Abstract
OBJECTIVES:
This study was to explore the prognostic value of connective tissue growth factor (CTGF) expression in endometrial cancer (EC).
METHODS:
We compared CTGF expression in 198 samples from patients with endometrial cancer and 50 samples from patients with healthy endometrial tissues as determined by immunohistochemistry.
RESULTS:
Expression of CTGF was significantly higher in endometrial cancers as compared to normal endometrial tissues. Positive CTGF expression displayed a strong association with CA125 level, histological grade, depth of myometrial invasion and the International Federation of Gynecology and Obstetrics (FIGO) stage. Our findings revealed histological grade, depth of myometrial invasion, FIGO stage, vascular/lymphatic invasion, and the CTGF expression are related to 5-year survival in patients with endometrial cancer. Positive CTGF expression, lymph node status, as well as vascular/lymphatic invasion, were identified as independent prognostic factors in endometrial cancer.
CONCLUTIONS:
Over-expression of CTGF is an independent prognostic factor that will allow the successful differentiation of high-risk population from the group of patients with stage III-IV endometrial cancer. The up-regulation of CTGF may contribute to the progression of endometrial cancer and serve as a new prognostic biomarker in patients with endometrial cancer survival.
Introduction
Endometrial cancer (EC) is one of the most common malignancies affecting the female reproductive tract [1]. However, the molecular and genetic events underlying the development of these tumors remain obscure [2]. The disease most commonly presents with abnormal uterine bleeding and consequently, is often detected in early stages (FIGO stage I) [3]. However, some patients present with advanced stage disease. 35% of the endometrial cancers are diagnosed in the advanced stage of the disease, and with significantly diminished lifetime expectancy [4]. Hysterectomy with bilateral salpingo-oophorectomy is the primary treatment for patients with this localized disease. Thus, there is an urgent need for new therapeutic targets and strategies for endometrial tumorigenesis, both of which may be realized through improved understanding of the molecular mechanisms governing this disease.
Connective tissue growth factor (CTGF), also known as CCN2, belongs to the CCN (CTGF/Cysteine-rich 61/Nephroblastoma) family [5]. All CCN family members are secreted proteins associated with the extracellular matrix (ECM) which play an essential role in processes such as placentation, embryo implantation, embryogenesis, differentiation, and development. These proteins are also implicated in wound healing, fibrotic disorders, and tumorigenesis [6, 7]. Interestingly, the role of CTGF varies depending on the type of cancer. For example, CTGF is an oncogenic factor which promotes tumor progression in pancreatic, prostate, liver, and breast cancer, ovarian cancer as well as sarcomas [8, 9]. Conversely, CTGF functions as a tumor suppressor in the lung, and oral squamous cell cancers [10, 11, 12]. CTGF has been used as a prognostic marker in esophageal squamous cell carcinoma, gastric and lung cancer [10, 13]. However, the expression pattern and functional mechanisms of CTGF in EC have not been established.
In the present study, we examined the expression of CTGF in normal and malignant endometrial tissue in order to determine its involvement in malignant progression. Additionally, we evaluated whether CTGF expression is associated with clinical-pathologic parameters and prognosis in patients with EC.
Materials and methods
Patient characteristics and specimens
One hundred ninety-eight patients with endometrioid adenocarcinoma were enrolled in the present study. All patients had undergone radical hysterectomy with the same group gynecologists in our institute from January 2007 to December 2008. No neoadjuvant chemotherapy treatment before surgery. All the patients were staged according to the FIGO classification system, and patients with advanced stage received the standard chemotherapy and radiotherapy. The mean age of patients was 53 years (range, 22–75 years). 194 (97.8%) patients without lymph node metastasis and the mean preoperative CA125 levels was 41.72 U/mL (range, 3.14–425.1 U/mL). Besides, 50 endometrial tissues from the cases with hysteromyoma at the same period were included in this study, the mean age was 39 years (range, 34–62 years) and the mean CA125 level was 21.1 U/mL. The Consent forms were got from all cases, and this study was approved by the Ethics Committee of Harbin Medical University Cancer hospital.
Connective Tissue Growth Factor (CTGF) Expression in normal endometrial tissues and cancer endometrial tissues
Connective Tissue Growth Factor (CTGF) Expression in normal endometrial tissues and cancer endometrial tissues
Abbreviation: CTGF, connective tissue growth factor.
Representative images of CTGF staining in normal endometrial tissues and endometrial cancers. (a-b) normal endometrial tissues showed negative (a) and 1+ (b) CTGF staining. (c-f) endometrial cancer tissues showed negative (c), 1+ (d), 2+ (e), 3+ (f) CTGF staining, respectively. Magnification 100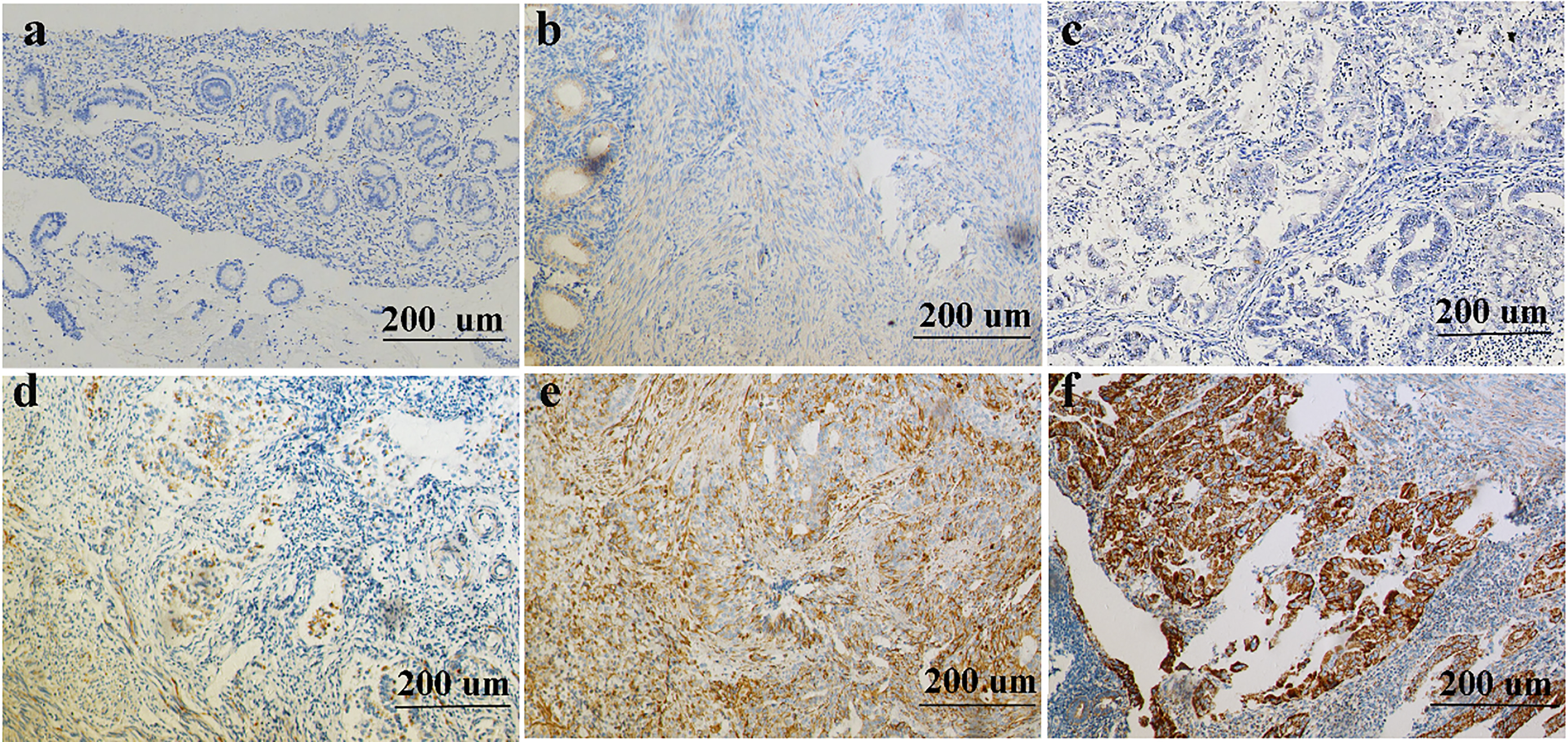
For immunohistochemical staining, four
Staining evaluation
The scoring procedure was carried out by two experienced independent pathologists. Evaluation of CTGF expression was scored according to the percentage of positively stained cells in the field of view, as follows: negative (0), no staining; weak (score 1+), 0–19% of cells stained; moderate (score 2+), 20–49% of cell stained; and strong (score 3+), 50–100% of cells stained. Slides with 0 or 1+ staining were classified as low expression, and slides with 2+ or 3+ staining were classified as a high expression [14].
Statistical analysis
Chi-square tests were applied to analyze the categorical variables. The p-values were shown for the difference of these variables between the CTGF expressions. The 5-year survival rate was analyzed with Kaplan-Meier curve analysis and tested with the log-rank test. COX regression models were performed to test the risk factors. Significance tests were two-tailed and p-values less than 0.05 were considered statistically significant. All analyses were performed using the SPSS statistical (version13.0; SPSS, Chicago, IL, USA) and MedCal.
Results
Expression of CTGF in EC tissues is significantly higher than normal endometrial tissues
CTGF was observed in both glandular and stromal with different coloration. Also, as shown in Fig. 1, CTGF was localized mainly in the cytoplasm or membrane of normal and tumor cells. Normal endometrial tissues exhibited weak or no CTGF immunoreactivity (Fig. 1a and b). However, endometrioid adenocarcinoma tissues showed no or weak CTGF immunoreactivity (Fig. 1c and d), moderate or strong CTGF expression (Fig. 1e and f). As EC specimens were examined, negative CTGF expression was detected in 95 (48%) of 198 patients, whereas positive CTGF expression was detected in 103 (52%) of 198 patients. In normal endometrial tissue, 16% (8 of 50) of samples stained positive for CTGF (Table 1, Fig. 2). As shown in Table 1 and Fig. 2, the level of CTGF was significantly higher in EC tissues compared to the normal endometrial tissue (
Connective tissue growth factor (CTGF) expression in endometrial cancer is significantly higher than normal endometrial tissues (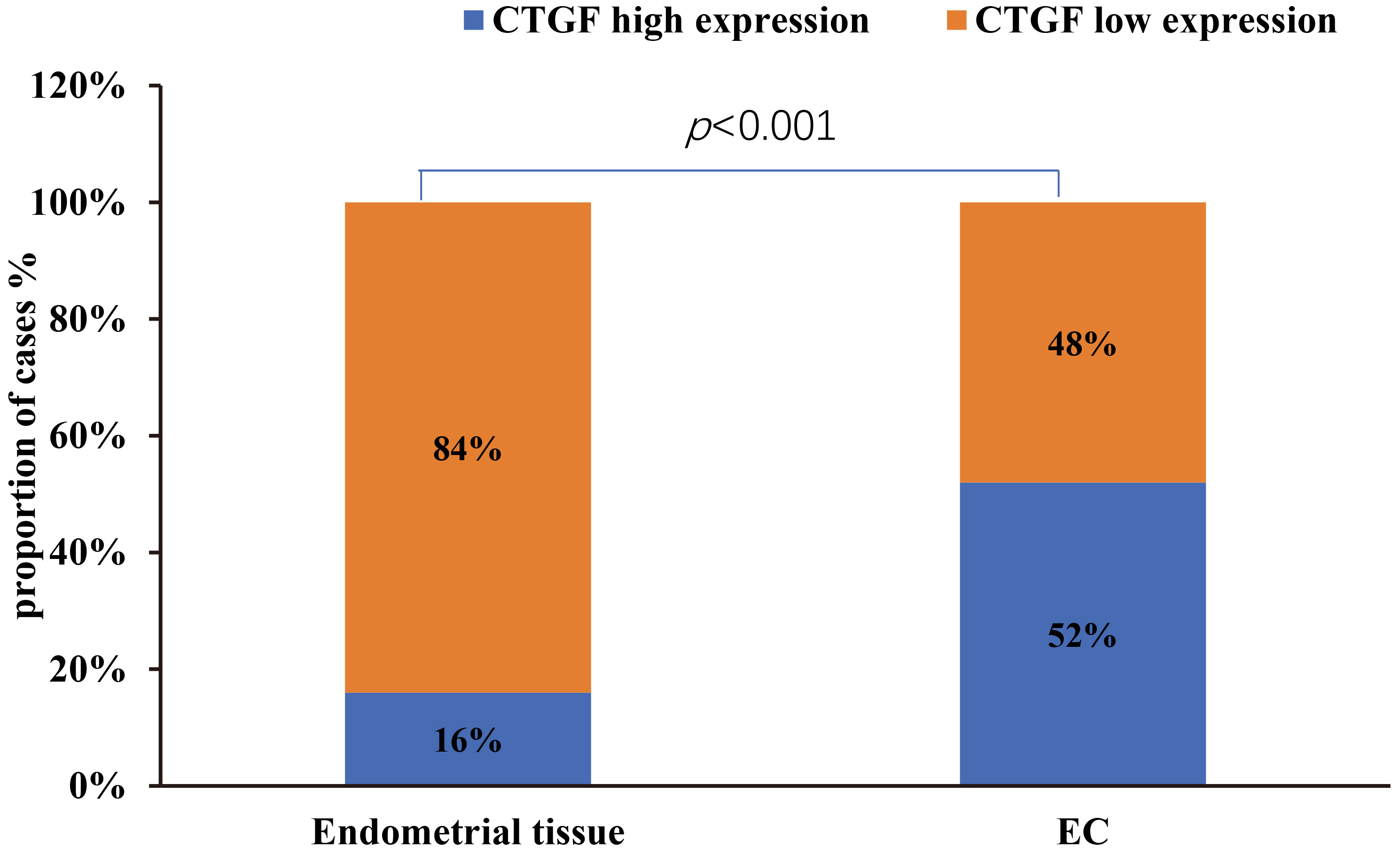
Relationships between CTGF expression and clinical variables were analyzed for patients with EC. As shown in Table 2, there was no significant correlation between level of CTGF and age (
Association between CTGF Expression (positive/negative) and Clinicopathologic Parameters
Association between CTGF Expression (positive/negative) and Clinicopathologic Parameters
Abbreviation: FIGO, International Federation of Gynecology and Obstetrics; G1, well differentiated; G2, moderately differentiated; G3, poorly differentiated; # Chi-square corrected.
Correlation of CTGF staining intensity with CA125. Distribution of CTGF staining intensity in endometrial cancers of different CA125 levels. The 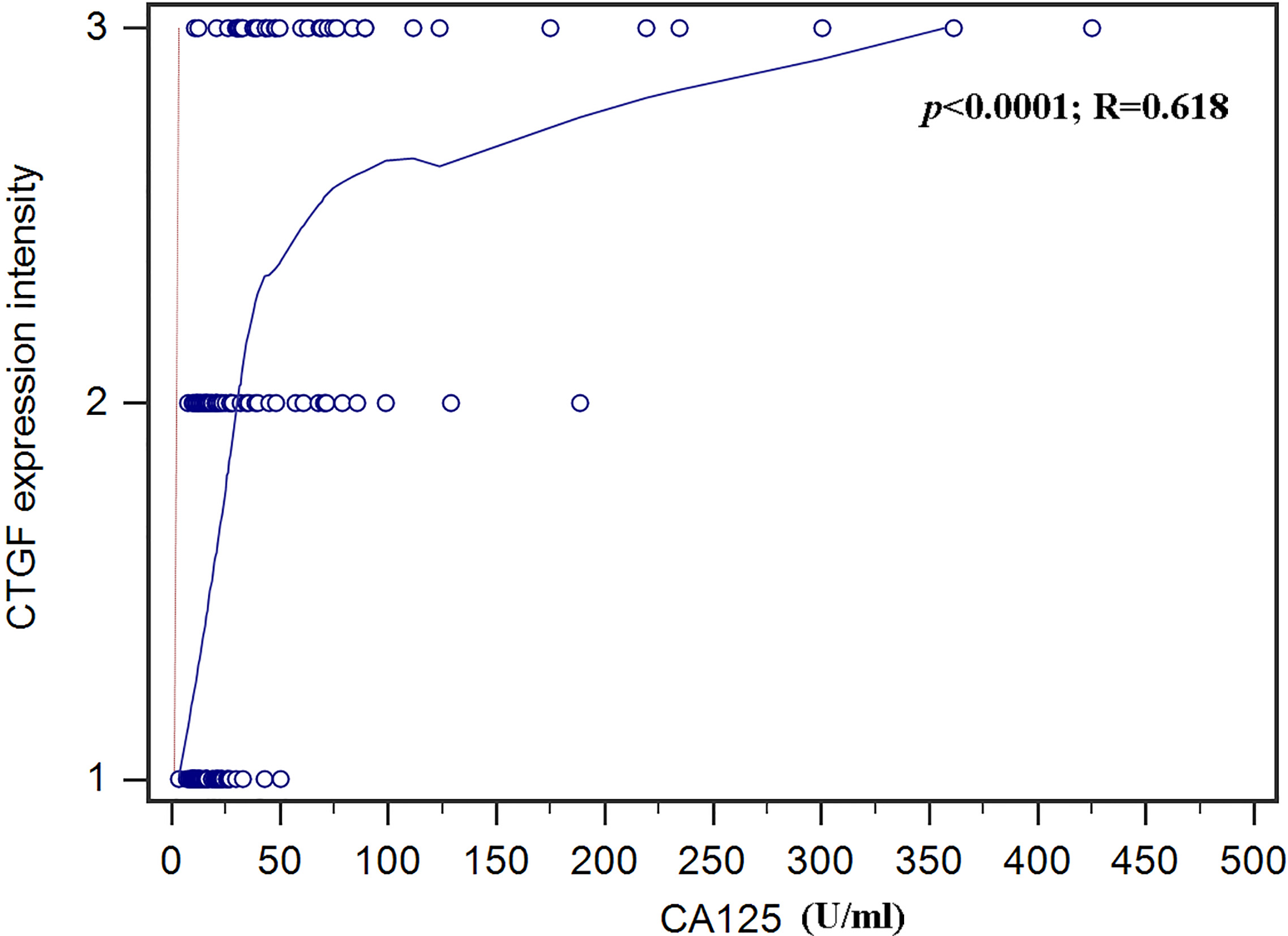
Table 3 demonstrates CA125 levels (
Univariate Cox-regression analysis of various clinicopathological parameters and their use as prognostic markers for disease-specific 5-year survival in EC patients
Univariate Cox-regression analysis of various clinicopathological parameters and their use as prognostic markers for disease-specific 5-year survival in EC patients
Abbreviation: CI, confidence interval; HR, hazard ratio; G1, well differentiated; G2,moderately differentiated; G3, poorly differentiated; FIGO, International Federation of Gynecology and Obstetrics.
Multivariate Cox-regression analysis of various clinicopathological parameters and their use as prognostic markers for disease-specific 5-year survival in EC patients
Kaplan-Meier survival curves for 198 patients with endometrial cancer, grouped according to CTGF expression. 5-year survival was significantly lower in patients with high CTGF expression than in patients with low CTGF expression (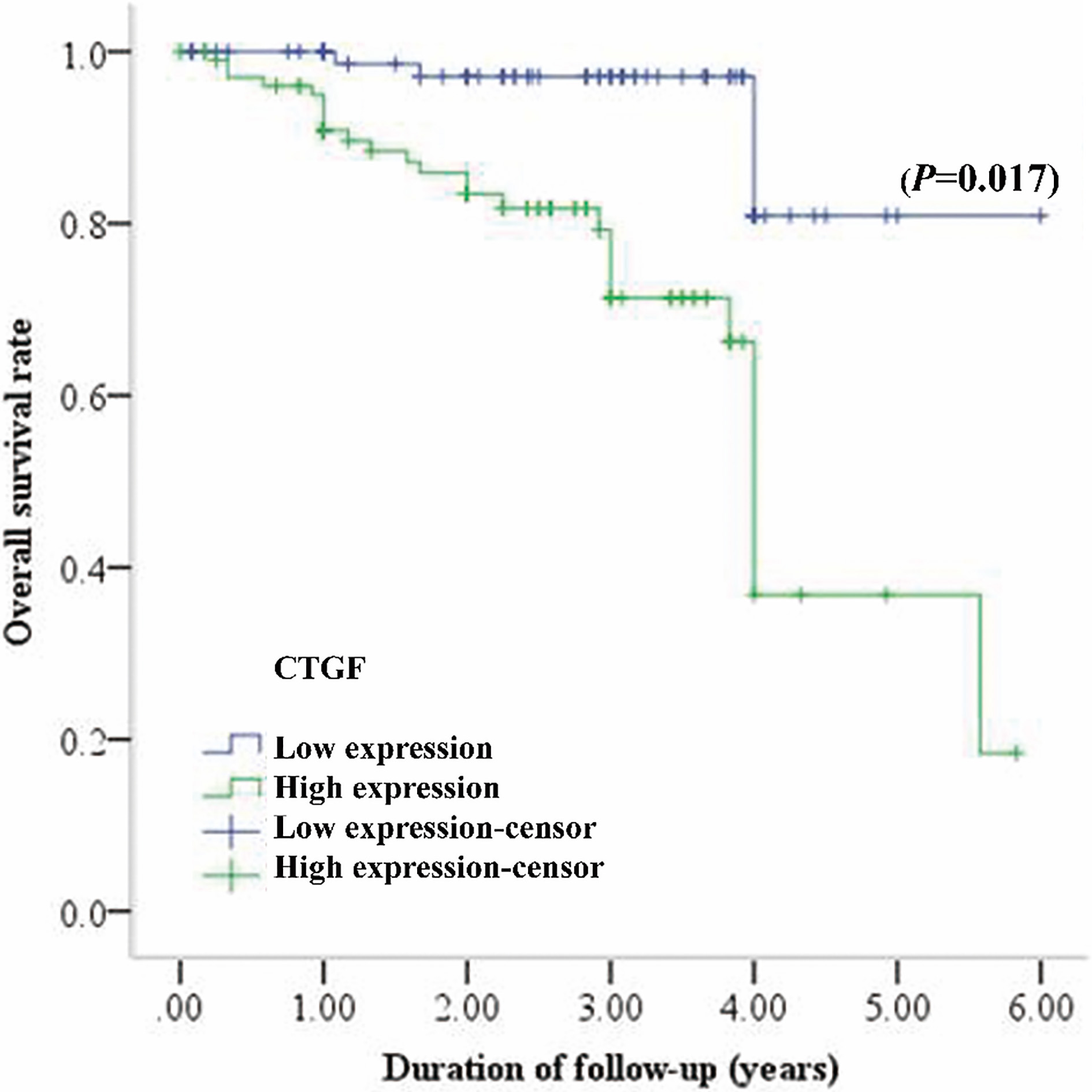
Historically, the age of onset of EC is mostly after menopause. However, the incidence of EC in premenopausal women has increased dramatically due to earlier-onset obesity in the past ten years [15]. Even though there are effective treatment methods, survival is still based on the stage and histology of the diagnosis. Most EC patients with stage I-II will have a favorable prognosis, whereas patients with stage III or IV will have a worse likelihood of survival [16, 17, 18]. Although CA125 remains as one the most widely used biomarkers for the potential diagnosis and prognosis of EC [19, 20, 21, 22], it demonstrates several limitations, including the occurrence of false-positive diagnosis and over-treatment due to the reduced sensitivity and specificity of CA125 level testing [23, 24]. Therefore, it is of a high clinical significance to find specific valuable biomarkers which can screen high-risk women, make a preoperative stratification of patients as high-risk or low-risk categories, support personalized therapeutic approaches.
In the present study, we detected high CTGF expression in 52% of EC patients, which is significantly higher than 16% in normal endometrial tissues. The CTGF expression was predominantly observed in the cytoplasm and membrane of cancer cells. The rate of positive CTGF expression was increased significantly in poorly-differentiated EC, levels of CA125 expression
Interestingly, a recent study demonstrated that CTGF expression was increased in epithelial ovarian cancer tissues, and its expression correlated with the stage of this disease. The CTGF levels in stage III-IV of ovarian cancer were significantly higher than stage I-II [28]. These are consistent with our findings and indicate that over-expression of CTGF may contribute to the progression of EC. The mechanism of CTGF in mediating the EC progression is unclear. However, CTGF had been found to accelerate tumor progression through mediating tumor-stroma interactions between hepatoma cells and hepatic stellate cells [29]. There is a positive feedback loop between CTGF and chemokine ligand 18 in hepatocellular carcinoma metastasis [30]. Thus, promoting tumor-stroma interactions might be one of the mechanisms which are mediated by CTGF to accelerate tumor progression.
Univariate and multivariate analysis revealed that high CTGF expression was a powerful independent predictor for the poor survival of EC patients. The overall 5-year survival rate of EC patients with a positive CTGF expression and a negative CTGF expression was 36.8% and 80.9%, respectively. Our results indicated that over-expression of CTGF might play a role in the aggressive behavior of EC. These are consistent with various studies which showed that CTGF is associated with a more aggressive phenotype and with tumor progression in kind of solid tumor tissues [8, 31, 32, 33, 34, 35, 36, 37, 38, 39]. Dr. Kang and colleagues found that CTGF is a critical component of the gene signature that facilitates osteolytic bone metastasis in breast cancer cells [40]. Administration of FG-3019, a humanized anti-CTGF monoclonal antibody, inhibits tumor growth and metastases in xenograft and orthotopic models of pancreatic cancer in mice [35, 41]. Therefore, CTGF up-regulation in EC may promote the tumor growth and metastases which is associated with a lower survival rate of the patients.
The mechanisms responsible for CTGF over-expression in EC patient needs further investigation to elucidate. Recent studies demonstrated that microRNAs are essential mediators in the mechanism of human cancer. MiR-143 was reported to target CTGF and exert tumor-suppressing functions in epithelial ovarian cancer [42]. In addition to MiR-143, MiR-26b was also found to govern the CTGF expression in osteosarcoma. When MiR-26b levels sharply increased, the CTGF expression was downregulated [43]. Based on these findings, we speculated that MiR-143 and MiR-26b might be a potential regulator of CTGF. However, further investigations are still needed to verify the above-mentioned underlying mechanisms.
In conclusion, our findings indicated that on clinical inspection, EC patients with increased CTGF expression have more chance to develop to the advanced stage of EC and shorter survival time. Mainly, CTGF seemed to be an independent prognostic factor that will allow the successful differentiation of high-risk population from the group of patients with stage III-IV disease. Moreover, CTGF seems to be a novel biomarker for predicting the prognosis of EC patients. Also, over-expression of CTGF in EC patients results in poor survival.
Footnotes
Acknowledgments
I want to express my most profound appreciation to all those who provided me the possibility to complete this manuscript. A special gratitude I give to our department director Dr. Yu, whose contribution to stimulating suggestions and encouragement, helped me to coordinate my clinical work especially in writing this manuscript. Furthermore, I would like to acknowledge with much appreciation for the crucial role of Dr. Jarrett, who provided language help. Special thanks go to my teammate, Dr. Lu, who was involved in conceiving the experimental design.
Conflict of interest
None declared.
