Abstract
BACKGROUND:
Spinal cord injury (SCI) is a disabling condition characterized by multilevel skeletal muscle impairment and rapid cortical and trabecular bone loss. Rehabilitation is a cornerstone of the long-term management of patients with SCI; however, the optimal rehabilitation strategy for improving bone health has not been fully characterized.
OBJECTIVE:
To characterize the current evidence supporting different rehabilitation interventions improving bone health in patients with SCI.
METHODS:
On November 17
RESULTS:
Out of 499 records, 11 RCTs met the eligibility criteria and were included. Electrical stimulation combined with physical exercise was assessed by 5 studies, standing intervention was assessed by 3 studies, vibration was assessed by 1 study, ultrasound therapy was assessed by 1 study, and electroacupuncture combined with a pulsed magnetic field was assessed by 1 study. The rehabilitation intervention was administered combined with pharmacological treatment (3 studies) or alone (8 studies). Positive effects in terms of BMD were reported by 3 studies. The quality assessment revealed some concerns in 9 out of 11 studies, in accordance with the Cochrane Risk of Bias assessment – version 2.
CONCLUSION:
Our data suggest that multicomponent interventions including rehabilitation might be considered a suitable option to improve bone health management in SCI patients. Further studies are mandatory to characterize the optimal combination of non-pharmacological interventions reducing bone loss and improving the risk of fractures in patients with SCI.
Introduction
Spinal cord injury (SCI) is a disabling neurological condition characterized by the loss of both motor and sensory function and visceral dysfunction [1, 2, 3, 4]. The prevalence of SCI is highly heterogeneous among different countries, ranging from 13.0 per million to 163.4 per million people, with a significant proportion of traumatic spinal cord injuries affecting young adults [5, 6, 7]. Regrettably, SCI is related to extremely high healthcare and assistance costs, while social and personal costs cannot be quantified [5, 6, 7].
After spinal cord injury, bone loss is a common complication related to skeletal muscle system disuse and impaired mechanical stimuli [3, 8]. More in detail, osteoporosis generally affects the skeletal system caudal to the spinal cord damage, with bone loss occurring most rapidly and significantly in the distal femur and proximal tibia [9, 10]. Within two to three years of SCI, individuals show a 50–100% trabecular bone mineral density (BMD) reduction, and 40–80% lower cortical bone mass [11, 12, 13]. As a result, SCI patients have a 20- to 100-fold higher fracture risk compared to the general population [14]. Therefore, it has been proposed that the comprehensive management of bone loss should be performed in patients with SCI, including both pharmacological and non-pharmacological approaches [15].
In this context, growing evidence highlighted the crucial role of a comprehensive rehabilitation program targeting both physical and psychosocial impairment of SCI patients [8, 16, 17, 18, 19, 20]. To date, several studies reported positive effects of physical exercise and physical activity on bone health in several disabling conditions [21]. On the other hand, rehabilitation might effectively target not only bone health but also balance and the risk of falls [22].
Despite these considerations, the optimal rehabilitation strategies to prevent bone loss in people with SCI have yet to be fully characterized [23, 24, 25, 26]. Furthermore, to our knowledge, no previous systematic review including only randomized controlled trials (RCTs) assessed the effects of specific rehabilitation protocols on bone health of SCI patients.
Therefore, the objective of this systematic review was to provide evidence supporting different rehabilitation strategies for improving bone health in patients with SCI. Moreover, we aimed at characterizing the effects of specific rehabilitation prescriptions in bone health in order to pave the way to an evidence-based approach preventing bone loss in people with SCI.
Methods
Registration
This systematic review of RCTs follows the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) statement [27]. A preliminary search has been performed in the international prospective register of systematic reviews (PROSPERO) for comparable review protocols in progress, without finding similar studies. The systematic review was then submitted to PROSPERO and accepted on 27
Search strategy
The literature search has been performed from onset up to November 17
Selection criteria
In accordance with the PICO model [28], we considered eligible all randomized controlled trials (RCT) satisfying the following criteria:
P) Participants: adult (aged more than 18 years) male or female patients with spinal cord injury, without restriction in terms of time of injury. I) Intervention: all rehabilitation interventions administered as exclusive or integrated therapeutic intervention. C) Comparator: any comparator. O) Outcome: the primary outcomes were bone macroscopical effects, assessed by: i. Bone Mineral Density (BMD); ii. Bone Mineral Content (BMC); iii. Trabecular Bone Thickness (TBTh) and Trabecular Bone Separation (TBSp); iv. bone strength indices [Compressive Strength Index (CSI), Cortical Thickness Index (CTI), Cortical Bone Volume (CBV), Buckling Ratio (BR), and Torsional Strength Index (TSI)]; v. quantitative bone ultrasound (qUS) parameters; vi. Cross-Sectional Area (CSA) of red and yellow bone marrow and cortical and trabecular bone. The secondary outcomes were: i. changes in body composition; ii. change in bone biomarkers; iii. hormonal and metabolic changes; iv. changes in physical functioning and physical performance.
Only RCTs published in peer-reviewed International Journal were included. No publication date restriction was applied.
The exclusion criteria were: i. participants with pregnancy, clinical instability (defined as hemodynamic alterations, respiratory events, abnormal laboratory values, reduced level of consciousness or temperature alterations), or palliation; ii. studies involving animals; iii. doctorate theses, conference proceedings, and reviews of the literature; iv. not RCT studies; v. language other than English.
Firstly, the articles resulting from the database-specific search strings were examined for duplication removal. Subsequently, the titles and abstracts were screened by two investigators that independently excluded records that did not meet the inclusion criteria. Lastly, the selected articles were examined in full text, and the articles included in the review were thus extracted. Any conflicting record along this chain was discussed between the two investigators, and in case of unresolved disagreement, a third reviewer was involved to reach consensus.
All data were extracted by two independent reviewers through Excel database. Any difference was solved by discussion between the two reviewers or by consulting a third reviewer.
The data extracted were: i. authors, journal, publication year, and nationality; ii. study design; iii. sample characteristics [number of participants, mean age and age range, gender, time of injury, lesion level and grade, baseline body mass index (BMI) and osteoporosis grade]; iv. comparator characteristics; v. intervention characteristics [type, duration, frequency, and intensity of treatment in the experimental group and in the control group(s)]; vi. primary and secondary outcomes; vii. duration of the study (with possible follow-up); viii study results.
Data were independently synthesized by two reviewers. A qualitative analysis of the evidence was performed, given that the heterogeneity of the study samples, the type of intervention and the outcome measures did not allow a quantitative analysis of the data.
A subgroup analysis was performed based on participant characteristics, time from SCI, type of intervention, and outcome assessed.
Quality assessment and risk of bias
The qualitative analysis of the studies included in the review was carried out using the PEDro scale [29], which consists of 11 items. In particular, the first item assesses the external validity (or applicability) of the study although is not considered for the purposes of the total score; items 2–9 evaluate the internal validity of the study, while items 10–11 evaluate the interpretability of the results based on the statistical information. Each criterion has “yes” and “no” response options, 1 point for each item whose answer is “yes”, 0 points for items whose answer is “no”. The maximum score obtainable is 10/10. Scores from 9 to 10 were considered “excellent”, 6 to 8 were considered “good”, 4 to 5 were considered “fair”, and
The risk of bias assessment of the studies included in the review was performed following the Cochrane Risk of Bias assessment – version 2 (RoB 2) [30], a validated tool for assessing the risk of bias in RCTs. It includes five different domains, each subdivided in criteria that are judged individually (low, high, unclear risk of bias). The overall judgment of a domain derives from the overall judgment of the various criteria. Studies were considered with low risk of bias if all domains evaluated had low risk of bias. Studies in which at least one domain presented unclear risk, were considered studies with some critical issues. Lastly, studies were considered with high risk of bias if at least one domain had a high risk of bias [30].
Results
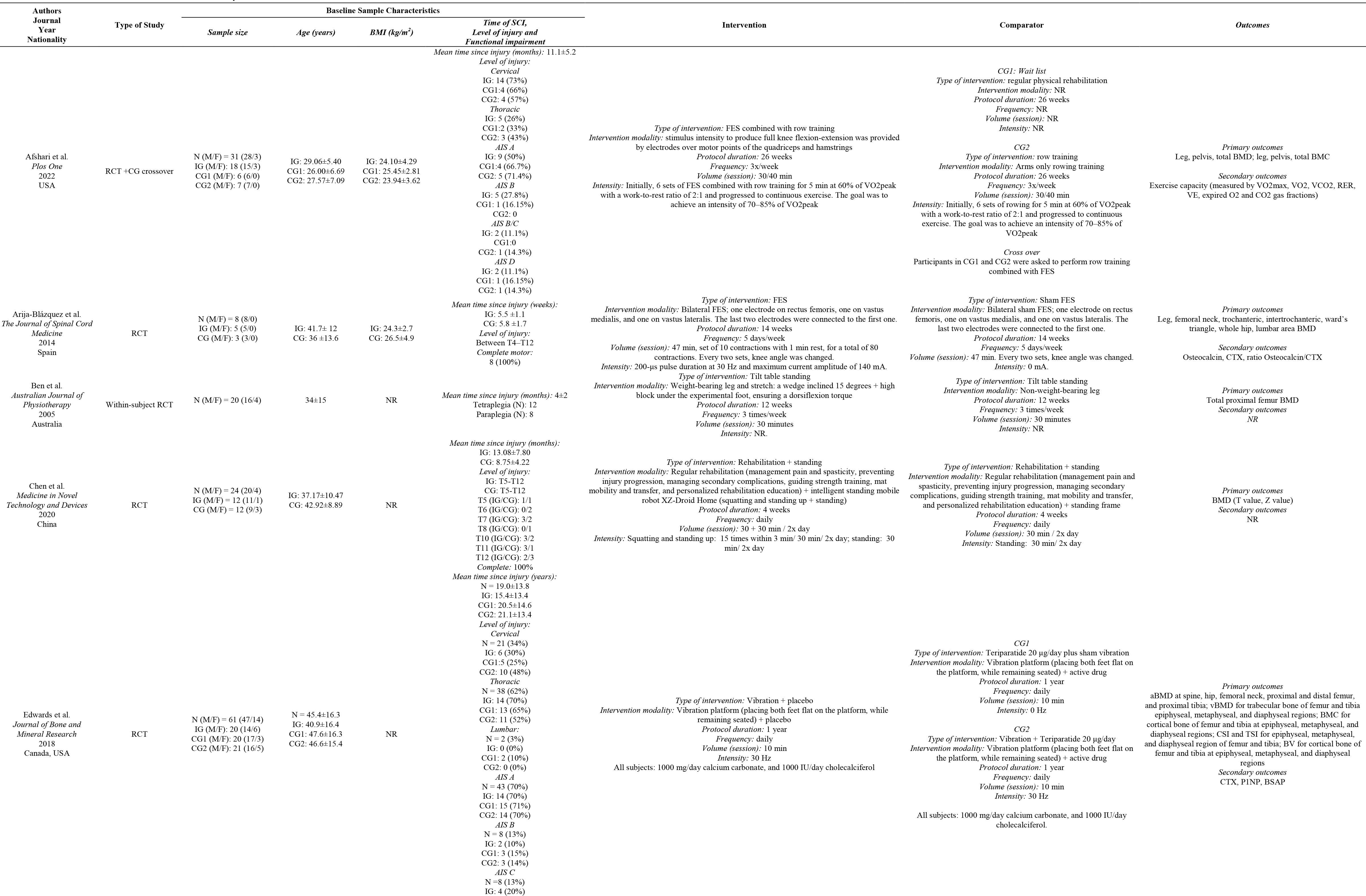
PRISMA 2020 flow diagram.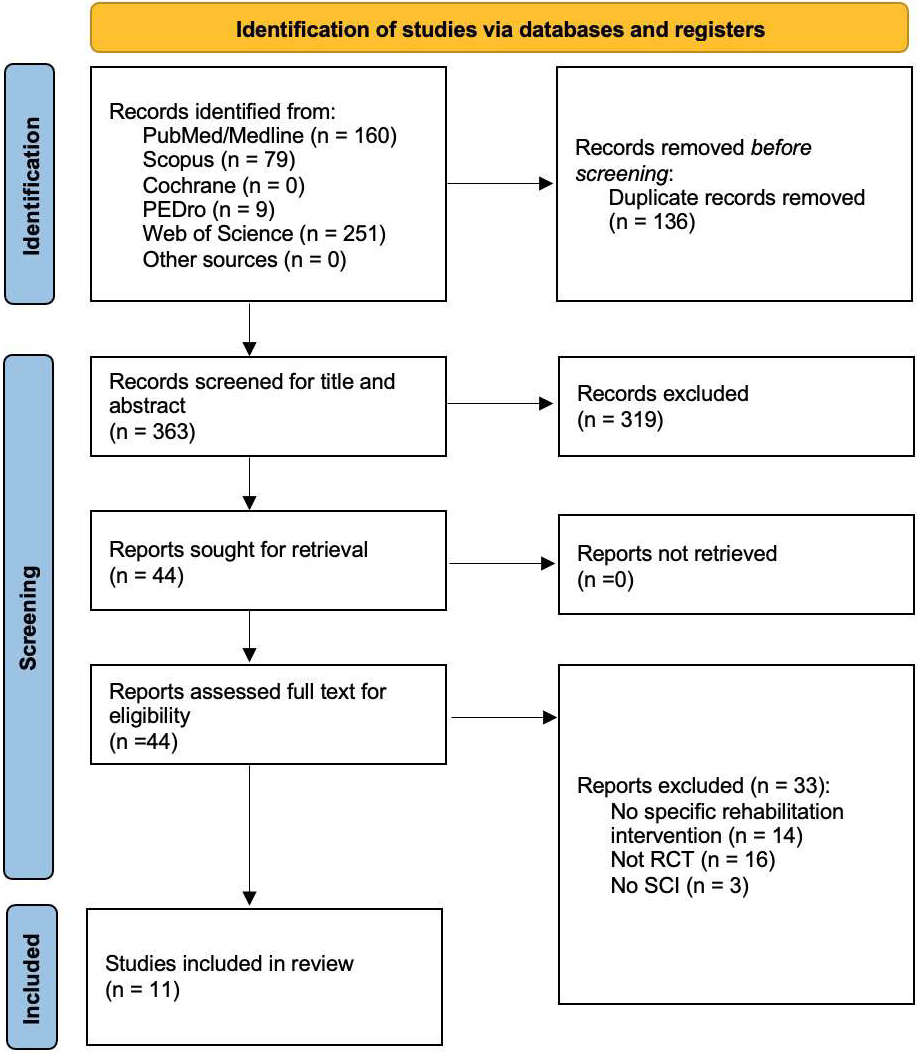
Overall, a total of 499 records resulted from the 5 databases assessed, while no other records were identified from other sources. After duplication removal, a total of 363 studies were assessed for eligibility and screened for title and abstract. As a result, 319 records were excluded, and 44 studies were sought for retrieval and then screened in full text. Lastly, 11 studies [31, 32, 33, 34, 35, 36, 37, 38, 38, 40, 41] were included in the present systematic review. Figure 1 shows the PRISMA flow diagram, reporting further details about the article selection process. Supplementary Table 2 summarizes the reason for exclusion of the remaining 33 studies assessed full-text for eligibility.
The following studies were included in the present systematic review: Afshari et al. [31], Arija-Blázquez et al. [32], Ben et al. [33], Chen et al. [34], Edwards et al. [35], Groah et al. [36], Holman et al. [37], Morse et al. [38], Shackleton et al. [39], Warden et al. [40], Xu et al. [41].
The studies included were published between2001 [40] and 2022 [31, 39] and were conducted in America (USA
The present review included a total of 301 patients (250 males, and 51 females). The patients were characterized by a mean age ranging between 23.9
Patients with cervical SCI were assessed in five studies [31, 35, 37, 39, 40]; patients with thoracic SCI were evaluated in eight studies [31, 32, 34, 35, 37, 39, 40, 41], and patients with lumbar SCI was assessed in two studies [35, 41]. Two studies did not clarify the level of injury [33, 38]. Asia Impairment Scale (AIS) was the most used tool used to characterize the functional impairment of SCI (
Literature search strategy on different databases
Literature search strategy on different databases
Table 1 summarizes in detail the sample characteristics of both intervention groups and comparator groups of each study included in the present review.
Heterogeneous rehabilitation modalities were assessed by the RCTs included. In particular, rehabilitation interventions currently proposed were:
Functional Electrical Stimulation (FES) combined with physical exercise was assessed in five studies [31, 32, 36, 37, 38]. Afshari et al. [31] positioned electrodes on the quadriceps and hamstrings muscles and delivered a stimulus to produce full knee flexion-extension during arms rowing training. The group compared this intervention against conventional physical rehabilitation arm and rowing training arm [31]. Similarly, Morse et al. [38] assessed the effects of FES and rowing training combined with Zoledronic acid 5 mg/100mL [38]. Arija-Blázquez et al. [32] performed bilateral FES, placing three electrodes on quadriceps muscle to produce contraction, comparing it to sham FES [32]. Groah et al. [36] performed FES bilaterally on the quadriceps muscle combined with non-specified individualized inpatient rehabilitation program, comparing it to the same rehabilitation program without FES [36]. Holman et al. [37] used FES to perform open kinetic chain neuromuscular electrical stimulation-evoked resistance training, with electrodes applied on distal and proximal thigh. The intervention group (IG) was also treated with transdermal testosterone patches, compared with transdermal testosterone patches alone [37]. Standing modalities were studied in threeRCTs [33, 34, 39]. More in detail, Ben et al. [33] tested weight-bearing and stretch achieved through a block under the experimental foot combined with a tilt table standing. The control was the opposite foot, without weight-bearing [33]. Chen et al. [34] assessed an intelligent standing mobile robot (XZ-Droid Home) with which the patient performed squatting and standing up, and tested it against a regular standing frame. Both IG and control group (CG) underwent rehabilitation, comprising management of pain and spasticity, preventing injury progression, managing secondary complications, guiding strength training, mat mobility and transfer, and personalized rehabilitation education [34]. Lastly, Shackleton et al. [39] used robotic locomotor training to achieve exoskeleton walking in IG. This training modality was compared to activity-based training, which focused on prehabilitation, muscle recruitment, posture and joint stability, resistance and endurance training, pregait, and gait training [39]. Vibration therapy was studied by Edwards et al. [35]. The group used a vibration platform over which the patient placed both feet flat while remaining seated. This IG was compared to the administration of teriparatide 20 Ultrasound (US) therapy was assessed by Warden et al. [40]. The authors applied active coupled US-heads on medial and lateral surfaces of the calcaneus, comparing it with sham treatment on contralateral calcaneus [40]. Electroacupuncture combined with a pulsed magnetic field was assessed by Xu et al. [41]. More in detail, electroacupuncture was performed by treating points GB 34 and GB 39 on both sides. This treatment was combined with XT-20 0 0B Osteoporosis Treatment Apparatus (pulsed magnetic field below the level of spinal cord injury), Reneed tablet, and rehabilitation treatment (standing, limb air pressure, limb function training) [41].
Rehabilitation was combined with pharmacological and nutritional intervention as it follows. Morse et al. [38] treated IG patients with a one-time infusion of Zoledronic acid 5 mg/100mL solution [38]. Holman et al. [37] used transdermal testosterone patches in both IG and CG. In the study by Edwards et al. [35], control groups received teriparatide 20
BMD was evaluated in eight studies [31, 32, 33, 34, 35, 36, 39, 41]. Afshari et al. [31] assessed leg, pelvis and total BMD, and reported a significantly lower decrease in pelvis BMD (
BMC was assessed in three studies [31, 35, 40]. Afshari et al. [31] did not find significant differences in leg, pelvis, and total BMC (
Trabecular bone thickness and trabecular bone separation parameters were assessed by Holman et al. [37] for distal femur and proximal tibia, though the authors did not perform statistical inference and multiple comparisons of the results [37].
Bone strength indices were assessed in two studies [35, 38]. Edwards et al. [35] computed CSI and TSI for epiphyseal, metaphyseal, and diaphyseal regions of femur and tibia, without reporting significant differences (
Bone ultrasound attenuation (BUA) and speed of sound (SOS) were evaluated in one study [40]. Although these parameters had a significant worsening over time (
Main characteristics of the studies included in the systematic review
Main characteristics of the studies included in the systematic review
Holman et al. [37] assessed the yellow and red bone marrow CSA and cortical bone CSA at proximal, middle, and distal femoral levels, though the authors did not perform statistical inference and multiple comparisons of the results [37]. Further details are shown in Table 2.
Changes in bone biomarkers were assessed in four studies [32, 35, 36, 41].
Serum alkaline phosphatase (ALP) was analyzed in the study by Xu et al. [41], reporting a significant difference in the intragroup analysis in both groups and in the intergroup analysis ( Procollagen type I C-peptide (PICP) was analysed in one study [41]. Xu et al. reported a significant difference in the intragroup analysis in both groups ( Bone gamma-carboxyglutamic acid containing protein (BGP) was analyzed in one study [41], reporting a significant difference in the intragroup analysis and in the intergroup analysis ( Osteocalcin was evaluated in two studies [32, 36], without reporting significant results ( Urinary N-terminal telopeptide (N-TX) was evaluated in one study [36], at 6 weeks and at 3 months after intervention without reporting significant differences in intragroup and between group analysis ( Collagen type 1 cross-linked C-telopeptide (CTX) was analyzed in two studies [32, 35], which did not report significant changes following intervention [32, 35]. The ratio Osteocancin/CTX was calculated in one study [32], that did not report significant changes [32]. Type 1 procollagen amino-terminal propeptide(P1NP) was analyzed in one study [35], which did not report significant changes following intervention [35]. Bone-specific alkaline phosphatase (BSAP) was analyzed in one study [35], which did not report significant changes following intervention [35].
Lastly, changes in physical functioning and physical performance were assessed by Afshari et al. [31], reporting significant improvement in the IG (
Cochrane Risk of Bias assessment – version 2 (RoB 2).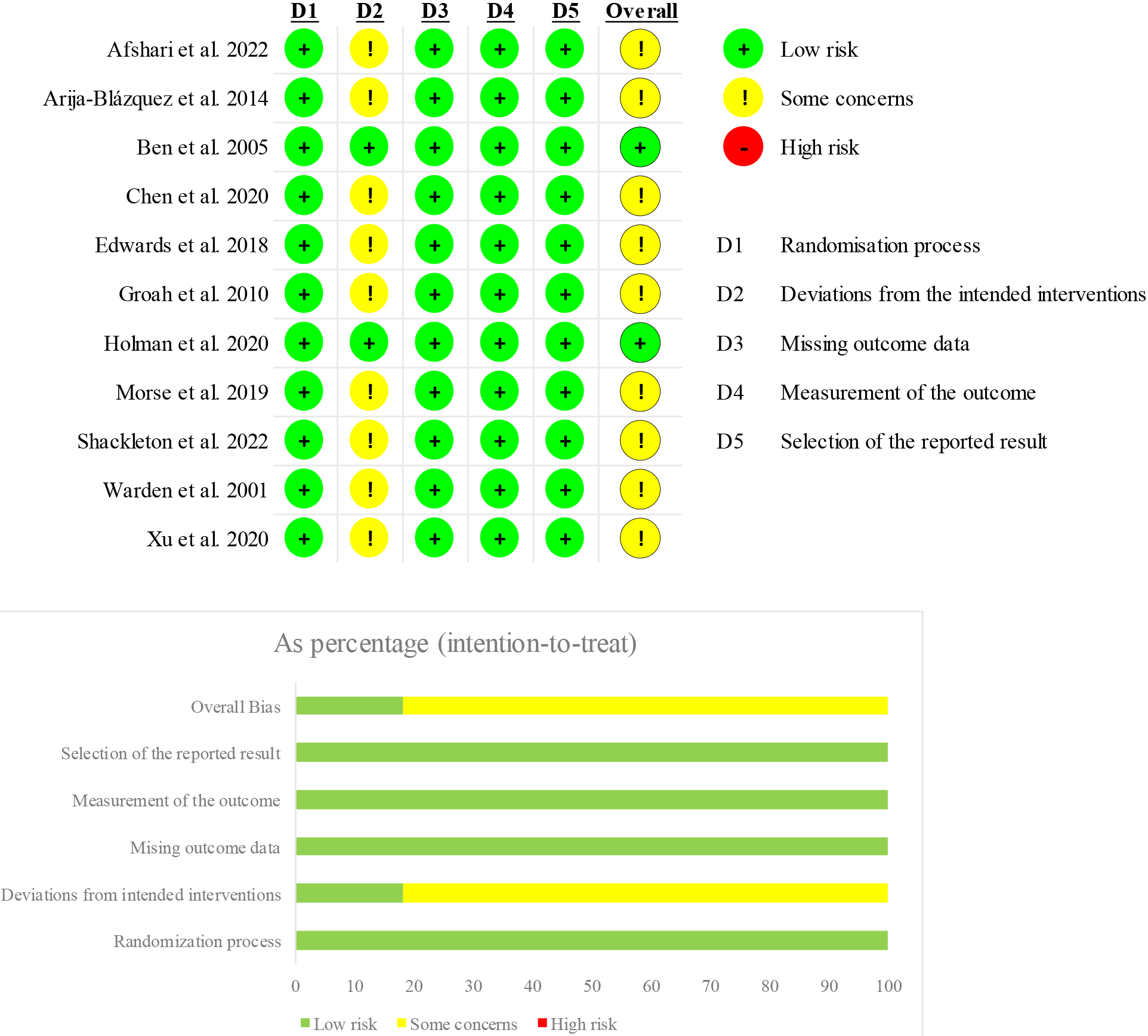
Main findings of the studies included in the systematic review
Qualitative assessment of the studies included using the PEDro scale
Note: Eligibility criteria item does not contribute to total score. 0
In accordance with the PEDro scale [29], two studies resulted in poor quality [31, 38], three studies were characterized as fair [36, 39, 41], while the remaining six studies resulted in good quality scores [32, 33, 34, 35, 37, 40]. Table 3 shows in detail the results of the PEDro scale for each item.
The risk of bias assessment was performed according to the Cochrane Risk of Bias assessment – version 2 (RoB 2) [30]. Some concerns emerged from this analysis in domain 2, specifically “Deviations from the intended interventions”, due to lack of an appropriate analysis estimating the effect of assignment to intervention. This resulted in total outcome of “some concerns” for 9 studies out of 11 [31, 32, 34, 35, 36, 38, 39, 40, 41]. Further details about the risk of bias assessment of each study are shown in Fig. 2.
Discussion
Rehabilitation is currently considered a cornerstone in the complex framework of care of SCI patients, with growing research suggesting several therapeutic strategies to improve bone health in these patients [3, 8, 16]. However, bone loss still remains a major issue in SCI patients and the optimal rehabilitation approach to prevent this burdensome condition has not been standardized yet.
Thus, this systematic review of RCTs aimed at summarizing the current evidence supporting the different therapeutic strategies to improve bone health in SCI patients trying to characterize the specific biological effects of a comprehensive rehabilitation approach to these patients.
Interestingly, our findings identified several therapeutic interventions to improve bone health in SCI patients, including FES, physical exercise, vibration therapy, standing, ultrasound, and electroacupuncture combined with pulsed magnetic field. Taken together, the results of the present systematic review underlined that several instrumental therapies were assessed to improve bone health of SCI patients, but with conflicting results and only 3 studies suggested positive effects in terms of BMD improvement at lower limb level [31, 33, 34].
While the effects of physical therapies on bone health are still controversial in the current literature [42, 43, 44], physical exercise is considered a non-pharmacological intervention supported by several guidelines in both osteoporosis prevention and management, due to its multitarget effects on the whole musculoskeletal system, improving both BMD and reducing the risk of falls [45, 46, 47]. Despite these considerations, few studies [31, 32, 36, 37, 38] assessed the effects of physical exercise on SCI patients. However, people with SCI could be affected by detrimental musculoskeletal impairment, with crucial implications in biological responses to physical exercise [48, 49]. Indeed, as part of normal bone metabolism in adult individuals, bone tissue undergoes a continuous process of resorption and formation; when this turnover is at equilibrium, it results in unchanged bone mass, while when there is unbalance, it results in bone changes (growth, aging) and pathology. More in detail, osteoclasts initiate bone resorption, osteoblasts are responsible for bone formation, while osteocytes are the cells dedicated at maintaining bone tissue [50]. On the other hand, as early as 1892, it was determined that mechanical loads in live beings can influence bone architecture. This concept is known as Wolff’s law, which was further explored till defining that loading on load-bearing bones determines the majority of their strength in adult life [51]. In this context, voluntary load bearing is lacking in SCI patients’ life. In this context, it has been suggested that spasticity might have a protective role in bone tissue in patients with neurological disability [52, 53], highlighting the close link between bone and muscle tissues due to their cross-regulation promoted by mechanical forces, in accordance with the mechanostat theory theorized by Frost [54]. On the other hand, it has been proposed that FES treatment might have positive effects on patients affected by spasticity [55]. Despite these considerations, our review underlined that no studies assessed spasticity combined with rehabilitation interventions to manage bone health in SCI patients and more in general the impact of spasticity on bone in these patients is far from being fully characterized.
On the other hand, muscle contraction has been targeted by different rehabilitative programs, including FES and vibration therapy, aiming at increasing mechanical stimuli on the bone and optimizing the protective role of muscle contraction on bone loss in these patients [31, 32, 35, 36, 37, 38].
In accordance with the International Osteoporosis Foundation, non-pharmacological intervention should be considered the first-line therapy in osteoporosis management [56]. However, in patients with a higher risk of fracture, a pharmacological approach is necessary to increase BMD and reduce the risk of fractures [57, 58]. In line with these findings, several studies included in the present review assessed the role of rehabilitation combined with both antiresorptive drugs (zoledronate [38]) and anabolic therapies (teriparatide [35] and testosterone [37]). The recent study by Dionyssiotis et al. [15] reported that level 1 supports intravenous zoledronic acid for preventing sublesional decline, while few studies are currently available about the effects of anabolic treatments for osteoporosis management in this specific population [15].
Lastly, a specific nutritional approach should be part of a comprehensive rehabilitation intervention, given the large consensus about the role of micronutrients in the management of bone health [57]. Despite this evidence, it was surprising to notice that just one study [35] assessed the effects of a combined intervention including rehabilitation and vitamin D
Interestingly, when consulting existing literature, previous reviews assessed the effects of rehabilitation interventions on bone health management of SCI patients. In particular, the review by Sutor et al. [10] assessed the effects of physical exercise and physical activity, providing interesting perspectives about the pathophysiology and molecular mechanisms underpinning physical activity positive effects on bone health of SCI patients. However, several studies with heterogenous designs were included and the authors did not apply a systematic approach. Similarly, the review by Chandrasekaran et al. [59] assessed the effects of FES in patients with SCI. Remarkably, the study highlighted the positive impact of FES in terms of muscle weight, muscle cross-sectional area, and physical functioning. However, the authors did not focus only on bone tissue modifications and the literature was assessed without a systematic approach.
Therefore, to the best of our knowledge, the present study is the first systematic review of RCTs assessing the effectiveness of different rehabilitation strategies to manage bone loss in SCI patients. Altogether, our data highlighted a large gap of knowledge about the potential role of rehabilitation interventions on bone health of SCI patients. On the other hand, a multitarget approach might further implement the synergisms between non-pharmacological and pharmacological interventions, with positive effects not only on bone health but also on functional outcomes of patients with SCI.
Despite these considerations, we are aware that this study has several limitations. In particular, the high heterogeneity of the study interventions limits to draw a quantitative synthesis of the study results. However, this is the first study addressing in a systematic way the effects of rehabilitation interventions on bone health management of SCI patients. In addition, this is the first systematic review assessing the effects of different rehabilitation modalities in this field, despite currently available literature does not allow to compare the effectiveness of different approaches. On the other hand, it should be noted that most of the studies included in the present systematic review showed some concerns in risk of bias assessment, highlighting the need for good quality studies assessing rehabilitation intervention in preventing bone loss of SCI patients.
Lastly, the etiology of SCI lesion, its level, and completeness might severely affect the rehabilitation outcomes, with potential sought of bias in the data synthesis. Therefore, further stratification might improve knowledge about the optimal therapeutic treatment tailored to patient’s characteristics.
Conclusions
Altogether, findings of this systematic review underlined that several rehabilitation interventions have been studied to improve bone health in patients with SCI, with promising results reported for physical exercise combined with FES and vibrations. However, several questions are still open about the synergisms of different rehabilitation strategies and the role of a specific rehabilitation approach combined with pharmacological management should still be characterized. Further good-quality studies are needed to improve knowledge about the optimal rehabilitation approach in SCI, improving not only functional outcomes but also bone health of these frail patients.
Ethical approval
Not applicable.
Funding
The authors report no funding.
Informed consent
Not applicable.
Author contributions
Conceptualization, L.L., A.d.S., and M.I.; methodology, L.L., A.d.S., and M.I.; investigation, L.L., A.F., and A.T.; writing – original draft preparation, L.L. and A.F.; writing – review and editing, A.d.S, and M.I.; figures, S.M.; visualization, C.C., S.M, and A.A.; supervision, A.d.S. and M.I. All authors have read and agreed to the published version of the manuscript.
Supplementary data
The supplementary files are available to download from http://dx.doi.org/10.3233//BMR-230006.
Footnotes
Acknowledgments
The authors want to acknowledge Enrico Cavallo for his support of this work.
Conflict of interest
The authors declare that they have no conflict of interest.