Abstract
BACKGROUND:
Ring Box Protein-1 (RBX-1), a component of SCF E3 ubiquitin ligases, has a crucial role in bladder urothelial cell carcinoma (UCC) carcinogenesis and progression.
OBJECTIVES:
In the present study, it is aimed to determine the expression of RBX-1 protein in bladder UCC and the association between tumor grade, stage and RBX-1 expression.
METHODS:
Ninety UCC samples and 20 samples containing foci of normal bladder urothelium were recruited and analyzed immunohistochemically in terms of RBX-1 expression. Immuno-reactivity scoring system (IRS) was used to determine RBX-1 expression levels.
RESULTS:
RBX-1 overexpression was associated with high tumor grade (
CONCLUSIONS:
In bladder UCC, RBX-1 overexpression is associated with high tumor grade and advanced stage and represents biological potential of invasiveness and aggressive disease. Results of the present study have to be supported with further studies to reveal clinical and therapeutic implications of RBX-1 overexpression in bladder UCC.
Introduction
Among all cancer types, bladder cancer (BC) is the ninth and most common malignancy of the urinary tract accounting for more than 380,000 novel cases and 150,000 deaths annually [1]. More than 90% of all initial cases are diagnosed histopathologically with urothelial cell carcinoma (UCC), 70% of them are non-muscle invasive bladder cancer (NMIBC) and the remaining 30% muscle-invasive disease (MIBC) [2]. MIBC has known to have a high risk of local invasion and distant metastasis [3].
Definite diagnosis is possible with pathological specimen investigation obtained via transurethral resection of bladder tumor (TURB) enabling histopathologic diagnosis, staging, grading, and further treatment decision-making process for bladder UCC [4]. Tumor stage, and grade are well known histopathologic parameters to have importance in prediction of prognosis [5]; however, they have been accepted insufficient to provide definitive prognosis in bladder UCC [6]. Due to a need have arised to develop personalized anticancer therapies and to improve prediction of disease outcome, novel molecular markers that contribute to bladder UCC carcinogenesis and progression have constituted a main issue of many investigations recently.
Ring Box-1 (RBX-1) protein, a RING component of SCF (Skp-1, cullins, F-box proteins) E3 ubiquitin ligases, plays an important role in cell cycle-related protein activities and numerous cellular processes such as DNA replication, signal transduction and gene transcription [7, 8, 9]. RBX-1 degradation or overexpression is present in multiple human cancer types such as colon, lung, melanoma, prostate and necessary for cancer cell growth and survival [8, 9, 10]. In a recent study evaluating RBX-1 expression in bladder UCC [11], knockdown of RBX-1 expression has been shown to prevent bladder cell growth and induce G2 cell cycle arrest and p53-dependent cell senescence, which is an important mechanism in bladder UCC carcinogenesis and progression [12]. Although the role of RBX-1 in bladder UCC has been established in several studies, the association between RBX-1 expression and clinicopathologic prognostic parameters of bladder UCC has not been well documented yet. According to our literature research, there is a single study determining the association of RBX-1 protein overexpression with prognosis of bladder UCC, including NMIBC only [6].
In the present study, we aimed to determine the association between RBX-1 expressions and tumor grade and stage in bladder UCC, comprising both MIBC and NMIBC.
Materials and methods
Patients groups
This retrospective study was approved by local ethics committee of Selcuk University in accordance with the Helsinki Declaration. The present study comprised ninety consecutive patients who underwent TURB and whose pathological diagnosis was bladder UCC between January 2010 and December 2013.
Tumor grade, bladder wall invasion and invasion depth (T stage) analyses were based on hematoxylin-eosin stained slides and re-evaluated by two pathologists (EC, PK). Besides the 90 UCC samples, 20 samples containing foci of normal bladder urothelium were also included.
Tumor grading and staging were based on World Health Organization (WHO)/International Society of Urological Pathology (ISUP) classification of tumors of urinary tract (2004) criteria and Union International Contre le Cancer (UICC) 2009 TNM system.
Immunohistochemistry
Paraffin-embedded tissues of chosen slides were collected and 4
Evaluation and scoring of immunohistochemically stained slides
The immunohistochemical RBX-1 stained slides were evaluated and scored by two pathologists (EC, PK) blinded to patients’ data. On light microscopic evaluation of each 4
Statistical analysis
All statistical analyses were performed using SPSS 20 (Statistical Package for Social Sciences) package program. In order to test normality of distribution Kolmogorov-Smirnov Z test was employed. Parametric analyses methods were employed for normal distributions and in abnormal distributions non parametric tests were preferred. Chi-square test, One-way ANOVA test with Tukey HSD post hoc test and nonparametric Kruskall Wallis test were used to determine the relation between RBX-1 expressions and clinicopathological data. All calculated P values were two-tailed, and
Results
Patients’ clinicopathologic characteristics
Ninety patients diagnosed with bladder UCC were evaluated in the present study. Out of these 90 patients, eighty-two were male and 8 female. Patients’ age ranged between 37 and 85 (mean 63 years).
Clinicopathologic characteristics of the patients are summarized in Table 1.
Clinicopathologic characteristics of the patients and related RBX-1 expressions
Clinicopathologic characteristics of the patients and related RBX-1 expressions
The urothelium of non-tumoral bladder tissue showed weak, low and mainly cytoplasmic immunohistochemical RBX-1 expression (X100).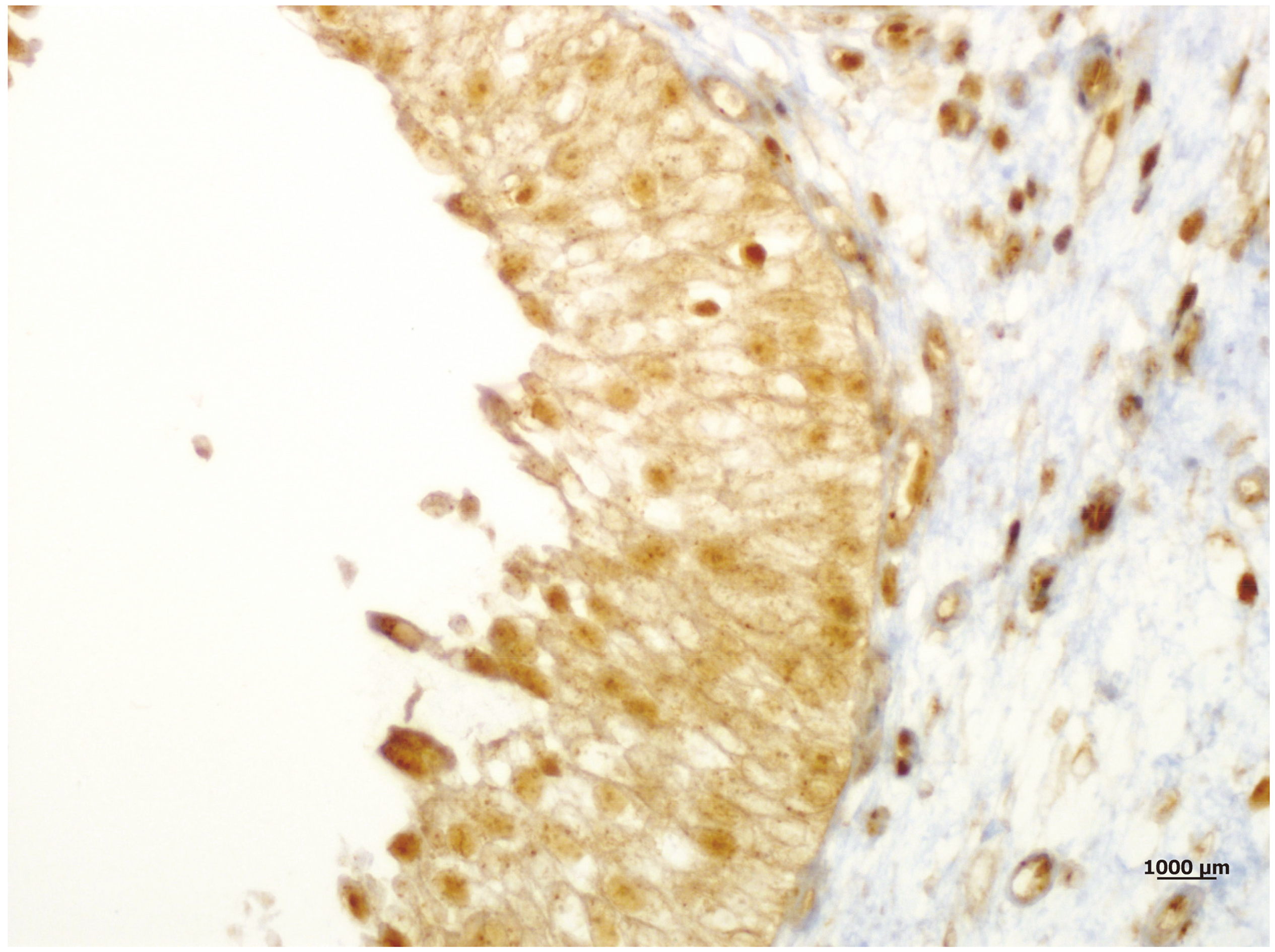
Low (1+) cytoplasmic and focal nuclear staining of RBX-1 in a noninvasive (pTa) low grade UCC sample (X100).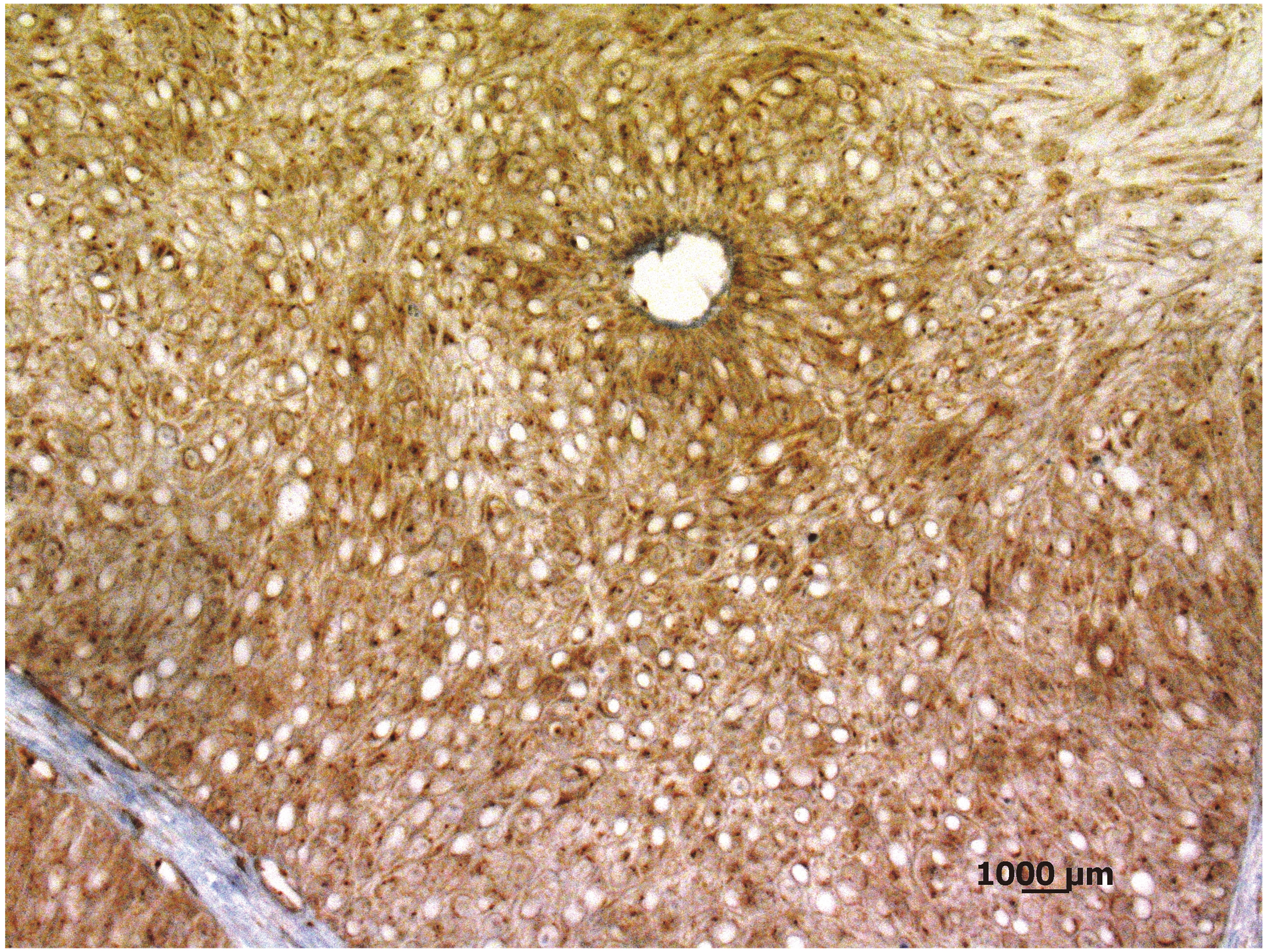
Moderate (2+) both cytoplasmic and nuclear staining of RBX-1 in a low grade pT1 tumour tissue (X100).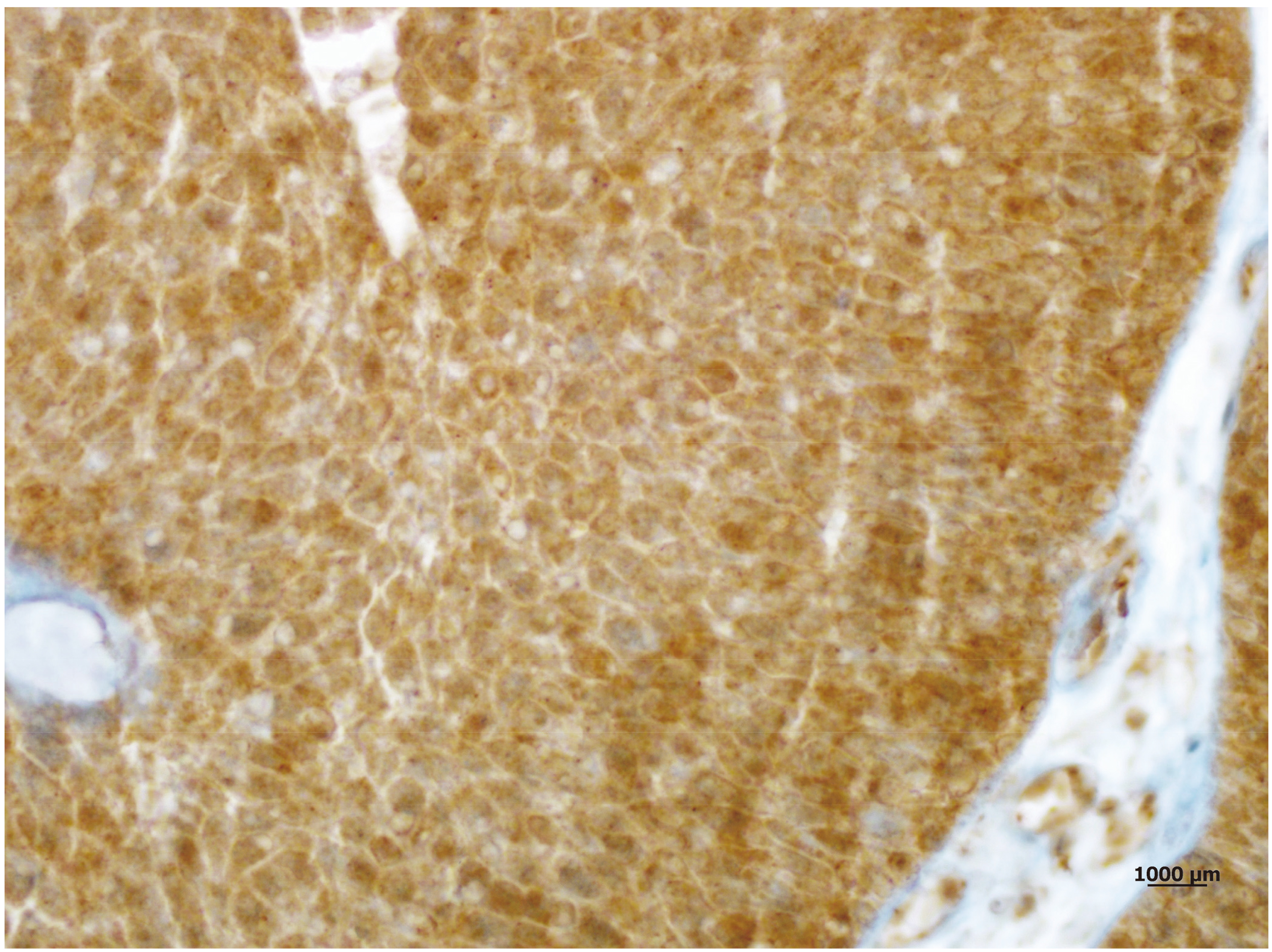
Strong and diffuse (3+) both cytoplasmic and nuclear staining of RBX-1 in a high grade muscle invasive tumour sample (X100).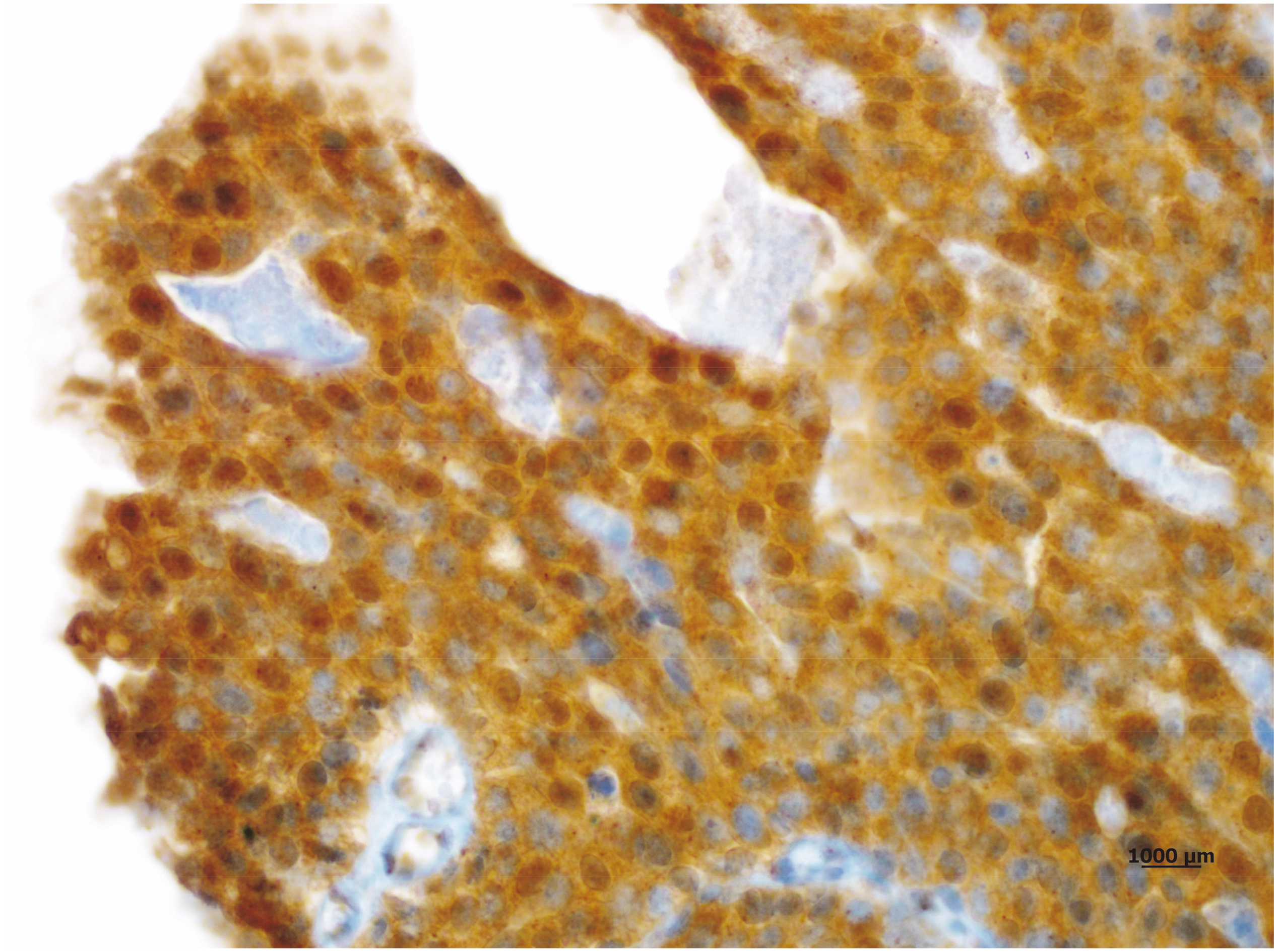
IRS scores of LG (low grade) and HG (high grade) tumors (Data are expressed as Mean 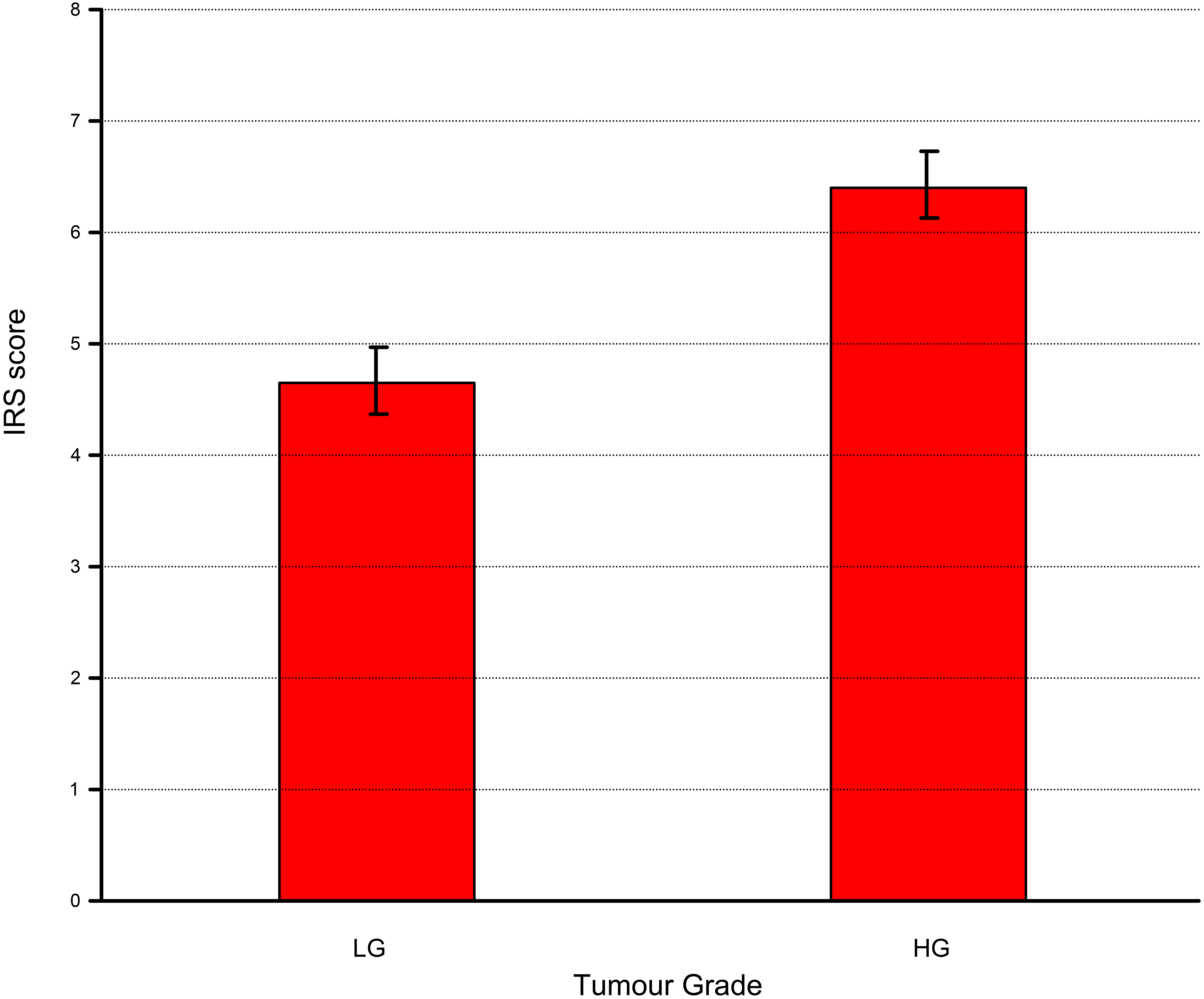
Association of IRS scores with tumor stage (LS: Low IRS, HS: High IRS) (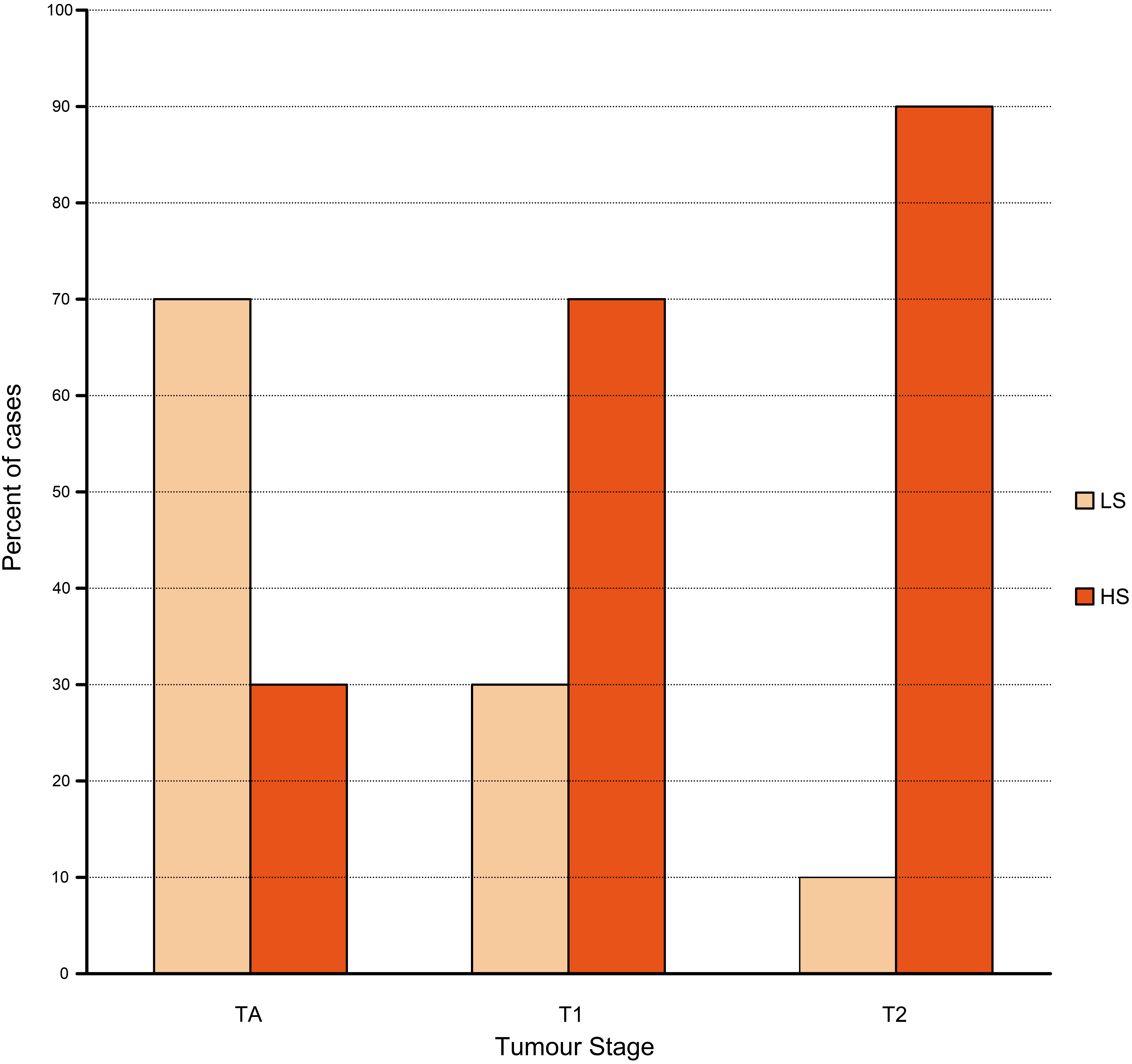
Immunohistochemical RBX-1 staining revealed the following; whereas RBX-1 expression in both nuclei and cytoplasm of the tumor cells of UCC tissue samples was significantly higher (Figs 2–4), the urothelium of non-tumoral bladder tissues exhibited weak, low and mainly cytoplasmic staining (Fig. 1). Among the 90 carcinoma tissue samples, 33 (36.7%) cases showed low expression and 57 (63.3%) cases showed high expression of RBX-1 protein.
Relation of RBX-1 expression and clinicopathologic parameters
Table demonstrates the relation between RBX-1 expression and clinicopathologic parameters. RBX-1 expression was associated with tumor grade (
Hence, following conclusions can be drawn: Compared to high grade tumors, low grade tumors exhibited lower RBX-1 statistically significant expression (
Discussion
In the present study, we aimed to determine RBX-1 expression in bladder UCC including NMIBC and MIBC and revealed that RBX-1 was overexpressed in bladder cancer tissues compared to normal urothelium. This finding is consistent to previous studies performed in bladder UCC and gastric cancer [6, 13]. We also found RBX-1 overexpression to be related with poor prognostic parameters such as high tumor grade and advanced tumor stage.
Bladder UCC is a tumor with variable behavior potential, ranging from a favourable outcome to aggressive clinical course [14]. Although NMIBC (Ta and T1 tumors) constitute a majority of bladder UCCs with a ratio of 70% [2, 14] they have a high risk of recurrence up to 80% [3, 14] and high capacity of converting into invasive and metastatic tumors [15]. According to EORTC risk evaluation of intermediate- and high-risk Ta and T1, tumors found in NMIBC recur frequently and progress to muscle-invasive disease [15]. Moreover, invasive bladder UCCs including T2 tumors may progress to lethal disease with metastasis in more than 50% of the patients [14]. Hence, early detection is essential to prevent tumor progression to lethal disease.
Alterations of various cellular processes have been suggested in bladder UCC. On molecular basis; noninvasive UCCs are characterized by activation of the Ras-MAPK pathway, while invasive bladder tumors represent alterations in p53 and retinoblastoma (RB) pathways [16, 17]. In high-risk MIBC, changes in P53, RB, and PTEN markers indicate poor prognosis [18]. Likewise, changes in proliferative markers such as MIB-1 and PCNA indicate a high grade and poor prognosis [19]. Recently, some investigations have been performed to determine novel biomarkers associated with tumor invasiveness. In their study comprising 2317 patients with bladder UCC, Ohlson et al. found tumor grade and invasiveness to be associated with fatty acid binding protein levels [20]. In another study, Choi et al. evaluated the association of CD 24 overexpression with bladder UCC invasiveness and suggested that CD 24 overexpression can be an indicator of stromal invasion and high tumor grade in bladder UCC [3]. Although there are studies focused on gene and cytogenetic marker expressions related with the predictability of UCC course, currently there is no noninvasive standard evaluation procedure for determining UCC prognosis and invasiveness.
A recent study has investigated the association of RBX-1 overexpression with prognosis of NIMBC [6]. RBX-1 was first cloned in 1999 by 4 independent groups via either biochemical purification or yeast 2-hybrid screening [7]. It is a molecule playing an important role in normal cellular activity as well as in various oncogene and tumor suppressive gene activities [7, 21]. The complex consisting of Skp1, Cullin, F-box protein (SCF), a Ring box protein, has a crucial activity in the ubiquitination of the cell cycle-related proteins or other proteins such as DNA replication, signal transduction, gene transcription and development. If RBX-1 protein is overexpressed, SCF recognizes and promotes p21 or p27 protein degradation leading to cell unregulated proliferation which induces a primary alteration in cancer cells [22]. The role of cell cycle control regulators including p21, p27 and Rb were revealed in both NMIBC and MIBC pathogenesis [17]. Wang et al. [11] performed a study to determine potential mechanisms related with RBX-1 inactivation in bladder UCC. In their study, they revealed that G2 arrest resulted from RBX-1 inactivation was associated with upregulation of p21 and p27, and also emphasized that cellular senescence induced by RBX-1 knockdown acts through p53/p21 pathway. As a conclusion; they suggested RBX-1 plays an important role in tumor progression and could be a potential anticancer agent in bladder UCC.
Coleman et al. defined in their review article on the diagnostic and prognostic markers p53, cytokeratin 20, E-cadherin, Ki67, and, more recently, CD44 as useful techniques indicating cell-cycle progression and gene mutation. In high-grade in situ and invasive UCCs, the mutataion and overexpression of all these markers were observed [23].
Immunohistochemical staining enables the documentation of RBX-1 expression. Whereas low RBX-1 expression is determined with immunohistochemical staining in many normal human tissues like lung, liver and breast, malignant tumors including colon, ovarian stomach, breast carcinomas and malignant melanoma exhibit increased RBX-1 expressions [7, 8, 10, 18]. Similary, in the present study, we determined higher RBX-1 expression in bladder UCC tissue compared to normal urothelium.
According to our knowledge, in the current literature, there is a single study evaluating association of RBX-1 overexpression with prognostic parameters in bladder UCC including pTa and pT1 tumors [6]. In it, low and cytoplasmic RBX-1 expression was evident in normal bladder mucosa, whereas RBX-1 overexpression was detected in both cell nucleus and cytoplasm in cancerous bladder tissue. Likewise, in the present study, in normal bladder epithelium both RBX-1 staining and cytoplasmic RBX-1 expression were low; whereas in UCC tissue samples, staining were intensive in both nucleus and cytoplasm indicating that RBX1-associated ubiquitination and disruption of nuclear proteins may conduce to initiation and progression of bladder cancer. The same study underlined that RBX-1 overexpression was related with higher tumor grade and stage, as well as, cases with RBX-1 overexpression exhibited poor prognosis and high recurrence rate. Our result indicating RBX-1 overexpression in high grade tumors is in line with the findings of Wang et al. study [6].
The unique aspect of the present study is to evaluate expression of RBX-1 in MIBC in addition to NMIBC. RBX1 overexpression in pT1 tumors compared with pTa tumors was also similar to the findings of the Wang et al. study. Moreover, the findings of the present study have shown that RBX-1 expression levels in MIBC (pT2) tumors were higher than pTa tumors alone and pTa & pT1 tumors (NMIBC) combined in total. In the light of these findings, we suggest that RBX-1 overexpression can be established for having a substantial role in tumor progression and can be an indicator of invasiveness and aggressive clinical behavior in bladder UCC.
Some of the considerable limitations of the present study are the following: the study was performed in a single center with patients who did not have uniform or randomized treatment or follow-up and examined RBX-1 expressions in only TURB specimens. In order to establish a causal relationship in order to present RBX-1 as a prognostic biomarker further prospective studies with larger patient series including radical cystectomy materials are to be conducted.
Conclusions
RBX-1 overexpression in bladder UCC is associated with high tumor grade and advanced stage and represents biological potential of invasiveness and aggressive disease. Further studies are needed to reveal the clinical and therapeutic implications of RBX-1 in bladder UCC.
Conflict of interest
The authors declare that they have no competing interests.
Footnotes
Acknowledgments
The authors thank Dr. Tahir Kemal Sahin for statistical evaluation of the study, and Dr. Enver Ahmet Demir and Dr. H. Serdar Gergerlioglu for their help in preparing graphs. This study was supported by grants from Selcuk University Scientific Investigations Projects Coordinatorship with the project number 14401053.
