarcoma, 10 breast cancer, 23 endometrial endometrioid carcinoma 11 and lung cancer. 9 Cell migration and invasion are also inhibited by miR-206 in rhabdomyosarcoma, endometrial endometrioid carcinoma and lung cancer cells. In vivo, miR-206 acts as a tumour suppressor in rhabdomyosarcoma in xenotransplanted mice. 24 Additionally, miR-206 is markedly decreased in OR alpha-positive human breast cancer tissues, and transfection of miR-206 into MCF-7 breast cancer cells inhibits cell growth in a dose-dependent manner. 25 miR-206 is significantly downregulated in laryngeal squamous cell carcinoma tissue, 26 and inverse correlations have been shown between miR-206 level and tumour grade, nodal metastasis and clinical stage in laryngeal squamous cell carcinoma. 26 It has also been reported that miR-206 is one of five miRNAs that exhibit stage-dependent differential expression in human colorectal cancer. 27 Furthermore, serum miR-206 levels might be used to distinguish rhabdomyosarcoma from nonrhabdomyosarcoma tumours. 28 Studies using quantitative PCR, Western blot analysis, and reporter assays and bioinformatic prediction programmes, have identified several targets of miR-206, including the met proto-oncogene (hepatocyte growth factor receptor), 24 notch3, 29 and vascular endothelial growth factor. 26 The molecular genetic basis of carcinogenesis and cancer progression is complex, however, and the connection between miRNAs and target mRNAs is not necessarily ‘one to one’. One miRNA can have over 100 mRNA targets, 30 and one mRNA can be regulated by a variety of miRNAs. 31 Thus, the potential regulatory circuitry associated with miR-206 may be enormous, and identification of the complex molecular network involved in its function remains to be determined.
In conclusion, miR-206 is frequently downregulated in breast cancer tissue, and decreased miR-206 levels are associated with advanced clinical stage and shorter overall survival, indicating that miR-206 may play an important role in tumour initiation and in the development and progression of breast cancer. Although the precise molecular mechanism surrounding its downregulation requires clarification, miR-206 might be a candidate therapeutic target and a potential biomarker for diagnosis and prognosis in breast cancer.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
