Abstract
BACKGROUND:
LAMP2 is one of the major protein components of lysosome. In addition to the expression on the lysosomal membrane, LAMP2 has also been found relocalizing to the cell surface of some highly metastatic tumor cells.
OBJECTIVE:
The aim of this study was to detect the expression levels of LAMP2 and discuss its roles in esophageal squamous cell carcinoma (ESCC).
METHODS:
Six hundred and ten tissue samples of ESCC were collected to construct tissue microarrays, which were stained by immunohistochemistry.
RESULTS:
After immunohistochemical staining, 596 patients including 460 men and 136 women were analyzed. The LAMP2 expression levels were significantly different based on degrees of histological differentiation (
CONCLUSIONS:
Our results demonstrated that LAMP2 expression levels correlated with tumor histological differentiation and TNM stages. High expression of LAMP2 predicts poor prognosis in patients with ESCC.
Introduction
Esophageal squamous cell carcinoma (ESCC), the fourth most common cause of cancer-related deaths, is one of the most common cancers, especially in China [1, 2]. The fiveyear survival rate of patients with advanced ESCC is still very low [3, 4, 5]. Less than 30% ESCC patients could achieve early diagnosis and treatment. To improve clinical outcome of ESCC, novel molecular biomarkers used for early diagnosis and predicting prognosis have been widely investigated [6, 7].
Lysosomal associated membrane proteins (LAMPs) are major protein components of the lysosome playing an important role in the lysosome-mediated physiological processes [8, 9, 10]. The heavy glycosylation of the LAMPs creates a sugar coat or glycocalyx on the inner side of the lysosomal membrane, which protects the membrane from the hydrolytic enzymes and degradation [11, 12]. Despite having 37% amino acid sequence homology, LAMP1 and LAMP2 are distinct proteins for their separate chromosome localizations. Mice deficient in both of them are embryonic lethal, but viable and fertile upon loss of any one of them, suggesting that LAMPs are, to some degree, functionally redundant in vivo. However, LAMP2 seems to have some specific functions, because LAMP2 single deficiency has more severe consequences than LAMP1loss [13].
In addition to the expression on the lysosomal membrane, LAMP2 has also been found relocalizing to the cell surface of some highly metastatic tumor cells, such as melanoma and colon cancer [14, 15], playing an important role in cell-cell adhesion and cancer cell migration. These observations indicate that LAMP2 may be important for the metastasis of malignant tumors.
In this study, we detected the LAMP2 expression in tissue microarrays (TMAs) [16, 17, 18] by immunohistochemistry (IHC) to analyze the correlation between the LAMP2 expression levels and clinicopathological characteristics of ESCC patients, and evaluated the possibility of LAMP2 as a biomarker for ESCC prognosis.
Materials and methods
ESCC patients and samples
A total of 610 patients with ESCC were enrolled in this study from thoracic surgery department of the Affiliated Hospital of Jining Medical University from 2008 to 2014. All patients did not receive radiotherapy or chemotherapy before surgical resection. Informed consents were obtained from all enrolled patients. The clinical stages were defined according to the 7th edition of TNM classification of the International Union Against Cancer (2010) [19]. Patients’ clinical information was collected from medical records, and follow-up data were collected by telephone interview. This study was reviewed and approved by the Medical Ethics Committee of the Affiliated Hospital of Jining Medical University.
LAMP2 Expression and Clinicopathological Characteristics in ESCC
LAMP2 Expression and Clinicopathological Characteristics in ESCC
Paraffin-embedded tissue blocks of the ESCC patients were used to construct TMAs. The pathologist microscopically examined hematoxylin and eosin-stained sections and selected representative areas, excluding tumor necrosis. Cores (0.6 mm) of malignant tissues were spotted in duplicates. We constructed 9 TMA blocks containing a total of 610 tissue samples of ESCC. There were up to 70 patient tissue samples on each slice. 5-micrometer sections were cut from completed array blocks and transferred to adhesive slides. Then, the sections were stained with haematoxylin and assessed for adequate tumor representation.
Besides, 60 patients’ paraffin-embedded tissue blo-cks were randomly selected to making routine pathological sections for immunohistochemical staining, which were used to examine the consistency between TMAs and conventional tissue slices.
Immunohistochemistry (IHC)
TMAs were stained using the rabbit anti-LAMP2 monoclonal antibody (1:50, Proteintech, 10397-1-AP), and the microwave was used to perform the antigen retrieval. The immunostainings, performed according to the manufacturer’s instructions, were completed by the technical staff of the Department of Pathology. Then, the TMAs were counterstained with haematoxylin. LAMP2 antibody omission was used as a negative control. Finally, the TMAs were scanned with CaseViewer 1.4.
The immunohistochemical scores were based on the percentage of LAMP2 positive cells and their staining intensity [20]. Regarding the staining intensity, the score “0” corresponds to no staining, “1” to faint staining, “2” to moderate staining and “3” to intense immunostaining. The necrotic areas were not included. Moreover, the invasion zones were not included. Regarding the percentage of positive tumor cells, the score “0” corresponds to 0–2% positive cells, “1” to 2–25% positive cells, “2” to 25–50% positive cells, “3” to 50–75% positive cells and “4” to 75–100% positive cells. Then intensity and percentage scores were multiplied to conclude a total score (from 0 to 12). The patients were divided into four groups according to the total scores. “
Statistical analysis
Statistical analysis between groups was performed using Pearson chi-squared (
Results
Clinical data
All 610 patients were diagnosed as ulcerative squamous cell carcinoma according to the pathological results. Lymphatic metastasis was present in 281 cases, but no distant metastasis was found. After immunostainings, 14 patients’ tissues were failed to stain due to the tissues removal. The remaining 596 patients were comprised of 460 men and 136 women, ageing from 34 to 83 years (mean age: 60.9 years). The clinicopathological characteristics and the IHC results were listed in Table 1.
Expression Level of LAMP2 in ESCC
In ESCC tumor cells, the positive immunostaining was mainly located at the cytoplasm (Figs 1, D&E). LAMP2 expression levels in ESCC tissues were significantly different among the different subgroups. The LAMP2 expression levels were significantly different based on degrees of tumor histological differentiation (
The correlations between LAMP2 expression levels and clinical data were analyzed by spearman correlation coefficient. LAMP2 expression levels were negatively correlated with degrees of tumor histological differentiation (Fig. 2A,
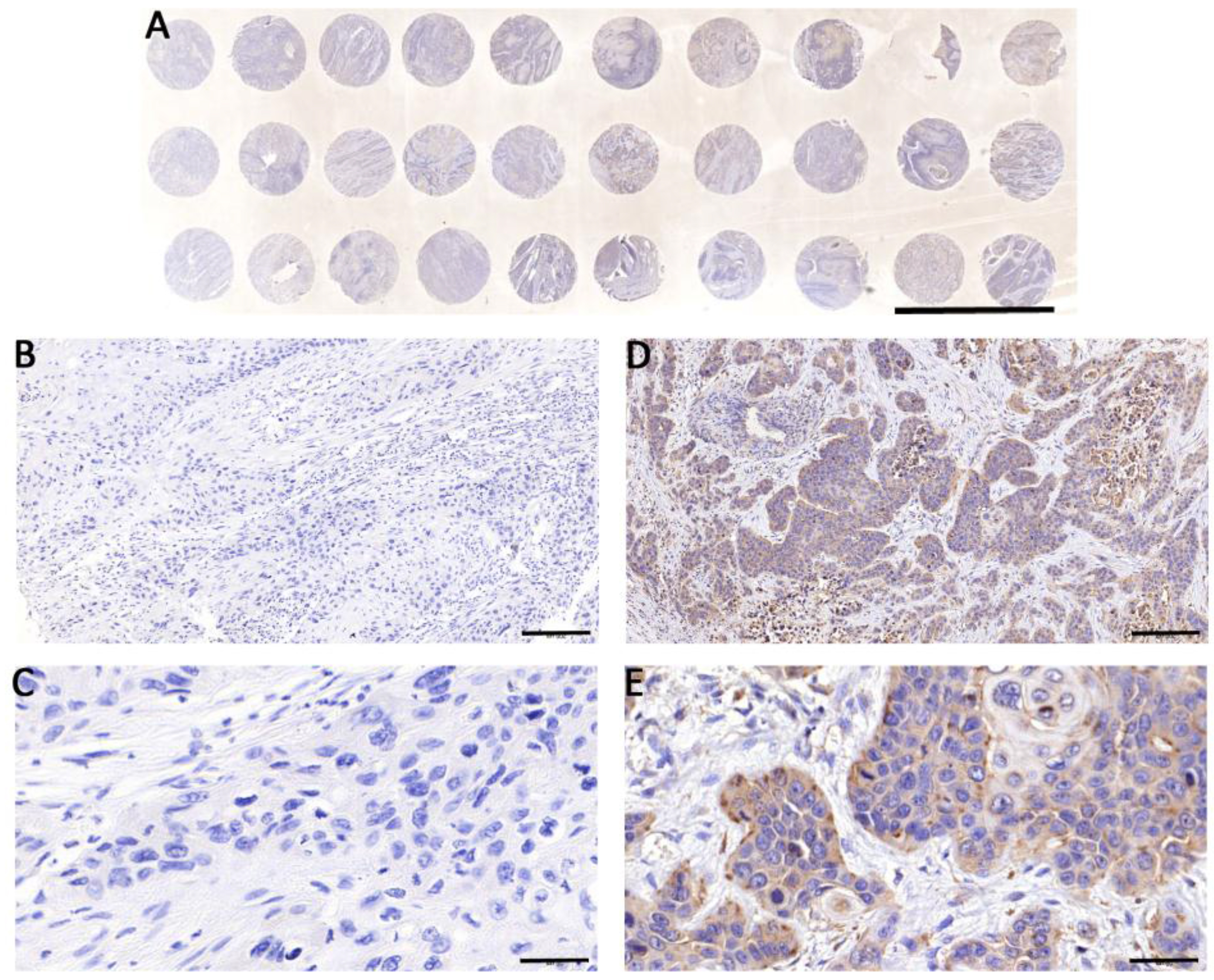
Immunohistochemistry Stainning of TMAs of the ESCC Patients. A: TMAs of the ESCC patients. B: Negative case of immunohistochemical staining (10
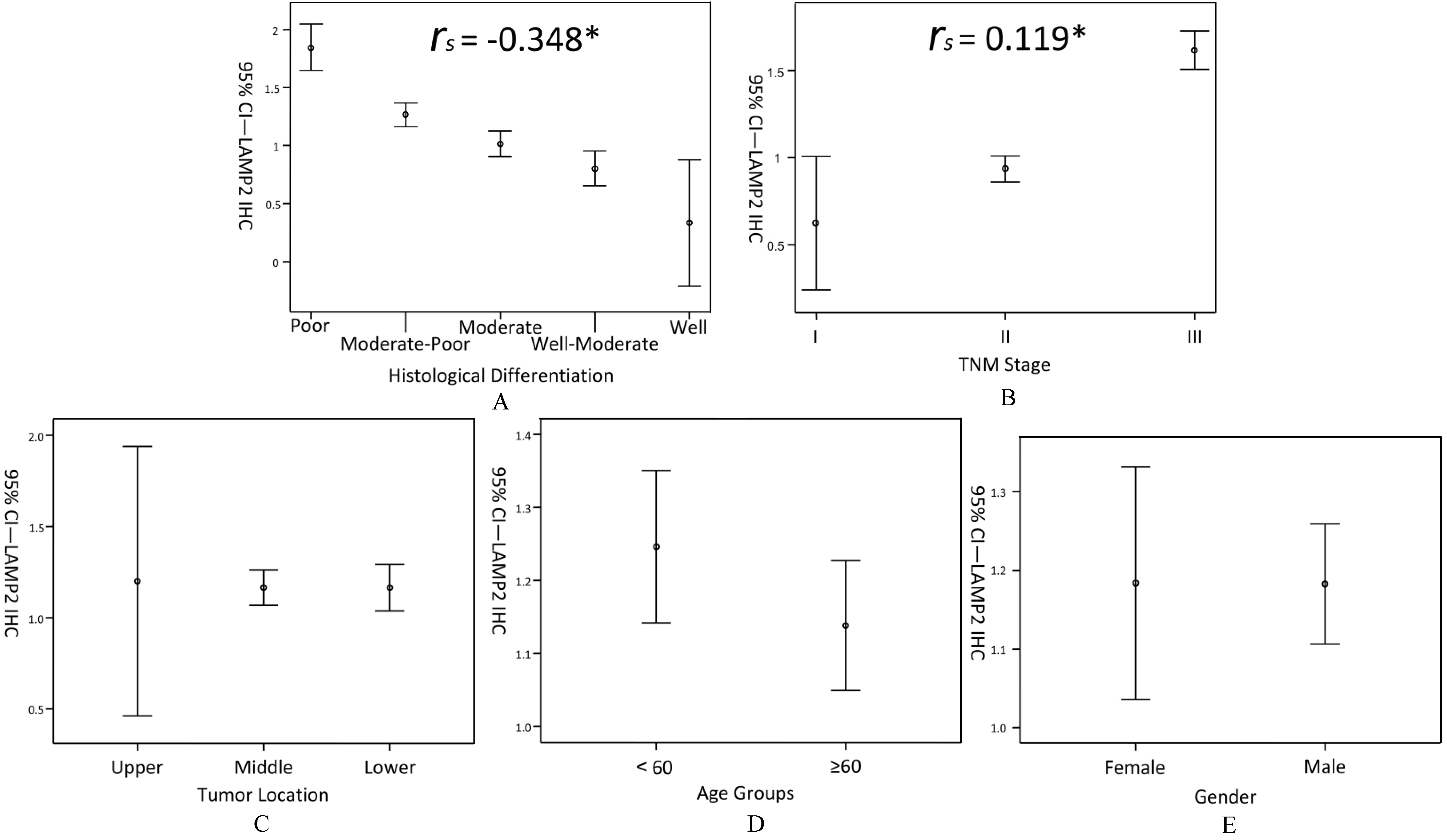
Expression Level of LAMP2 in ESCC Tissues. Samples were divided into different subgroups for statistical analysis. Expression levels (95% CI) were correlated with degrees of tumor histological differentiation (A), TNM stages (B), but not with tumor location (C), age (D) and gender (E). *,
Logistic regression analysis between LAMP2 expression and clinicopathological characteristics was performed. In this analysis, the positive degrees of LAMP2 were redefined. “
Kaplan-Meiersurvival curves, used to analyze the correlations between LAMP2 expression levels and patients’ prognosis, indicated that patients with a higher expression of LAMP2 showed worse prognosis (Fig. 3F,
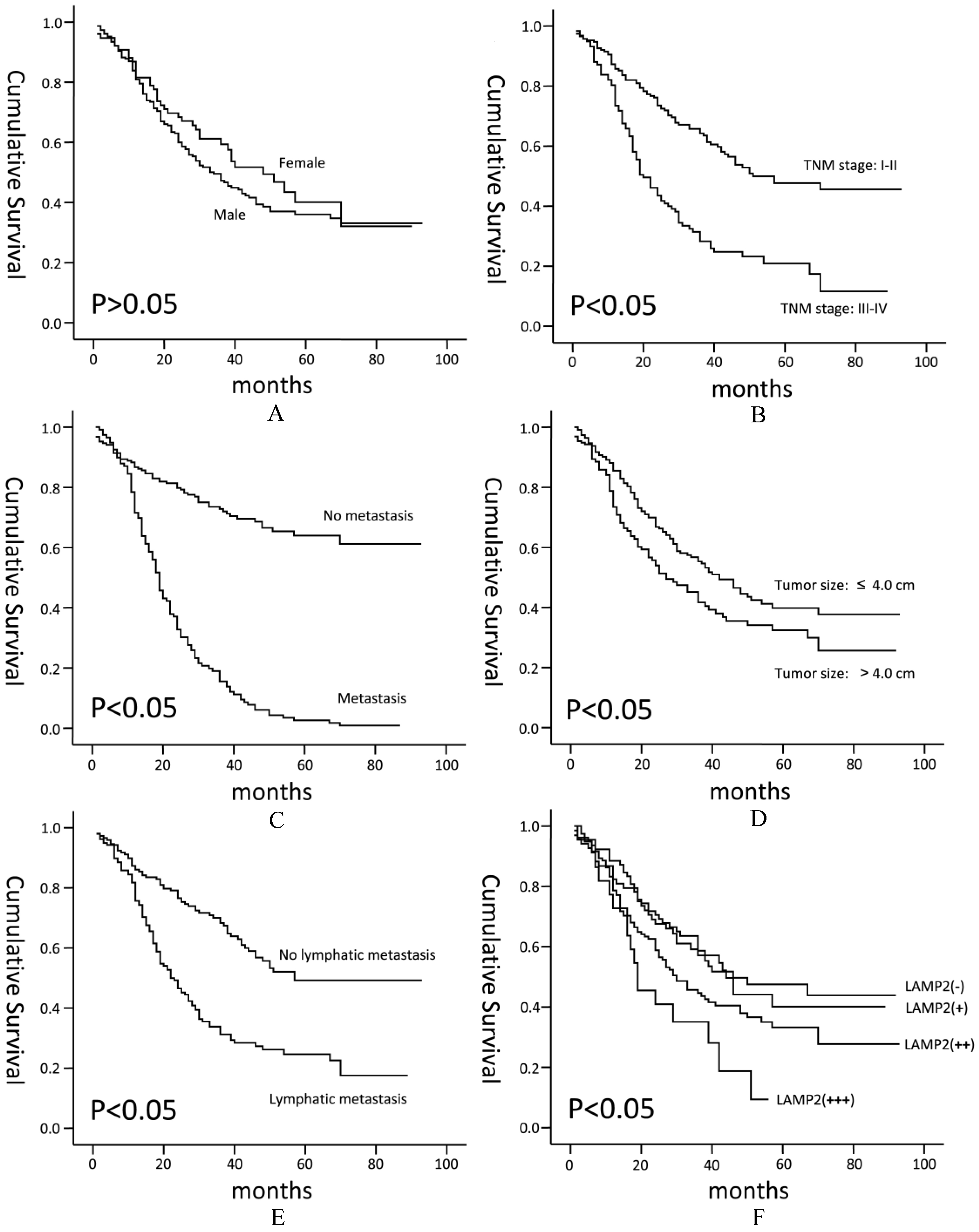
Kaplan-Meier Survival Curves for ESCC Patients. Kaplan-Meier survival curves for ESCC patients between different subgroups were made. Patients with a higher expression of LAMP2 (F) showed worse prognosis (
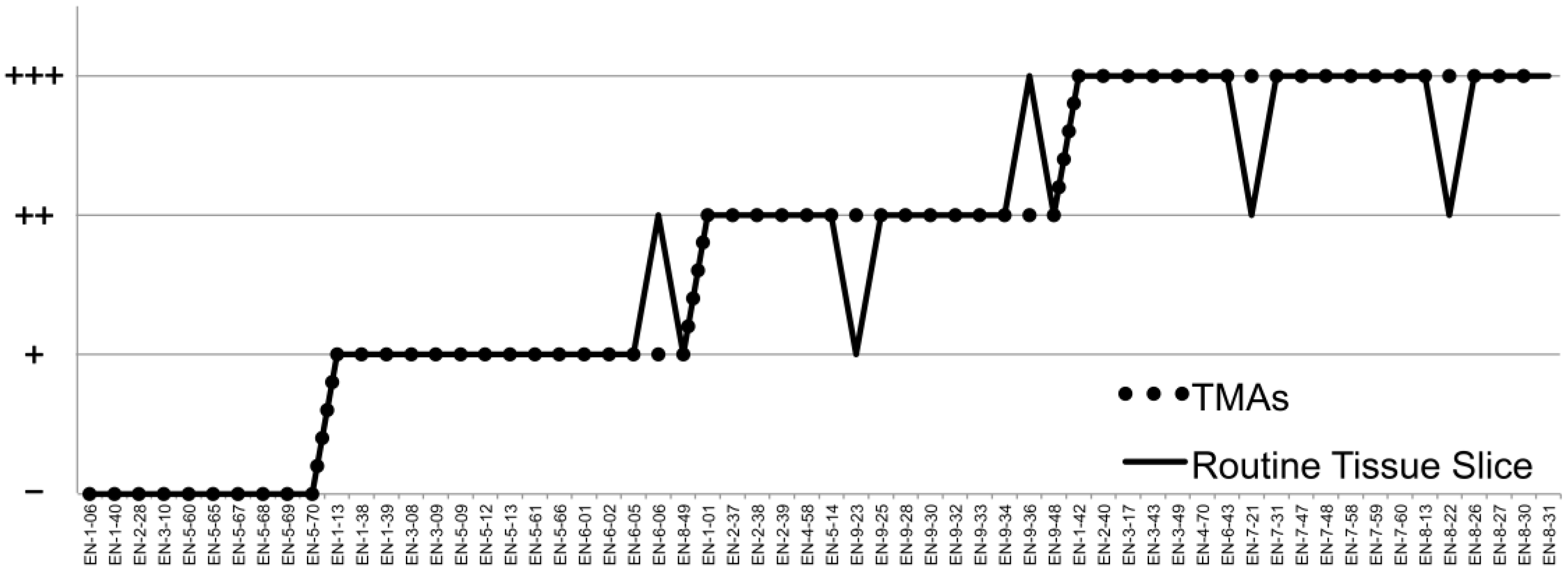
Comparison of Consistency between TMAs and Routine Tissue Slices. 60 patients’ cancer tissues were randomly selected to making routine tissue slices for immunohistochemical staining to examine the consistency between TMAs and conventional slices. Results of 55 cases were consistent with TMAs.
In the present study, the results showed that the LAMP2 expression levels were significantly different among different TNM stages and degrees of tumor histological differentiation, and that LAMP2 expression levels were negatively correlated with degrees of tumor histological differentiation, but positively with TNM stages (Table 1, Figs 2A & B). The results of correlation analysis and survival curves were consistent with the previous studies which showed that LAMP2 was highly expressed in many cancers, especially in the metastatic cancers [14, 15, 23, 24]. LAMP2 plays an important role in the regulation of autophagy which is involved in cancer cell metabolism [25]. Therefore, the results of this study may imply the enhanced autophagy activity correlated with LAMP2 expression in tumor cells. However, the detailed molecular mechanism needs to be further determined.
LAMP2 expression levels were negatively correlated with degrees of histological differentiation in our study. The worse the degree of histological differentiation, the higher the level of LAMP2 expression. The relationships between LAMP2 and cell differentiation have been reported [26, 27]. This might be related to the glycosylation of LAMP2 [28]. The weak positive correlation between the expression of LAMP2 and TNM stages observed in this study might be associated with the enhancement of autophagy activity in tumor cells [29], because LAMP2 was located on surface of lysosome, the final destination of the autophagy, which involves importantly in cancer cell metabolism. We would continue to study the roles of LAMP2 in ESCC in the following research.
Additionally, we conducted a comparison of LAMP2 expression levels between different genders in consideration of the location of LAMP2 gene on the X chromosome (Xq24) and the gender differences in the incidence of esophageal cancer [1, 2]. According to our results, there was no difference in the expression of LAMP2 between genders (Table 1,
TMAs, arranging many different individual specimens at regular arrays on the same slides [18], were characterized by large scale, high throughput, standardization. The biggest advantage was that all samples were tested under identical conditions, and that the experimental errors were reduced greatly. In this study, we used TMAs to detect the expression levels of LAMP2 in order to reduce the artificial operation errors among samples. But the other problem was that, compared with the traditional tissue sections, the detected area of each sample was greatly reduced, increasing the impact of tumor heterogeneity [30, 31]. With this in mind, we compared the consistency of TMAs and conventional pathological tissue slices. The ratio of consistency was 91.7% (55 of 60, Fig. 4).
Besides, there were other defects in the present study. Firstly, adjacent carcinoma tissues were not involved in this study. The differences between tumor and adjacent tissues could not be analyzed. Secondly, the sample size of some subgroups was small (N
In conclusion, our results demonstrated that LAMP2 might be involved in the histological differentiation, progression and prognosis of ESCC. The molecular mechanism would be discussed in the following research.
Footnotes
Acknowledgments
This study was supported by the following science foundations: Science and Technology Development Project of Jining Science and Technology Bureau (2015-57-35), Scientific Research Project of Jining Medical University (JY2015KJ023), and the Key Project of Scientific Research Program of Affiliated Hospital of Jining Medical University (MP-2015-002), Shandong, People’s Republic of China. All the authors have read the journal’s authorship agreement and policy on disclosure of potential conflicts of interest.
Conflict of interest
The authors declare no conflict of interest.
