Abstract
BACKGROUND:
The present study aimed to illustrate the clinical value of contrast-enhanced computed tomography (CECT) in predicting gastric cancer (GC) recurrence and metastasis.
METHODS:
From March 2012 to March 2014, 131 GC patients who underwent initial treatment at the Tumor Hospital of Harbin Medical University were enrolled in this study, and 64-slice spiral CT was used for CECT. CECT parameters were recorded, including CT values in the arterial and venous phases, as well as the enhancement patterns in the arterial phase. All GC patients were followed for one year. The receiver operating characteristic (ROC) curve was used to evaluate the clinical values of the CECT parameters in predicting GC recurrence and metastasis.
RESULTS:
Significantly higher CT values in the arterial and venous phases were found in patients with higher tumor node metastasis (TNM) staging, lymph node metastasis (LNM), advanced Borrmann type and postoperative chemotherapy. TNM stage I-II patients showed less significant enhancement and significant heterogeneous enhancement than TNM stage III-IV patients. Compared with patients without recurrence and metastasis, patients with recurrence and metastasis demonstrated higher CT values in the arterial and venous phases, as well as a higher proportion of significant enhancement and significant heterogeneous enhancements. For the CT value in the arterial phase, the area under the ROC curve was 0.858, with 70.3% sensitivity and 95.7% specificity, and in the venous phase, it was 0.801, with 73.0% sensitivity and 83.0% specificity.
CONCLUSION:
Our study suggests that CECT has a high accuracy rate for predicting postoperative GC recurrence and metastasis.
Introduction
Gastric cancer (GC), also known as stomach cancer, develops in the lining of the stomach [1]. According to statistics, 952,000 new GC cases occurred in 2012, accounting for approximately 6.8% of all types of human cancer, making it the fifth most common malignancy worldwide; furthermore, half of all cases occur in East Asia, primarily in China [2]. GC is a multifactorial disease caused by the interaction of genetic and environmental factors, including diet, Helicobacter pylori, heredity, age and precancerous lesions [3]. Although GC treatment consists of adjuvant chemotherapy and/or chemoradiation and surgery, surgical resection of the tumor and lymph nodes remains the only curative therapy for localized GC [4, 5]. Nevertheless, over half of radically resected GC patients relapse locally or with distant metastases [5]. Clinical studies have shown that the survival rates and survival times of GC patients are closely associated with gastric wall invasion (T staging), lymph node metastasis (LNM) (N staging) and distant organ metastasis of gastric carcinomas (M staging) [6, 7]. Therefore, it is crucially important to preoperatively determine the invasion depth in the gastric wall and lymph node involvement. Currently, efforts have been directed toward improving GC diagnosis through double contrast radiography, endoscopy and biopsy, but the three major qualitative diagnostic methods are limited to observation of the mucosal surface [8, 9, 10]. With technical improvements in imaging techniques, computed tomography (CT) has shown good performance in the preoperative evaluation of GC staging.
A CT scan, also known as X-ray CT, uses computer-processed combinations of X-ray images taken from various angles to generate cross-sectional images of specific areas of a scanned object, allowing the user to identify the target inside the object without surgery [11]. Based on different scanning modes, CT scans are classified as follows: CT plain scan, enhanced scan, CT angiography and three-dimensional (3D) CT gastrography [12, 13]. Contrast-enhanced CT (CECT) increases the detection rate and diagnostic accuracy of lesions due to its better ability to differentiate the densities of lesions and adjacent normal tissues [14]. The merit of CECT is that either tumor extension or distant metastases might be evaluated in a single examination, and in detecting metastases, CECT is superior technique compared to plain CT [15]. It has been reported that CECT was useful in performing preoperative advanced GC staging [16], while few studies have systematically explored the relationship between the clinical features, recurrence and metastasis of GC and CECT. Therefore, this study aimed to further investigate the relationship between CECT and the clinical features, recurrence and metastasis of GC.
Materials and methods
Ethics statement
This study was approved by the Institutional Review Board of Harbin Medical University Cancer Hospital, and written informed consent was obtained from each eligible participant.
Subjects
From March 2012 to March 2014, 131 patients (96 males and 35 females, ranging in age from 26 to 80 years [mean age, 57.81
CECT
The 64-slice multidetector-row CT (Toshiba Medical Systems, Crawley, UK) was used for the CECT sans, with a 0.8 CT interval. Before undergoing CECT, the patients routinely fasted for 4
Association between the clinical features of GC patients and their pre-surgery CECT values
Association between the clinical features of GC patients and their pre-surgery CECT values
Note: GC, gastric cancer; HU, Hounsfield Units; CECT, contrast-enhanced computed tomography; TNM, tumor node metastasis;
The CECT images were analyzed by 2 (blinded) experienced doctors from either the radiology department or the nuclear medicine department, and they based their diagnoses on these images. When the diagnoses conflicted, the doctors conferred further to reach a final conclusion. The slices with the highest uptake of fluorodeoxyglucose (FDG) in focus were selected, and the region of interest (ROI) was used to delineate the focus contour. The ROI included the whole focus but avoided the focuses with hypermetabolism. The maximized standardized uptake value (SUVmax) was calculated using total body weight (TBW). The central slices of focus in the CT plain scan image and image of the stomach phase were found, and the ROI was drawn in the same region with same shape and size. The ROI included solid parts of focuses and parts with the most enhancements and avoided the cavity and necrosis. CT enhancement value
Association of TNM staging with enhancement patterns in the arterial phase of GC patients
Association of TNM staging with enhancement patterns in the arterial phase of GC patients
Note: TNM, tumor node metastasis; GC, gastric cancer.
All participating GC patients were followed-up for one year. After surgery, the patients were followed-up monthly. The postoperative recurrence and metastasis of GC were pathologically confirmed through surgery or gastroscopy and, eventually, recorded.
Statistical analysis
SPSS software (version 19.0, SPSS Inc., Chicago, IL, USA) was used for the statistical analysis. The measurement data are expressed as the mean
Results
Relationships between the clinical featuresof GC patients and CECT values before surgery
The correlation between the clinical features of GC patients and the values of pre-surgical CECT is shown in Table 1. The CT values in the arterial and venous phases showed no correlation with age, gender, smoking or alcohol consumption (all
Relationships between TNM staging and enhancement patterns in GC patients in the arterial phase
Based on the CT images of GC patients, the enhancement patterns in the arterial phase were categorized as slight enhancement (
Association between recurrence and metastasis and pre-surgical CECT values and certain clinical features in GC patients
The 131 patients were followed-up postoperatively for one year, during which 37 patients demonstrated recurrence and metastasis. As shown in Table 3, compared with patients without recurrence and metastasis, those with recurrence and metastasis showed higher CT values in the arterial and venous phases, as well as a higher proportion of significant enhancements and significant heterogeneous enhancements (all
Association between recurrence and metastasis and pre-surgery CECT values and certain clinical features in GC patients
Association between recurrence and metastasis and pre-surgery CECT values and certain clinical features in GC patients
Note: HU, Hounsfield Units; CECT, contrast-enhanced computed tomography; TNM, tumor node metastasis; GC, gastric cancer.
The ROC curves were used to evaluate the sensitivities and specificities of the CT values in the arterial and venous phases in predicting GC recurrence and metastasis, and the results are shown in Fig. 1. Figure 1A shows the ROC curve of the predictive value of the CT value in the arterial phase, and the area under the curve (AUC) was 0.858, with 70.3% sensitivity and 95.7% specificity. As for the ROC curve of the predictive value of the CT value in the venous phase (Fig. 1B), the AUC was 0.801, with the sensitivity and specificity being 73.0% and 83.0%, respectively.
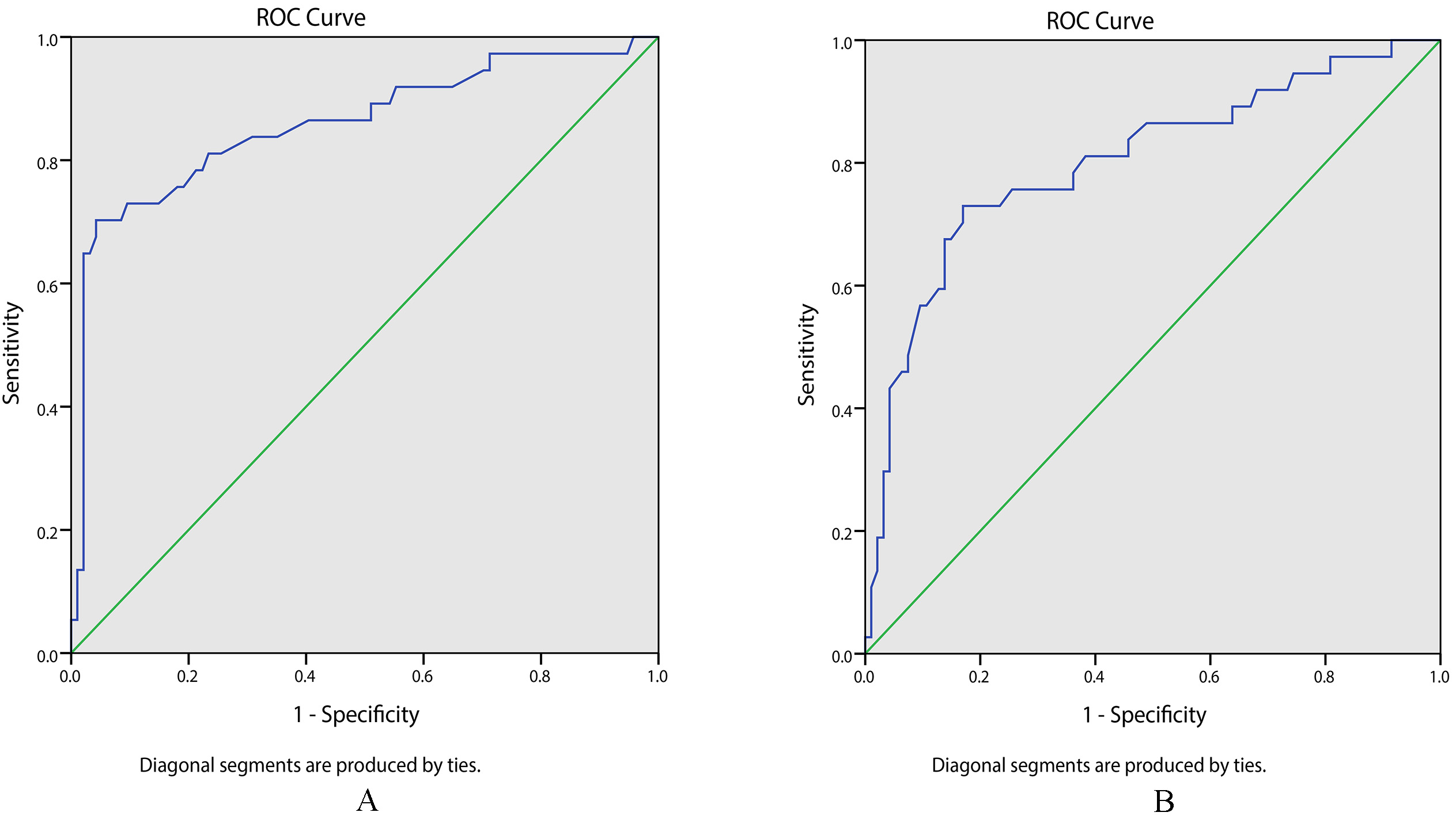
The ROC curves of CT values in predicting GC recurrence and metastasis. Note: A, the ROC curve of the arterial phase CT value in predicting GC recurrence and metastasis; B, the ROC curve of the venous phase CT value in predicting GC recurrence and metastasis; ROC, receiver operating characteristic; CT, computed tomography; GC, gastric cancer.
In this study, the preoperative data concerning CECT parameters and clinical features, as well as the recurrence and metastasis of 131 GC patients, were retrospectively analyzed to explore the association between CECT and recurrence and metastasis in GC patients. The results indicated that in GC patients, CECT showed higher accuracy in determining TNM staging and played an important role in predicting postoperative recurrence and metastasis.
GC is a heterogeneous and multifactorial disease, the incidence and mortality of which vary geographically, with the highest rates in East Asia [17]. However, among different therapeutic methods, surgery has been considered as the only curative treatment for GC [5]. To identify the most adequate therapy, it is necessary to accurately determine preoperative staging using CT, the standard imaging modality for preoperative staging [16]. CECT has become the preferred method for GC staging due to its high spatial resolution, tissue resolution and 3D reconstruction techniques [6]. In the present study, the clinical features of GC patients were studied, and it was found that the CT values in the arterial and venous phases were significantly and positively correlated to higher TNM staging, LNM, advanced Borrmann type and postoperative chemotherapy. Confirming these results, Ba-Ssalamah et al. have reported that textural gray-level patterns on CECT images, which reflect tumor heterogeneity, may indirectly capture the cell morphology [18], and morphology showed a close relationship to lymph node involvement [19]. Similarly, a previous study has also reported that CECT showed better diagnostic performance for primary tumors and regional LNM, as well as peritoneal seeding, exhibiting high diagnostic value for preoperative staging of advanced GC [16]. It has been reported that CECT provided useful information, such as gastrointestinal neuroendocrine carcinoma locations, density, enhancement pattern, and some metastases, further increasing the diagnostic accuracy [20]. The maximum HU value represents the highest degree of tumor enhancement; a published result has shown that the parameter was significantly higher in patients with poor prognoses, which may reflect the higher vascularity that characterizes more aggressive tumors [21].
However, it has also been found that patients with TNM stage I-II showed less significant enhancement and significant heterogeneous enhancement than those with TNM stage III or IV, which implied a high accuracy for CECT in determining TNM staging. TNM staging includes local tumor depth invasion (T), regional lymph node invasion (N), and distant metastases (M) [22]. Similar to most solid tumors, GC requires neovascularization to supply additional nutrients for growing metabolic demands and to provide potential routes for tumor dissemination and metastasis. Angiogenesis develops as the GC continues to develop, thus, the peak enhancement increases correspondingly [23]. Thus, it is reasonable that high TNM staging is closely related to increased neovascularization and significant enhancement. A recent study has demonstrated that enhanced CT imaging showed higher accuracy in predicting T3 and T4 stages of GC preoperatively [6]. Hallinan et al. have reported that texture analysis for GC could represent an additional, prognostic indicator over conventional methods, such as tumor volume, which is known to be correlated with TNM stage and prognosis [24]. In addition, Lee et al. have noted that contrast-enhanced helical CT could also be successfully used to preoperatively evaluate the staging of remnant GC in patients who have undergone gastric resection [25].
In this study, the diagnostic significance of the arterial phase CT value and the venous phase CT value in predicting GC recurrence and metastasis was confirmed due to their high accuracy rates; in addition, the AUCs of the corresponding ROC curves for the arterial and venous phases were 0.858 and 0.801, respectively. In terms of GC recurrence and metastasis, D2-40 positive lymphatic vessel density (LVD) and CD34 positive microvessel density (MVD) were two valuable parameters used to detect the invasion and metastasis of tumor cells in lymphatic and blood vessels [26]. Because the CECT values were shown to positively correlate with MVD, an increased CT value indicated increased angiogenesis, as well as a higher possibility of tumor invasion and metastasis [27, 28]. Regarding GC, a published study has proven the accuracy of multidetector-row CT in diagnosing lymph node metastasis [29].
Conclusion
In conclusion, our study demonstrates that CECT is highly accurate in determining TNM staging for GC patients and that it plays an important role in predicting postoperative recurrence and metastasis. Nevertheless, this study only investigated the diagnostic value of CECT in GC patients with early recurrence and metastasis. Further studies are needed to evaluate the clinical value of CECT in predicting late recurrence and metastasis.
Acknowledgments
The authors thank everyone who assisted with editing this paper.
Conflicts of interest
The authors have declared no conflicts of interest regarding the publication of this paper.
