Abstract
OBJECTIVE:
This study aims to evaluate the diagnostic value of ultrasound in thyroid Hürthle cell tumors.
METHODS:
A retrospective analysis was carried out on 27 patients with thyroid Hürthle cell tumors, in terms of the size, shape, boundary, echo, aspect ratio, cystic degeneration, calcification, peripheral halo sign and blood supply of the tumor, through surgical pathological validation. Then, these were compared with postoperative pathological results.
RESULTS:
The maximum diameter of the thyroid Hürthle cell tumors is between 0.6 cm and 4.6 cm. It had an oval nodule with clear boundaries, an aspect ratio
CONCLUSION:
Thyroid Hürthle cell tumors have nodules with even or uneven echoes on the background of the normal echoes of the thyroid, with an aspect ratio of
Introduction
Hürthle cells are also known as oxyphil cells. They are a kind of large and polygon cells that are rich in cytoplasm. An hematoxylin-eosin dyed cytoplasm is red eosinophilic and granular. These particles are mitochondria with abundant peroxidase in the cytoplasm under an electron microscope. Thereafter, researchers have found this type of cell in some thyroid neoplasms and non-tumor tissues such as thyroiditis and nodular goiter one after the other. Ewing [1] clearly defined the thyroid Hürthle cell neoplasm (HCN) in1928. HCN, which is a rare thyroid neoplasm, consists of more than 75% of Hürthle cells and a little colloid ingredient, approximately accounting for 4.5–10% of all types of thyroid neoplasm [2].
HCN is classified as a subtype of thyroid follicular cell tumors by the World Health Organization (WHO). However, its invasion is far stronger than follicular cell tumor. Furthermore, malignant HCNs can more easily metastasize into the surrounding tissues, blood vessels and lymph nodes, compared with other differentiated thyroid carcinomas. Once tumor metastasis occurs, treatment would be very difficult
Data and methods
General data
The data of 27 thyroid HCN patients who were examined and diagnosed by the Pathology Department of Peking University First Hospital Ultrasonic Diagnosis Center between January 2008 and March 2016 were collected. The age of these patients was between 27 and 69 years old, with a mean age of 40.8 years old. Among these patients, 20 patients were female and seven patients were male. Ultrasonography revealed that one patient had thyroid carcinoma and 18 patients had thyroid follicular cell tumors, and eight patients had goiter. Other concomitant thyroid space occupying lesions include six thyroid hyperplastic nodules, one papillary carcinoma, and one follicular cell tumor.
Ultrasonography
GE Logiq 9, SEMENS S2000, PHILIPS iu22, ALOKA F75 and ESAOTE Mylab90 ultrasonic devices are applied used, with a real-time linear array high frequency probe and at a frequency of 10–14 MHz. Using the thyroid conditions preset in the devices, and adjustments performed on the depth, gains and focusing locations according to lesion conditions to adjust the depth, gains and focusing locations, the images are adjusted to be its best, in order to observe the location, size, shape, aspect ratio, echo type, boundary, and peripheral low level echo halos, and to determine whether there are cystic changes, and calcification and via the color Doppler flow imaging (CDFI) of the thyroid tumor. The echo types include the low echo, medium echo and medium-low mixed echo.
Sonographic findings of 27 Hürthle cell neoplasm
Sonographic findings of 27 Hürthle cell neoplasm
Even low-echo Hürthle cell tumor.
HCN ultrasonic findings
There are no diffuse lesions in the thyroid parenchy-ma among the 27 patients. These nodules are located in the right lobe (10 nodules), left lobe (15 nodules) and isthmus (two nodules). The maximum diameter of the nodules was between 0.6 cm and 4.6 cm, with an average of 2.3 cm, among which eight nodules revealed even low echoes (Fig. 1), 13 nodules revealed even medium echoes (Fig. 2), six nodules revealed medium-low mixed echoes (Fig. 3D), and 12 nodules revealed internal cystic changes in varying degrees (Fig. 3A, B and C). Refer to Table 1 for detailed conditions.
Even medium-echo Hürthle cell tumor.
Ultrasonographic evolution process of mixed echo Hürthle cell neoplasm in four years follow-up. (A) Evolution process of Hürthle cell tumor from mixed nodule to solid nodule 2008.5; (B) Evolution process of Hürthle cell tumor from mixed nodule to solid nodule 2010.1; (C) Evolution process of Hürthle cell tumor from mixed nodule to solid nodule 2011.9; (D) Evolution process of Hürthle cell tumor from mixed nodule to solid nodule 2012.10.
There were 25 cases of Hürthle cell adenoma (HCA). The proliferation of fibrous tissues appeared in the tumor’s peripheral envelope. There are two cases of Hürthle cell carcinoma (HCC), in which one had a maximum diameter of
Ultrasound findings and pathological comparison of solid Hürthle cell carcinoma. (B) Solid medium-echo thyroid Hürthle cell carcinoma, surrounded by blood flow, rich blood flow inside. (C) Postoperative pathologic findings of thyroid Hürthle cell carcinoma. Tumor cells were partially invaded, liking a mushroom cloud into adjacent thyroid, X40, HE.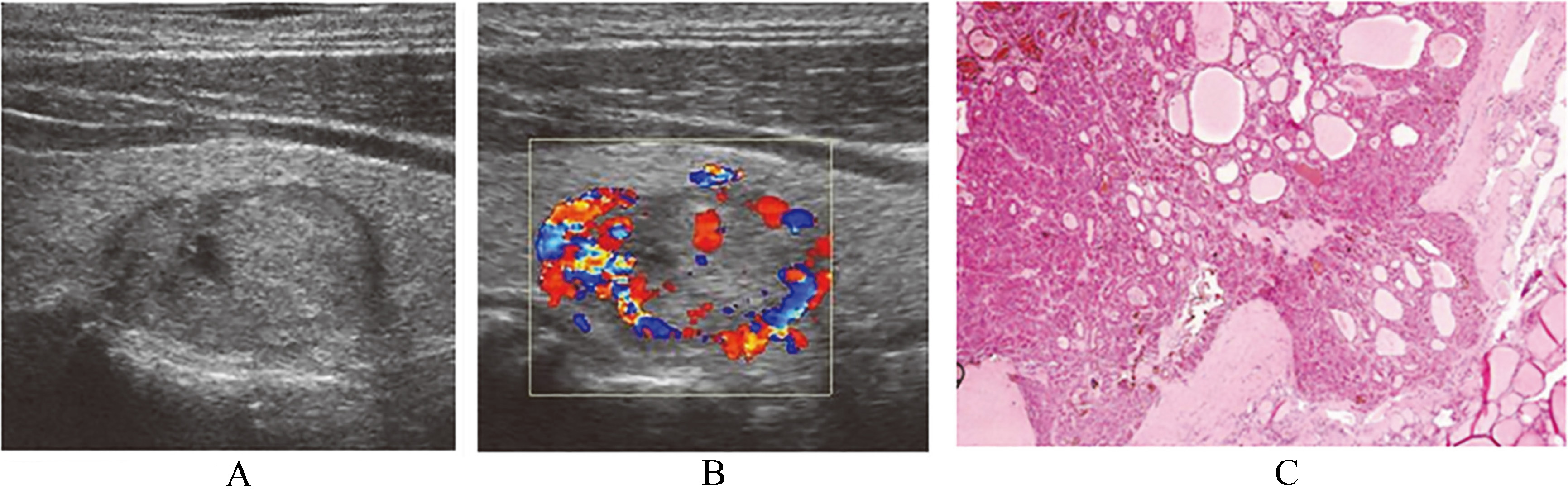
Ultrasound findings and pathological comparison of mixed Hürthle cell carcinoma. (A) Thyroid Hürthle cell carcinoma with mixed echo with cystic change, with strong echo inside, postoperative pathology confirmed as local fibrosis. (B) Thyroid Hürthle cell carcinoma with mixed echo with cystic change, surrounded by blood flow, rich blood flow inside. (C) Postoperative pathologic findings of thyroid Hürthle cell carcinoma, partial carcinoma invaded into capsule, X100, HE.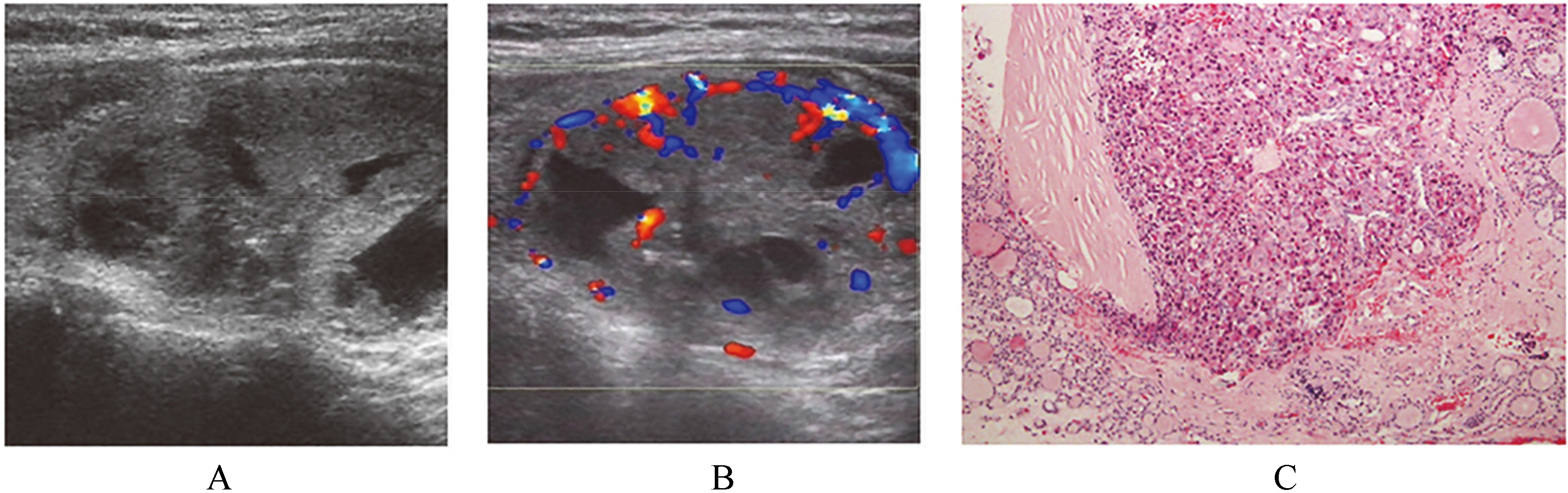
Ultrasound-guided fine needle aspiration (FNA) is the first choice [4, 5] in the preliminary confirmation of the nature of thyroid nodules. The introduction of The Bethesda System for Reporting Thyroid Cytopathology (TBSRTC) offered the opportunity to establish a uniform six-tiered reporting system for thyroid FNA. For the commonly encountered benign and malignant thyroid lesions in FNA specimens, the processing mode is clear. It can choose follow-up clinic or subtotal thyroidectomy. The “indeterminate for malignancy” has accounted for 36% of the total biopsy nodules [4]. For the purpose of assurance, most clinicians would choose to perform the complete surgical excision of these nodules. The excision extent is decided based on intra-operative pathological results. The third kind of nodule is mostly thyroid follicular cell tumors or a kind of more specific and rare tumor, namely, HCN. From the angle of biological behaviors, HCN has benign and malignant tumors that can be classified as HCA and HCC. The major evidence in diagnosing HCC is that Hürthle cells have invaded into the tumor envelope and (or) blood vessels and peripheral lymph nodes. Therefore, neither aspiration biopsy nor intra-operative frozen histology would be unable to estimate the nature of HCN. This nature can only be clarified when a complete excision of the tumor is performed and a pathological examination is conducted. Great disputes continue to exist in HCN classification and affiliation at present. Some scholars believe that HCN is a variant type of thyroid follicular cell tumor, while some scholars believe that HCN is a sort of independent tumor irrelevant to follicle tumors. In addition, some scholars believe that HCC should be classified into a subtype of papillary thyroid carcinoma [1, 2, 3]. HCN ultrasonogram shows the diversity. In this study, there are 27 cases of HCN, in which 29.6% had even lowecho nodules without cystic changes, and 48.1% and 22.2% of HCN had medium echo and medium-low mixed echo nodules, respectively. The incidence of cystic changes is 44.4%, which is similar to the incidence of HCN with 56.7% of cystic changes, as reported by Lee et al. [6]. The nodule’s internal hemorrhage and hyaline degeneration may be the pathological basis for the cystic changes of HCNs.
Lee et al. [6] reported that the incidence of calcification was 20% in HCNs. One patient with HCN was found with multiple “micro-calcification” in the thyroid cystic or solid nodules upon ultrasonic testing. This suggests that it is highly likely a thyroid malignant tumor (Fig. 5). The postoperative pathological examination for this patient shows that Hürthle cells invaded into the thyroid nodule envelope, and the patient is diagnosed with HCC. However, after comparing the ultrasonogram presentation and pathological results, the ultrasonography shows that the sign of “calcification” is not confirmed by pathological results. This “calcification” is actually internal colloid crystallization in the HCN, which forms an acoustic interface with great difference of acoustic impedance in the local nodule and causes a fake image similar to “calcification”, resulting in misjudgment. Therefore, this study concludes that nodular calcification is not a HCN feature. During diagnosis, care should be given in accurately forecasting malignant thyroid nodules that are to be likely “micro calcifications”. First, the calcification is mostly located in the solid lowecho zone of the thyroid nodule rather than in the junction of the cystic or solid structure or the cystic structure. These strong echoes are colloid crystallizations or fibrosis in this location. Second, gravel-like calcification is dotted rather than short and linear. Finally, because single gravel-like calcification has a small volume, it mostly shows a silent shadow or “comet tail” sign on the rear. These strong echoes may be Hürthle cell tumor specific.
A mixed echo nodule with medium and low echoes is the ultrasonogram performance of HCN, and forms a specific sign of “node-with-node”. Its formation mechanism has not been reported in documents. Figure 3 shows a procedure of the ultrasonogram of mixed echo HCN in four successive years of follow-up observation. It shows that HCN is mainly a cystic nodule at the earliest. Soon afterwards, cystic areas were gradually replaced by lowecho solid substances, finally forming a completely solid echo nodule with medium and low echoes. The reason that causes the HCN ultrasonogram evolution may be the stiff colloids that result from water that was gradually absorbed by epithelium in the HCN non-echo area and thin fluid area. This lowecho area is like a solid structure, but is actually thick sediment. If these liquid ingredients inside the nodules are not completely absorbed or the sound transparent property of colloids is good, it will form medium and low mixed echo nodules with partial cystic ingredients. There are three methods to identify this lowecho area by ultrasound: (1) in combination with medical history and attention to nodal evolution; (2) no space occupied effects in the lowecho area; (3) no blood flow in the lowecho area.
The halo sign means strip low echoes surrounded with thyroid nodules. The presence of a complete halo sign can reach 95% in estimating whether the thyroid nodules are benign or not [7]. These complete halo signs are surrounded in all HCNs of this group, in which 92.6% are benign and in accordance with results reported in literature. The pathological basis for the formation of HCN halo signs is the false capsules formed by HCN peripheral fibrillar connective tissues and chronic inflammation around the nodules caused by HCNs oppressing the peripheral thyroid parenchyma. The complete halo sign of peripheral HCN has reflected the HCN oppression to peripheral thyroid tissues rather than infiltration changes. Halo signs surround the partial papillary thyroid carcinoma, but are usually incomplete. Moreover, the tumor’s infiltration growth leads to uneven thickness in lowecho halos [8]. For HCN, regardless of benign or malignant tumors, peripheral halo signs are complete and even. HCN nature cannot be judged via the thickness of halo signs.
CDFI has some reminding effects in estimating the nature of thyroid nodules. HCN blood flow is rather regular and meets the features of benign thyroid tumors. Maizlin et al. [9] reported that 46.7% of HCNs were surrounded by blood flow and were rich with blood inside; and 40% of HCNs had blood flow inside. Lee et al. [6] reported that all HCNs were surrounded by blood flow and were rich with blood inside. Furthermore, 88.9% of HCNs revealed that they were peripherally surrounded blood flows, as well as blood flow inside in varying degrees in this group; and the rest revealed little blood flow inside. This speculates that the mechanism of forming blood flow is HCN rich in blood supply, and the tumor has not invaded the peripheral thyroid tissue. Therefore, these nodules are surrounded by blood flow. The performance is same to the “halo sign”, and indicates that the envelope structure is formed to surround the nodule. This performance is completely different from the apparent peripheral infiltration and non-surrounded blood flow in other types of thyroid malignant tumors. Similarly, the existence of blood flows is unable to act as an evidence to confirm HCNs as a benign tumor.
Lowecho HCN and common thyroid malignant tumors have a similar ultrasonogram feature, namely, “even and lowecho nodules without cystic changes”. This feature reflects the growth patterns of monoclonal proliferation in malignant tumor cells. It is an important standard of ultrasonography in deciding whether thyroid nodules are benign or malignant. Therefore, the lowecho expression of HCN cannot rashly exclude the possibility of malignancy at the first ultrasonic testing in this group, thereby requiring the performance of aspiration biopsy and an operation. However, the final pathology shows the benign results, and none of the cases are malignant. Even and lowecho expression may indicate a benign trend of HCN. The ultrasonogram features of lowecho HCN are different from other thyroid malignant tumors, which include: (1) regular and clear boundaries; (2) peripheral halos; (3) no peripheral burrs; (4) aspect ratio
Chen and Kushchayeva et al. reported that 5%–35% of patients underwent preoperative aspiration biopsy and were diagnosed with HCN, whose final operation pathology was HCC [10, 11]. The malignant rate of HCN is 7.4% in this group, which was in accordance with literature. Generally speaking, the ultrasonogram revealed the thyroid nodules with medium or medium-low mixed echoes, without regard to the diagnosis of thyroid malignant tumor. If these cystic changes have appeared inside the nodule, the possibility of a malignant tumor is even less. However, the ultrasonogram found out that HCN might be malignant tumor. Two cases of HCC have 2.1 cm of medium echoes and 4.6 cm of medium-low mixed echo nodules with cystic changes in this group. Both have benign features such as halo signs and peripherally surrounded blood flow (Fig. 4). The size of the nodules may be correlated to benign and malignant HCNs. Dahl et al. [12] reported 18 cases of HCC among the 26 patients with HCN with a diameter
Ultrasonography must be the primary choice at the diagnosis of many surgical diseases [13, 14]. HCN refers to nodules with even echoes and uneven echoes usually under a condition of normal thyroid. Its internal echoes vary. Aspect ratio was
