Abstract
The SerpinE2 pathway is evolutionarily conserved and plays an important role in tumorigenesis. SerpinE2 (a small ubiquitin-related modifier), like ubiquitin, conjugates SerpinE2 proteins onto lysine residues of target proteins. SerpinE2 over-expression has been found in several tumors. Here, we detected the level of SerpinE2 in 72 samples of EC tissue using immunohistochemistry to assess the role of SerpinE2 in EC prognosis. Meanwhile, we knocked down SerpinE2 by siRNA in the HTB-111 and Ishikawa EC cell lines and analyzed the viability and mobility change using an MTT assay, an annexin V/PI apoptosis assay, a wound scratch test and a transwell assay. A Kaplan-Meier analysis indicated a negative correlation between the level of SerpinE2 and the EC prognosis. Silencing SerpinE2 induced cell apoptosis and reduced the migration ability. Our data suggest SerpinE2 works as an oncogene in EC.
Keywords
Introduction
Endometrial cancer (EC) is one of the most common malignancies among females in both developed and developing countries. With the increase in obesity, improper application of hormone replacement therapy and aging populations, the incidence of EC has risen [1]. A noticeable symptom, vaginal bleeding, presents at an early stage in most EC patients, drawing their attention and generally resulting in a favorable prognosis. Although extremely rare, some patients present with primary bone pathology without vaginal bleeding [2]. In addition, up to 30% of EC patients were high-risk at the first visit, with the EC invading deep into the myometrium and showing local or extra pelvic metastasis [3]. For them, the cancer progresses more quickly and is resistant to contemporary therapy, and the prognosis is poor, with a five-year survival rate less than 50% [4]. Currently, selecting the optimized scheme for EC patients only depends on the surgical tumor stage (pTNM-system). For monitoring the response to therapy and detecting recurrent EC, CA125 and serum epididymis protein 4 (HE4) have been used as routine useful indices, and TP53, PTEN and PIK3CA might not be sufficient [5]. New predictive and prognostic biomarkers are urgently required.
Serine protease inhibitor E2 (SerpinE2), named glia-derived nexin or protease nexin-1, the phylogenetically closest relative of PAI-1, was first found to be secreted by fibroblasts and combined with proteases, such as thrombin, urokinase, and plasmin, to form complexes with certain serines [6]. Recently, SerpinE2 has attracted the attention of researchers because it targets the extracellular matrix and promotes the metastasis of malignant cells. SerpinE2 over-expression has been found to be a potential novel biomarker in many kinds of tumors [7]. For EC, the role of SerpinE2 has not been reported. Here, we measured the SerpinE2 level in 72 cases of EC using IH and analyzed its clinical significance. Moreover, we also knocked it down in an EC cell line to further verify its function.
Materials and methods
Patients and tissue samples
This study was approved by the Research Ethics Committee of the First Affiliated Hospital of Jinan University and the Nanfang Hospital. Written informed consent was obtained from all of the patients. Paraffin sections were obtained from 72 patients who were diagnosed with EC and enrolled at the department of Gynecology and Obstetrics Department of First Affiliated Hospital of Jinan and Nanfang Hospital the University between 2006 and 2010. The diagnosis of EC was confirmed based on FIGO guidelines (2009) and the 2008 WHO classification criteria. All of these patients underwent molecular and phenotypic classification and were distinguished as Types I (mainly endometrioid) and II (non-endometrioid). The follow-up was performed at 60 months by mail or phone.
Immunohistochemistry
IH was performed on EC paraffin sections using an Enhanced Polymer EliVision DAB Detection Kit (purchased from Maixin Biocompany, Fuzhou, China). Briefly, the paraffin-embedded tissue slides were heated, deparaffinized, and dehydrated, and then non-specific action was blocked by H
Cell culture and siRNA synthesis
HTB-111 and Ishikawa cells were maintained in DMEM medium (Gibco, Carlsbad, CA, USA) containing 10% heat-inactivated fetal bovine serum albumin (Logan, USA) in a 95% humidified incubator with 5% CO
Cell viability assay
The cells were seeded into 96-well culture plates at a density of 1.5
Cell apoptosis assay
The cells were seeded into 6-well culture plates. The transfer process was almost performed as described above, except the amount of Lipofectamine 3000 reagent differed according to the protocol (different wells were given different amounts of DNA and transfer reagent). After transfection for 24 h, both suspended and adherent cells were collect and labeled with FITC-labelled annexin V and propidium iodide (PI) (BD Bioscience, USA). A FACScan instrument (Becton Dickinson, USA) was used to count the cells (1
Wound-healing assay
The scratch wound assay was performed to detect the mobility of EC cells. HTB-111 and Ishikawa cells were seeded in six-well plates overnight; then, the cells were transfected with the siRNA-SerpinE2 or the NC control for 6 h. The cells were incubated in fresh DMEM complete medium overnight. Then, the confluent cell monolayer was scraped using a 200
Migration assay
After cells were transfected with siRNA-SerpinE2 for 6 h, they were digested with 0.25% trypsin and then seeded into the upper layer of a Boyden chamber (Haimen, Jiangsu province, China). After incubating for 8 h, non-migratory cells were removed from the top well. The cells were fixed with 4% paraformaldehyde and stained with crystal violet. Photo images of the plates were photographed.
Matrix metalloprotein (MMP) activity ELISA assay
Using a total MMP-2 and MMP-9 Quantikine ELISA Kit assay (R&D Systems, USA), we detected the activity of MMP-2 and MMP-9 according to the manufacturer’s instructions. After transfection of siRNA for 24 h, the cells were washed with serum-free medium. The medium was collected 24 h later and centrifuged at 500 rpm for 10 min. The MMP-9 antibody or MMP-2 antibody was coated on the 96-well plate, and the supernatant was added to plate and incubated at 4
Statistical analysis
One-way ANOVAs were used to identify the impact of clinicopathological factors and SerpinE2 expression in EC patients. Patient survival rates were calculated using the Kaplan-Meier method, and statistically significant differences in survival were identified using the log-rank test. All P values were two-tailed. Values of
Results
Association of SerpinE2 expression with clinicopathological characteristics of EC patients
Relationship between SerpinE2 expression and clinical features in endometrial cancer (
72)
Relationship between SerpinE2 expression and clinical features in endometrial cancer (
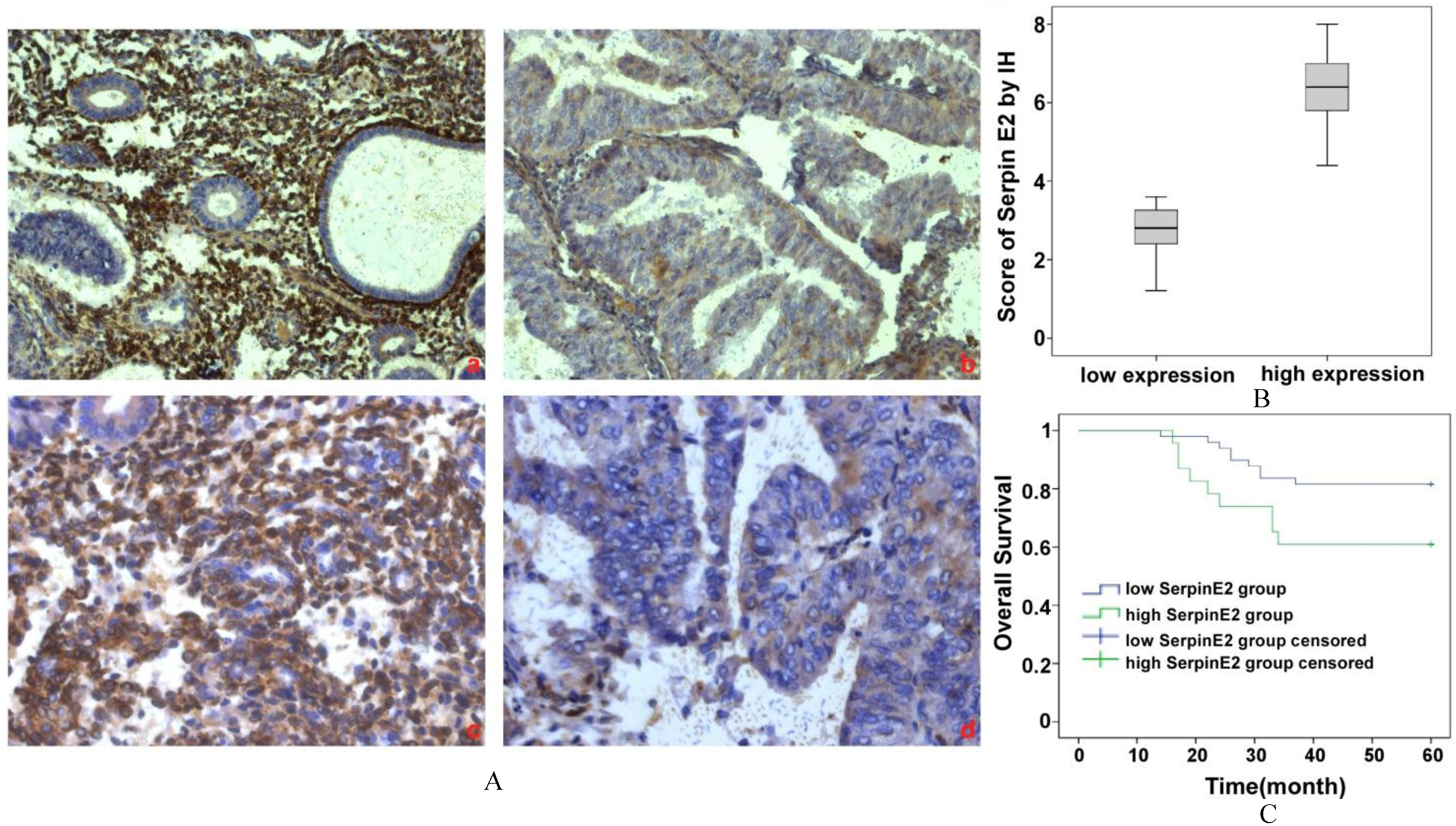
Expression of SERPINE2 in human EC tissue and its clinical value. (A) Immunohistochemical staining of SerpinE2 was performed, and representative images of weak or moderate staining (a and c) and strong positive staining (b and d) are shown. a) and b) are at 200
A total of 72 patients were enrolled in this study. Among them, 36 (36.11%) were
Using the Kaplan-Meier analysis, we compared the difference in the survival ratio between low and high SerpinE2 expression in EC (Fig. 1C). Among the 23 patients with high SerpinE2 expression, 9 patients died (60.9%). Among those with low SerpinE2 expression, 9 patients also died (81.6%, 9/49). When comparing the 5 year overall survival rate of the two groups, there was a significant difference between the high and the low SerpinE2 expression group (
The effect of SerpinE2 on the viability of EC cells
After a 24 h transfection of siRNA-SerpinE2 into EC cells, we measured the mRNA and protein levels of SerpinE2. The QRT-PCR results showed that siRNA reduced the SerpinE2 level to one third of the control levels (Fig. 2A). The gray value of the WB analysis showed that the protein level in the siRNA group was half of that in the NC group (Fig. 2B). To determine the effect of siRNA-SerpinE2 on the proliferation of HTB-111 and Ishikawa cells, an MTT test was performed. The results showed that the proliferation ratio of the siRNA group was 70–75% of that of the NC group in both the HTB-111 and Ishikawa cells (Fig. 2C). The apoptosis assay indicated that siRNA-SerpinE2 significantly induced apoptosis (Fig. 2D). Bajou reported that the anti-apoptosis factor, Bcl-2, is negatively regulated by SerpinE2 [8]. We clarified this by QRT-PCR and WB. siRNA-SerpinE2 up-regulated expression of Bcl-2 at both the mRNA and protein level (Figs 2E and F).
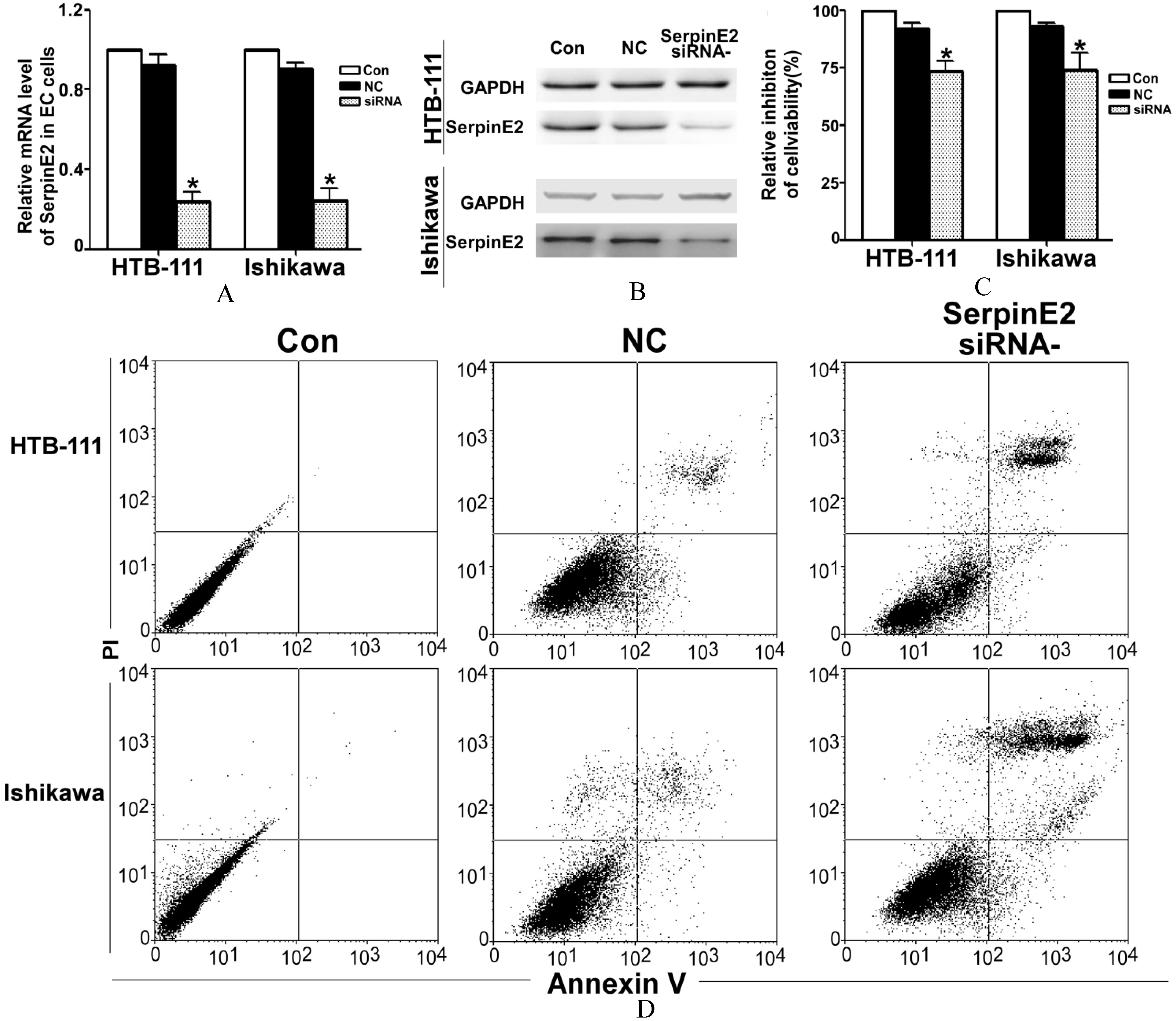
SerpinE2 took part in cell proliferation. (A) After transfection of siRNA-SerpinE2 into HTB-111 and Ishikawa cells for 24 h, we detected the SerpinE2 mRNA levels by QRT-PCR. Compared with the SerpinE2 levels in the untreated control and scrambled siRNA (NC) control groups, siRNA-SerpinE2 sharply decreased SerpinE2 expression (
Furthermore, we also measured the mobility change in EC cells after knock-down of SerpinE2 expression using the wound-healing assay. After cells were transfected for 48 h, the percentage of the wound healing area in the siRNA group was significantly less than that of the control group (Fig. 3A). The transwell assay revealed that there were fewer EC cells in the siRNA group than in the NC and untreated control group (Fig. 3B). The activity of MMP-2 and MMP-9 were both decreased in the siRNA-SerpinE2 group by approximately 65–70% of control (
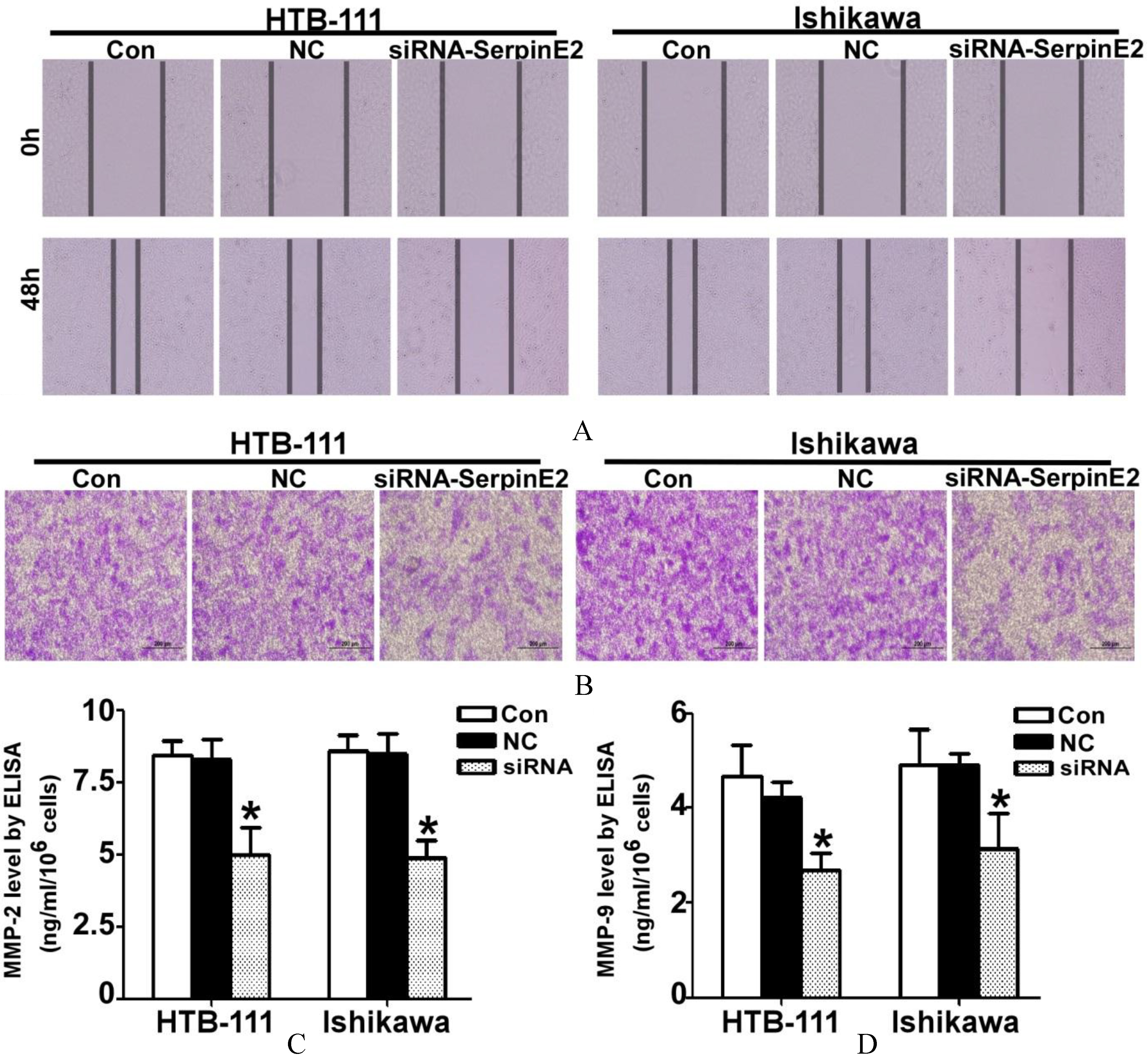
The mobility of EC cells was reduced with suppression of SerpinE2. (A) After HTB-111 and Ishikawa cells were transfected with siRNA-SerpinE2 for 48 h, the wound-scratch assay demonstrated that siRNA-SerpinE2 suppressed the healing of the EC cells; the wounded area was twice the size of that in the control groups. (B) The transwell invasion assay demonstrated that the invasive capacity of EC cells was reduced with siRNA-SerpinE2 treatment. (C) After HTB-111 and Ishikawa cells were transfected with siRNA-SerpinE2 for 48 h, the supernatant was collected to detect the MMP2 and MMP9 activity by ELISA. The mean absorbance value of MMP-2 and MMP-9 in the siRNA-SerpinE2 group was 35–40% of that of control (
Protein inhibitors are the key factors in the maintenance of protease activity, which is the last step in protease activity regulation. SERPINs are a superfamily of widely distributed and structurally related serine and cysteine protease inhibitors that regulate fibrinolysis, coagulation, complement activation, inflammation, cell mobility, cellular differentiation and apoptosis processes. SERPINs always form tight but reversible binary complexes that interact with proteases as inhibitors, substrates, or both and trigger conformational changes of both SERPINs and proteases [9]. SerpinE2/PN-1, unlike any other circulating SERPIN, is barely detectable in plasma but is expressed in tissues and mainly takes part in the regulation of extracellular matrix destruction [10]. Because of this feature, SerpinE2/PN-1 has been studied in the vasculature, nervous system and the behavior of cancer cells. Anoikis is described as apoptosis of adherent cells induced by the disruption of integrin-mediated cell-matrix interactions. Anoikis resistance is the key process of metastasis, and anti-apoptosis is its first characteristic. Numerous studies have suggested that SerpinE2 is required for over proliferation, anti-apoptosis and malignant transformation of tumor cells [11, 12]. Rossignol reported that over-expressed SerpinE2/PN-1/PAI-1 in Chinese hamster ovary fibroblast cells significantly inhibited the activity of plasmin and tissue-type plasminogen activator via the formation of inhibitory complexes and prevented cell detachment and apoptosis [13]. Rømer found that SerpinE2/PN-1/PAI-1 influences sensitivity to etoposide-induced apoptosis through the PI3K/Akt cell survival pathway by acting upstream of PI3K and Akt [14]. Bajou reported that SerpinE2/PN-1/PAI-1 protects EC cells from Fas/FasL-mediated apoptosis through Bcl-2 [8]. In our paper, our results also supported this conclusion. Silencing of SerpinE2 induced inhibition of viability and triggered apoptosis in EC cell lines. In addition, a large body of work has revealed that SerpinE2 is deeply involved in tumor metastasis. Smirnova found that blocking SerpinE2 caused a reduction in M2-like TAMs, diminished CCL2 and changed the collagen deposition, fiber structure and organization. SerpinE2 is one of the most important factors in tumor cell dissemination. Wu described that SerpinE2 promotes melanoma metastasis through the glycogen synthesis kinase 3
Our findings suggest that aberrant expression of SerpinE2 contributes to tumorigenesis in EC through the promotion of cancer cell proliferation and invasion. SerpinE2 may play important roles in the development of EC and work as a valuable marker for metastasis and, thereby, prognosis for EC patients.
Footnotes
Acknowledgments
This work was supported by Science and Technology Planning Project of Guangdong Province (No. 2016ZC0069), and the cultivate scientific research projects of Nanfang Hospotal (No. 2015Z006).
