Abstract
BACKGROUND:
Central sensitization cannot be demonstrated directly in humans. Therefore, studies used different proxy markers (signs, symptoms and tools) to identify factors assumed to relate to central sensitization in humans, that is, Human Assumed Central Sensitization (HACS). The aims of this systematic review were to identify non-invasive objective markers of HACS and the instruments to assess these markers in patients with fibromyalgia (FM).
METHODS:
A systematic review was conducted with the following inclusion criteria: (1) adults, (2) diagnosed with FM, and (3) markers and instruments for HACS had to be non-invasive. Data were subsequently extracted, and studies were assessed for risk of bias using the quality assessment tools developed by the National Institute of Health.
RESULTS:
78 studies (
CONCLUSIONS:
This review provides an overview of non-invasive markers and instruments for the assessment of HACS in patients with FM. Implementing these findings into clinical settings may help to identify HACS in patients with FM.
Keywords
Introduction
The term nociplastic pain is defined as “pain that arises from altered nociception despite no clear evidence of actual or threatened tissue damage causing the activation of peripheral nociceptors or evidence for disease or lesion of the somatosensory system causing the pai” [1]. Central sensitization (CS) can be described as “an increased responsiveness of nociceptive neurons in the central nervous system to their normal or subthreshold afferent input” and can therefore be an expression of nociplastic pain [2]. CS results in an enhanced nociceptive neural signaling, meaning that the stimulus intensity necessary to elicit the pain response is lowered, resulting in pain hypersensitivity [3]. This alteration in sensory processing systems is observed in animal experiments [4], and this phenomenon is supposed to be of value to explain multiple chronic pain conditions such as low back pain, osteoarthritis, temporomandibular disorders and fibromyalgia (FM) [5]. FM, with a worldwide prevalence of 2–4%, and temporomandibular disorders are among the most common causes of pain and disability related to CS [6]. Both disorder share features and are influenced by genetic, biological, and psychosocial factors, such as diet, obesity and stressful events [6, 7, 8]. Because there is no gold standard for the assessment of CS, the presence of it cannot be demonstrated directly in humans. Instead, studies used different proxy markers (signs, symptoms and tools) to identify factors assumed to relate to CS in human, that is, Human Assumed Central Sensitization (HACS). A proxy marker for HACS can be defined as an indirect measurable indicator of the assumed presence of CS. These proxy markers will further be referred to as ‘marker’ in this review. The term HACS has previously been defined in a review on HACS in patients with chronic low back pain [9]. As FM is related to CS, rehabilitative therapy could play a useful role in the improvement of pain-related and mobility symptoms [7]. Thus, improving the accuracy of FM diagnosis can aid in the rehabilitation process of patients with FM.
Because of the absence of a typical physiological abnormality specific to FM, the diagnosis of FM is based on clinical presentation only, with a diagnostic criterium using a list of eighteen body sites and experiencing pain in at least 11 of the 18 tender points [10]. The 1990 American College of Rheumatology (ACR) classification for diagnosis comprises the assessment of pain in eighteen body sites combined with the average scores of a self-administered questionnaire [11]. The revised 2010 ACR classification includes a calculation of the widespread pain index, a symptom severity scale and does not contain a tender point examination. Diagnostic studies in patients with FM were conducted with the aim of identifying HACS markers, varying from cerebrospinal fluid (CSF) and serum concentrations [12, 13, 14] to urinary metabolites such as creatine [11]. Simple clinical tests to objectively identify HACS markers may, however, contribute to setting more suitable and objective diagnosis which are clinically feasible. While there appear to be several studies available, an overview of the current state is missing. The aim of this study was to review the literature to determine non-invasive markers for the presence of HACS in patients with FM and the instruments needed for the assessment of these markers.
Eligibility criteria for study selection
Eligibility criteria for study selection
The search strategy started with a broad search regarding non-invasive markers of HACS for three chronic musculoskeletal pain diagnoses: fibromyalgia, chronic low back pain and osteoarthritis. Due to the vast amount of hits provided by the search, the authors decided to split it in three parts. Therefore, the current study constitutes the first part of a larger review about pain processing in chronic musculoskeletal pain disorders and is focused on markers for HACS in patients with FM. A second part is focused on HACS in patients with chronic low back pain and a third study focusses on HACS in patients with osteoarthritis and other painful syndromes. The current systematic review was reported based on the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) [15] and has been prospectively registered in Prospero (October 2020: CRD42020172382).
Search strategy
Three electronic databases (PubMed, EMBASE and PsycINFO) were searched on 01/04/2022. MeSH terms in PubMed were incorporated in the search string. Keywords were divided into the three following categories: the target
Eligibility criteria
Flow diagram of study selection process. *Consider, if feasible to do so, reporting the number of records identified from each database or register searched (rather than the total number across all databases/registers). **If automation tools were used, indicate how many records were excluded by a human and how many were excluded by automation tools. From: Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ 2021;372:n71. doi: 10.1136/bmj.n71. For more information, visit: 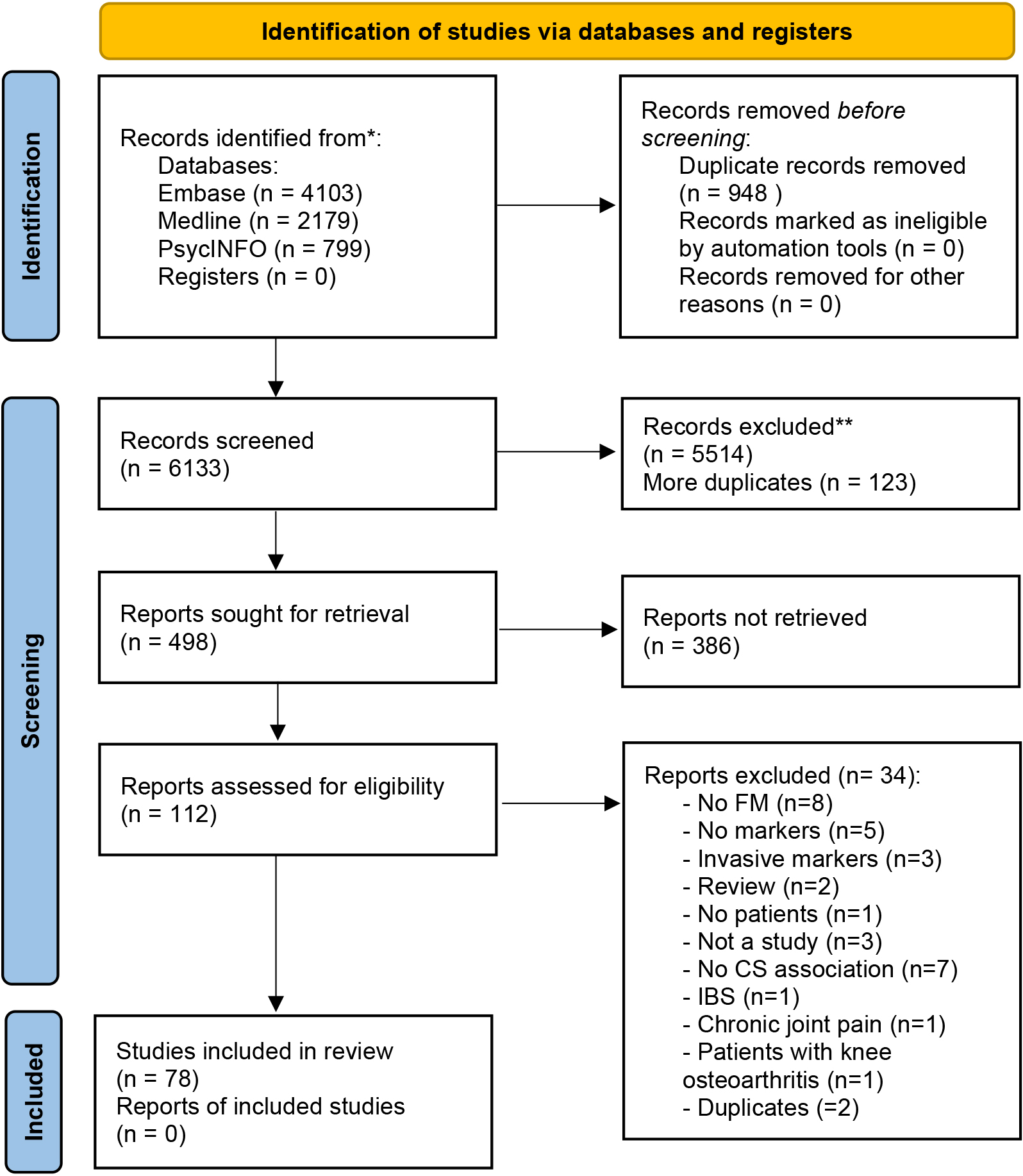
The eligibility criteria for the article selection are presented in Table 1. The following inclusion criteria were applied: (1) participants had to be adults (age 18 years or older; (2) patients had to be diagnosed with FM according to the American College of Rheumatology (ACR) criteria of 2010 and with the ACR criteria of 1990 for papers published before 201; (3) HACS markers had to be neurophysiological and non-invasiv; (4) the selected studies were published between 01/01/1994 and 01/04/2022. Articles were excluded if (1) included participants suffered from other forms of pain besides FM (but when patients with FM were compared to patients with other forms of pain besides FM these studies were included); (2) participants suffered from psychiatric comorbidity following specified DSM criteria; (3) the study designs were systematic reviews or meta-analyse; (4) the articles used only invasive markers such as blood tests.
The studies were screened (based on title and abstract) by three independent reviewers to exclude studies that were not specific to FM and the study aim. YS screened all, RS screened the first hal; and HT screened the second half. The reviewers subsequently selected articles for inclusion based on full text (
Data extraction
The data extraction process was performed by YS. Two researchers (RS and HT) reviewed the extracted data. The following information was extracted from each study and documented into a table: (1) the study (author names and publication date), (2) the population (number of participants with FM, number of healthy controls (HC) if applicable, age, gender and country, (3) study design, (4) aim of the study, (5) hypotheses, (6) inclusion/exclusion criteria, (7) assessment methods, (8) main findings and (9) definitions of HACS, nociplastic pain or hypersensitivity, when stated in the article.
Risk of bias and quality assessment
Quality assessment of the included articles was carried out using the National Institute of Health (NIH) Quality Assessment Tool for case-control studies, observational cohort and cross-sectional studies, and randomized controlled trials (RCTs) [16]. The NIH tool consists of 13 questions for case-control studies, 14 questions for cross-sectional studies and 14 questions for RCTs. Before assessing all the articles, YS, RS and HT first assessed 6 randomly chosen articles and then discussed it together to determine whether they all deduced the same understanding of the assessment questions. Possible answers for each question of the quality assessment were “ye”, “no”, “cannot determine, not applicable or not reported”. The answer ‘ye’ gave one point, whereas the other answers gave zero points to the study. An overall score between 0 and 13 for case-control studies or 0 and 14 for cross-sectional studies and RCT’s, was then calculated for each included study and the studies were subsequently judged as “good” (score of 75% or above), “fair” (score of 50–75%) or “poor” (score below 50%) quality [17]. Discussions between the three authors were held to solve any encountered disagreements.
The quality of the studies was taken into consideration when interpreting results. Markers identified from studies with a quality of at least ‘fair’ were interpreted as more reliable markers than those identified from studies ranked as ‘poor’ quality. Furthermore, conflicting outcomes from papers studying the same potential marker were considered as inconsistent results, consequently weighing the marker as ‘not valid’.
Study descriptives
The study descriptives of included articles are population (age and sex), country and number of included participants (patients and healthy controls). The results were divided into two main categories based on whether markers were detected by using measurements to assess peripheral or central manifestation of HACS.
Results
Search and selection
A total of 78 studies fulfilled the eligibility criteria (Table 1) and were included in this study. Peripheral manifestations of HACS include quantitative sensory testing. Central manifestations of HACS include electrophysiological techniques, conditioned pain modulation, pain anticipation and catastrophizing. Contrary to the peripheral manifestation of HACS, central manifestations are measurements of the CNS, such as brain perfusion using electrophysiological techniques and imaging.
Characteristics of included studies (
78)
Characteristics of included studies (
ACR: American college of rheumatology FM diagnosis criteria, AIC: anterior IC, ACC: anterior cingulate cortex, aMCC: anterior mid-cingulate cortex, APA: action potential amplitude, AUC: area under the curve, AS: pain after-sensation, ANS: autonomic nervous system, BP: blood-pressure, BOLD: blood-oxygen-level-dependent, BDI: Beck depression inventory, BP-PCS: Brazilian Portuguese Profile of chronic pain: screen, BPI: brief pain inventory, BRS; baroreflex sensitivity, BEI: baroreflex effectiveness, CC: cingulate cortex, C: cohort, cor: correlation, CLBP: chronic low back pain, CPM: conditioned pain modulation, CC: case control, CS: cross-sectional, CLP: chronic localized pain, CWP: chronic widespread pain, CSP: cutaneous silent period, CNS: central nervous system, CBP: chronic back pain, CLBP: chronic low back pain, CS: central-sensitization, C(D)T: cold (detection) threshold, CES-D: Center for Epidemiological Studies Depression Scale, CPT: cold pain threshold, CDC; centers for disease control, CFQ: Chalder fatigue questionnaire, CFS: chronic fatigue syndrome, CSS: central sensitization symptoms, CSI: central sensitization inventory, DM2: myotonic dystrophy type 2, DNIC: diffuse noxious inhibitory control, dif: difference, DLPFC: dorsolateral prefrontal, DEPS: depression scale, DBP: diastolic blood pressure, DBT: deep breathing test, DSM V: diagnostic and statistical manual of mental disorders, ES: explosive synchronization, EQ-5 L-5D: EuroQol The 5-level EQ-5D version, ED: electrodiagnostic, EDA: electrodermal activity, EPTT: electric pain tolerance threshold, EMG: electromyography, freq: frequency, FM: patients with fibromyalgia, fNIRS: functional near-infrared spectroscopy, FSS: fatigue severity scale, FP: frontopolar cortex, GMV: gray matter volume, HADS: hospital anxiety and depression scale, HbO: oxyhemoglobin, HC: healthy controls, HPT, heat pain threshold, HR: heart rate, HRV: heart rate variability, IBI: interbeat interval, ISI: interstimulus interval, IPL: inferior parietal lobule, IC: insular cortex, IPAQ: international physical activity questionnaire, IBS: irritable bowel syndrome, ILC: ipsilateral locus coeruleus, PDI: pain disability index, vs: compared to, pt: patients, y: years, w: weeks, VAS: visual analogue scale, LMP: local musculoskeletal pain, LOC: lateral occipital cortex, lPFC: lateral prefrontal cortex, MC: motor cortex, MPT: mechanical pain threshold, MPS: mechanical pain sensitivity, MVC: maximum voluntary contraction, MCV: medical college of Virginia pain questionnaire, MDT: mechanical detection threshold, MPQ: McGill pain questionnaire, MPFC: medial prefrontal cortex, MRS: multiple random staircase method, M1: primary motor cortex, NFR: nociceptive flexion reflex, NRS: numerical rating scale, NPQ: neuropathic pain questionnaire, OQS: Oviedo quality of sleep questionnaire, OFC: orbitofrontal cortices, stim: stimulation, OP: occipital pole, SF-MPQ: short-form of the McGill pain questionnaire, periph: peripheral, P-Ins: posterior insula, PPT: pressure pain threshold, PPI: present pain intensity, PPC: posterior parietal cortex, PS: peripheral sensitization, PAG; periaqueductal grey, PEP: pre-ejection period, PSQ-3: Pain and Sleep Questionnaire Three-Item, PASS: Pain anxiety symptom scales, PCS: pain catastrophizing scale, PrCG: pre-central gyrus, PCC: posterior cingulate cortex, Precun: precuneus, PL: paracentral lobule, PFC: pre-frontal cortex, QoL: quality of life, rCBF: regional cerebral blood flow, RLBP: recurrent low back pain, RCT: randomized controlled trial, ROI: region of interest, RSVP: rapid serial visual presentation, RVM: rostral ventromedial medulla, RMDQ: Roland-Morris Disability Questionnaire, ROC: receiver operator characteristics, SSR: sympathetic skin response, SREP: slowly repeated evoked pain, SPECT: single-photon emission computed tomography, signif: significant, S1&S2: primary and secondary somatosensory cortices, SMC: sensorimotor cortex, STPI: State-Trait personality Inventory (to assess anxiety), SF-36-PF: physical function subscale of the SF-36, SBP: systolic blood pressure, SPL: superior parietal lobule, STAI: State-trait anxiety inventory, SV: stroke volume, TS: temporal summation, , TSSP: temporal summation of second pain, T-T: threshold/tolerance, thal: thalamus, TMD: temporo-mandibular disorder, TPT: thermal pain threshold, cortex, TPR: total peripheral resistance, TSK: Tampa scale of kinesiophobia, VBM: voxel-based morphometry, VDT: vibration detection threshold, VGEE: generalized estimating equations, W(D)T: warm (detection) threshold, WU: wind up of pain, WU-AS: wind-up pain after-sensation, WPI: widespread pain index, +: positive, -: negative, r & l: right & left, ipsilat/contralat: ipsilateral/contralateral,
The study characteristics are shown in Table 2. In total, 2383 patients with FM, 1766 Healthy Controls (HC), and 1085 patients with other chronic pain conditions were included.
Peripheral manifestations of HACS were shown in the following studies: temporal summation of secondary pain (TSSP) and pain after-sensations (AS) were studied in eleven studies [18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28], the autonomic nervous system and slowly repeated evoked pain (SREP) sensitization were studied in five studies [29, 30, 31, 32, 33], quantitative sensory testing (QST) measures (heat, pressure and mechanical and sound ‘pressure’ pain thresholds) were used in thirteen studies [21, 23, 34, 35, 36, 37, 38, 39, 40, 41, 42, 43, 44] and the motor activity and FM was studied in four studies [36, 45, 46, 47].
Central manifestations of HACS were shown in the following studies: pain anticipation was studied twice [48, 49], conditioned pain modulation (CPM) was studied nine times [27, 29, 38, 43, 50, 51, 52, 53, 54], and three studies reported on the effect of distraction on pain [44, 55, 56], electrophysiological techniques were used in twenty-two studies [28, 44, 48, 49, 53, 55, 56, 57, 58, 59, 60, 61, 62, 63, 64, 65, 66, 67, 68, 69, 70, 71], laser-evoked potential (LEP) amplitudes were applied in four studies [72, 73, 74, 75], and brain region activation to stimuli and brain region connectivity were studied in sixteen studies [28, 33, 44, 60, 61, 62, 63, 64, 66, 69, 76, 77, 78, 79, 80, 81].
Risk of bias assessment of included studies (
78)
Risk of bias assessment of included studies (
Q: question, NR: not reported, NA: not applicable, CD: cannot determine. The quality of included studies was assessed using the National Institute of Health (NIH) Quality Assessment Tool for Case Control Studies (
Q: question, NR: not reported, NA: not applicable. The quality of included studies was assessed using the National Institute of Health (NIH) Quality Assessment Tool for Observational Cohort and Cross-Sectional Studies (
The quality of included studies was assessed using the National Institute of Health (NIH) Quality Assessment Tool for Controlled Intervention Studies (
The average risk of bias assessment scores was 39% for case control studies (
Measurements to assess peripheral manifestations of HACS
Quantitative sensory testing (QST)
Summary of temporal summation of second pain, pain after-sensation and pressure pain threshold findings
Summary of temporal summation of second pain, pain after-sensation and pressure pain threshold findings
TSSP: temporal summation of second pain, WU: wind-up of pain, FM: fibromyalgia,
Summary of electrophysiological technique findings in patients with fibromyalgia
FM: fibromyalgia,
Other markers of HACS include slowly repeated evoked pain (SREP) sensitization, the autonomic nervous system (ANS) response to pain measured with an electrocardiogram, electromyography and measures of the cardiovascular system [32], attentional task performance with concurrent pain stimuli [44], the relation between pain perception and motor activity and cutaneous silent period [96]. Patients showed SREP sensitization, measured as higher difference in VAS pain ratings between the last and first pain stimulation trials [30, 31] compared to HC. The stimulation trials consisted of a series of nine low-intensity pressure stimuli with 30 second interstimulus intervals. Patients also showed a positive correlation between clinical pain and SREP sensitization, and a lower pain threshold and pain tolerance compared to HC [31]. Patients demonstrated the need for longer interstimulus intervals (ISI) in order to perform an attentional rapid serial visual presentation (RSVP) task at 70% of optimal, compared to HC [44]. Furthermore, studies conducted with ECG on the ANS have shown that the abnormal ANS response to cold pressor tests in patients with FM is caused by lower effectiveness of the baroreflex responses, a homeostatic mechanism that helps to minimize considerable variations in blood pressure [29]. This was also demonstrated by lower baroreflex sensitivity (BRS) and baroreflex effectiveness (BEI) in patients with FM compared to HC during rest, as well as during the cold pressor test. Additionally, there was a positive correlation between BEI and heart rate variability (HRV). A negative correlation between BRS and BEI with cold pressor pain was also found, as well as between BEI and the pre-ejection period of the heart, the latter representing measures of the sympathetic influences on myocardial contractility. Furthermore, a reduced reactivity of blood pressure and cardiac stroke volume was demonstrated in patients compared to HC during the cold pressor test [32]. In contrast, one study showed no significant group difference in heart rate increase during cold pressor test but did underline a significantly higher heart rate in patients with FM compared to HC at rest [33]. Finally, one study demonstrated a negative correlation between motor activity and pain intensity [45]. There was a positive correlation between patients with FM who reported participating in regular physical activities and activity in pain regulatory regions of the brain (dorsolateral prefrontal cortex, posterior cingulate cortex, posterior insula) during painful stimulation [47]. On the contrary, lower PPT and higher pain ratings to pain stimulation during handgrip exercise was observed in patients with FM compared to HC [36]. Furthermore, a lower nociceptive withdrawal reflex threshold after stimuli in patients with FM compared to HC was shown in three studies [53, 57, 58]. Finally, a longer cutaneous silent period after painful electrical stimuli was measured in patients with FM compared to HC [96]. These findings are shown in Table 5 as well.
Measurements to assess CNS manifestations of HAC
Electroencephalogram (EEG)
Various electrophysiological techniques were used to assess HACS. Resting EEG measurements have shown a positive correlation between having FM and explosive synchronization conditions in patients with FM [59] (explosive synchronization being a condition in which a small perturbation leads to global propagation [59]). Furthermore, EEG has been helpful in demonstrating higher [72], lower [73] and identical [74, 75] amplitudes of the N1 and P2 components of laser-evoked potentials (LEP) in patients with FM compared to HC [72]. One study analyzed the biphasic N2P2 component of LEP’s and found an altered N2P2 habituation index in patients with FM [75] (habituation index representing whether or not subjects showed a decreased or increased response to stimuli repetition).
Brain activity and perfusion
Furthermore, fMRI was used to demonstrate higher mean amplitude of low-frequency fluctuations in the ventral hemicord, which turned out to be decreased in the dorsal quadrants of patient’ cervical spinal cord compared to HC [65]. Higher activation in fMRI-based neurologic pain signature regions was observed in patients with FM compared to HC during painful stimulation [61], indicating that this region is hyperactive in patients, suggesting to be an expression of HACS in patients with FM. fMRI also showed higher brain activity in different brain regions during identical pressure stimulation in patients with FM compared to HC [67]. When the stimulation intensity was adapted to create subjectively equal pain intensity for subjects in both groups, patients and HC showed similar fMRI activity [67]. This suggests that hyperactive regions can be an expression of HACS in patients with FM, as it was not activated in HC during identical pressure stimulation fMRI measures during TSSP, elicited by heat stimuli adjusted to individual’s pain threshold, also showed higher blood-oxygen-level-dependent (BOLD) activation patterns in the spinal cord of patients with and without FM. This activation also seemed to be associated with increased BOLD activity in the brainstem of patients with FM compared to HC [28]. Another study also demonstrated similar activation patterns in both groups after sensitivity-adjusted thermal stimuli, except for an increased activation of two brain regions in HC compared to patients with FM [44]. Furthermore, there was a positive correlation between the analgesic effect of the task and the BOLD activity detected on fMRI in both groups [44]. Brain perfusion analysis offered promising results using PET and SPECT neuroimaging. PET scan analysis showed increased regional cerebral blood flow (rCBF) bilaterally in the retrosplenial cortex (area that encodes sensory events, pain included) at rest in patients with FM compared to HC [68], increased rCBF in the parietal cortex and decreased rCBF in the retrosplenial cortex during painful stimulation compared to rest was also observed with PET scans in patients with FM [69], hyperperfusion in S1 and S2 areas of patients with FM was demonstrated using SPECT neuroimaging [70]. Functional near-infrared spectroscopy (fNIRS) measurements at the motor cortex (MO) showed greater hemoglobin-oxygen (HbO) concentration differences between two consecutive thermal stimuli in patients with FM compared to HC, suggesting a slower rate of cortical activation in the motor cortex of patients with FM [81]. In contrast, another study demonstrated a higher increase in HbO concentration in the left PFC between rest and cold pressor test in patients with FM compared to HC [33]. During CPT, patients with FM reached peak HbO concentrations faster than HC [33] and also demonstrated greater electrodermal activity amplitudes than HC [33].
Gray matter volume alterations
As final electrophysiological technique, voxel-based morphometry (VBM) analysis, showing gray matter volume alterations, yielded different results between FM patients and HC. Patients with FM presented with decreased grey matter volume in the anterior cingulate cortex [71] and increased grey matter volume in S1 bilaterally, compared to HC [60]. It was demonstrated that VBM-detected gray matter volume alterations in the anterior cingulate cortex are associated to HACS [71]. In contrast, the anterior cingulate cortex and amygdala volumetric changes are not associated with pain duration or functional disability. This suggests that these volumetric differences are not consequences of FM but could rather be a pre-condition for HACS development in FM [71], potentially making voxel-based morphometry a marker for HACS assessment. Additional results are displayed in Table 5.
Overview of the identified markers
Overview of the identified markers
*Markers identified from fair quality papers.
Pain during ascending (fingers first) and descending cold water immersion of the arm (elbows first) in HC and patients with FM was tested. One study demonstrated that HC felt less pain in their fingers during descending sessions compared to ascending, whereas patients with FM felt no difference [29]. Furthermore, it was demonstrated that patients with FM felt no changes in pain ratings after a pressure pain conditioning and a cold-water stimulation condition, whereas HC felt lower pain [50]. When comparing the efficacy of cold pressor test conditioning, one study [38] observed no CPM differences between both groups whereas another study [52] observed lower CPM efficacy in patients with FM compared to HC. When using tourniquet cuff conditioning, a study demonstrated that 95% of patients with FM showed inefficient CPM in comparison with zero HC cases [43]. However, lower PPT, HPT and higher pain ratings after a tourniquet cuff conditioning in patients with FM compared to HC were identified [51]. One study [53] observed that CPM decreases the nociceptive flexion reflex (NFR) amplitude in HC when painful conditioning was applied. However, in patients with FM, the nociceptive flexion reflex amplitudes were lower after applying non-painful conditioning CPM [53]. These findings are also shown in Table 5.
Pain anticipation and catastrophizing
Some studies were conducted on pain anticipation and catastrophizing in patients with FM. It was demonstrated that patients showed lower responses in the ventral tegmental area, a dopamine-rich region, during pain anticipation compared to HC [49]. It was shown that patients who were more prone to catastrophizing had a lower pain threshold with cuff algometry [48]. Loggia et al. demonstrated that patients displaying lower pain anticipation, showed reduced activity in the lateral prefrontal cortex (LPC). By means of mediation analyses, it was shown that this reduced activity mediates the hyperalgesic effect of catastrophizing [48]. Oliva et al. showed no difference in attentional analgesia during concurrent thermal painful stimuli, calibrated to each individual’s pain threshold, between groups: both groups demonstrated a decrease in pain score during the hard task compared to the easy task [44]. One study demonstrated lower blood-pressure and cardiac stroke volume reactivity during a mental arithmetic task in patients with FM compared to the reactivity of ANS parameters during the cold pressor test [32].
Table 6 shows an overview of the identified markers, with an asterisk next to the markers identified from fair quality papers.
Discussion
In this review, patients with FM showed differences on HACS markers compared to healthy subjects. The markers identified to assess
Measurements to assess peripheral manifestations of HACS
Peripheral assessments of HACS markers have provided inconsistent results. First of all, higher TSSP sensitivity in patients with FM compared to HC was shown in seven studies [19, 20, 23, 25, 26, 27, 28], with three of these studies being ranked fair quality [23, 27, 87], and four ranked poor quality [19, 20, 25, 26]. On the other hand, no TSSP difference between groups was found in two other studies [18, 21], with one study being ranked fair quality [21] and one with poor quality. From these findings, we cannot deduce that TSSP is a valid marker for the presence of HACS. However, the demonstrated higher pain after-sensation (AS) intensities [18, 26] and lower rates of pain AS decline [19, 20, 21] can support the suggestion to use them as markers for HACS in patients with FM. This is because HC showed opposite results and the higher pain sensation felt in patients with FM can be expressed through the higher pain AS intensities demonstrated in two studies [18, 26]. One study showed lower sound ‘pressure’ pain thresholds in patients with FM compared to HC, further expanding the noxious sensation spectrum of patients with FM to auditory mechanisms [42]. Regarding measurements of HPT and CPT (thermal sensory devices) [21, 34, 37, 39, 41] in patients with FM, one study [21] did not observe thermal pain threshold differences between patients with FM and HC. From these findings, and considering the fact that the study was only ranked fair quality, we cannot undoubtedly classify thermal pain thresholds as a usable marker for HACS identification in patients with FM. Lower MPT detected with pin-prick stimulators [34] in patients with FM compared to HC showed to be a promising tool for pain hypersensitivity detection in patients with FM. It is important to note that two of these studies [31, 41], were ranked fair quality. Furthermore, authors of several studies [23, 31, 34, 35, 36, 37, 38, 41, 42, 43, 44] demonstrated PPT measurements with pressure algometry to indicate the presence of hyperalgesia in patients with FM. However, one study found SREP specificity to be 25% higher (0.92) and sensitivity 4% higher (0.79) for discriminating between patients with FM and HC compared to PPT measurements (PPT 0.67) [31]. This may indicate that SREP, evoked by a series of pressure stimuli, is a better marker to discriminate for HACS between patients with FM and HC compared to PPT.The increased cutaneous silent period (CSP) duration after stimulation of the cutaneous nerve in patients with FM compared to HC represents a faster conduction of pain and longer period of sustained muscle contraction in patients [96]. This may suggest the effectivity of CSP as a peripheral marker for altered pain sensitization in FM.
By means of ECG, measuring ANS responses to cold pressor tests could be used in a clinical setting to detect abnormalities in the baroreflex responses in patients [29, 32]. The reduced baroreflex sensitivity and effectiveness during cold pressor test in patients with FM can be a manifestation of altered CNS activity. Additionally, the demonstrated reduced heart rate variability could be a result of a decreased baroreflex function in patients with FM [32]. Even though another study [33] showed no difference in heart rate increase during cold pressor test between both groups, results still indicated a higher heart rate and lower heart rate variability in patients with FM compared to HC at rest. Furthermore, the results on the correlation between pain modulation and motor features are inconsistent [36, 45, 46, 47], and we can therefore not deduce that assessing pain modulation during motor activity can be regarded currently as a valid marker.
Measurements to assess central manifestations of HACS
Feasibility of measurements of central manifestations of HACS in patients with FM was shown in several studies [28, 43, 44, 48, 49, 53, 55, 56, 57, 58, 59, 60, 61, 62, 63, 64, 65, 66, 67, 68, 69, 70, 71]. However, except for three [49, 61, 87], these studies were qualified as poor quality. Furthermore, an explosive synchronization network is a network where a small perturbation rapidly propagates throughout the whole network. Explosive synchronization (ES) networks detected with EEG have shown to elicit higher sensitivity to external stimulation than non-ES networks [59]. Lee et al. [59] concluded that the presence of ES conditions in the brains of patients with FM can be an underlying mechanism of hypersensitivity. ES conditions may thus be a potential marker for HACS. Studies on LEP, on the contrary, yielded inconsistent results. Therefore, it cannot be concluded that LEP amplitude analysis with EEG is a valid method to assess HACS in patients with FM [72, 73, 74, 75].
Conditioned pain modulation (CPM) can be described as a painful conditioning stimulus leading to decreased pain intensity of another noxious stimulus [97]. The reduced NFR after non-painful conditioning (mechanical stimulation) in patients with FM points towards the presence of altered pain inhibitory pathways [53]. However, this was performed in a poor-quality study, which suggests that NFR amplitude measured with EMG after the application of a painful conditioning cannot be considered a valid marker for HACS detection. One additional study showed the positive effect of attentional analgesia in patients with FM, putting forward the capability of patients with FM to modulate pain when the given stimulus is sensitivity-adapted and the attentional task difficulty is correctly calibrated [44].
fMRI was used in eleven studies [28, 44, 48, 49, 61, 62, 63, 64, 65, 66, 67] to examine various aspects of HACS in patients with FM. The variations of mean amplitude of low-frequency fluctuations (ALFF) in patients with FM indicate an imbalance between pain and sensory processes [65]. This suggests the presence of altered central nervous system activity in patients with FM [65]. fMRI showed increased connectivity between various brain regions in patients with FM compared to HC [63, 64, 66], reflecting the expression of HACS in pain processing mechanisms in patients with FM [63]. Furthermore, increased brain activation of pain-related areas during non-painful stimulation in patients with FM indicate physiological evidence of their increased pain perception [62]. Similar patterns of brain activation after sensitivity-adjusted painful stimuli in both groups also suggest increased pain sensitivity in patients with FM [28, 44]. The same study found a positive correlation between spinal activation during TSSP and increased BOLD activity in the brainstem, suggesting a different pain modulation mechanism in patients with FM [28]. The higher neurologic pain signature (NPS) responses, an fMRI-based neurologic correlate of physical pain, provides evidence of amplified pain processing and HACS in patients with FM [61]. These studies help us conclude that fMRI is a useful tool to help indicate the following markers of HACS in patients with FM: amplitude of low-frequency fluctuations (ALFF) variations, brain activity and connectivity differences, neurologic pain signature responses and pain anticipation dysfunction. On the other hand, rCBF variations indicate patient’ higher attention to innocuous sensory signals at rest. These findings make PET and SPECT imaging potential tools for the investigation of brain perfusion abnormalities [68, 69, 70]. Lastly, two studies [43, 51] point out the potential role of CPM assessment with tourniquet pain conditioning as a marker for HACS in patients with FM [51].
All taken together, seventy-four studies were ranked as poor or fair quality. Those studies indicate a high risk of bias, which should be taken into consideration when interpreting results. The markers identified from studies ranked as poor quality cannot be determined as being as valid as markers identified in higher quality papers. Out of the markers identified in this review, the following were suggested from at least fair quality papers: higher SREP sensitization (elicited by pressure stimuli) [30, 31], NPS response detected with fMRI [61], lower sound ‘pressure’ pain thresholds [42], brain perfusion differences [81, 87] and conditioned pain modulation with cuff pressure conditioning [51]. The lower pain AS decline rates were suggested from three papers [19, 20, 21], out of which only one is fair quality [21].
A limitation to this review is the fact that due to the heterogeneity of the studies, especially in the vast number of markers, measurements and differences in study protocols, a meta-analysis could not be conducted. The current study has implications in the clinical setting, because these findings can be utilized to construct a more objective diagnostic protocol for HACS assessment in patients with FM. Furthermore, assessing HACS development over time as a proxy for disease progression in the day-to-day clinical practice may be valuable. Questionnaires combined with a short battery of objective tests, grouping the markers and their respective tools could be a solution to objectively quantify patients pain markers. Markers that are best executable and affordable in daily practice are tourniquet cuff pressure conditioning [51] and pressure stimuli, the latter being derived from a fair quality paper [31]. By assessing these markers, HACS may be more objectively quantified Additionally, the diagnosis of HACS development over time can also be combined with methods which do not require questionnaires or markers [10]. Physicians could strengthen the diagnosis by assessing amplified pain distribution (number of pain regions and/or pain intensity per region) compared to previous assessments. This will ultimately help to make a personalized treatment plan for daily clinical practice. Studies have shown that the implementation of physical and pharmacological therapy in patients with temporomandibular disorders has led to the reduction of pain- and mobility-related symptoms [6]. Hence, FM, as an overlapping chronic pain disorder with relations to central nervous system dysfunction due to HACS, could also benefit from physical therapy for the rehabilitation of HACS and, as result, for the improvement of pain. Further research, however, is warranted to validate these hypotheses. It is important to note that there is currently no single test or gold standard that can identify patients with HACS and that a combination of different measurements could formulate a gold standard, possibly also combined with more invasive markers which were left outside the scope of the current study.
Conclusion
The current study identified non-invasive markers for peripheral manifestations of HACS in FM including quantitative sensory testing measurements and nociceptive flexion reflex assessment. This study also revealed that various techniques can be used to assess the aforementioned HACS. Among them are markers such as EMG for the assessment of nociceptive flexion reflex. Lastly, conditioned pain modulation by tourniquet cuff pain conditioning and techniques such as EEG, PET, SPECT, fMRI and VBM were also identified to be useful in the assessment of central manifestations of HACS. More studies should be conducted in order to determine which markers can clinically be used to identify HACS in patients with FM.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Competing interests
None to declare.
Supplementary data
The supplementary files are available to download from http://dx.doi.org/10.3233/BMR-220430.
