Abstract
BACKGROUND:
Knee osteoarthritis (OA) is a common and disabling disease among the elderly population. The optimal conservative treatment for knee OA is not well established.
OBJECTIVE:
This study aimed to assess the effectiveness of pulsed electromagnetic field (PEMF) combined with progressive resistance exercise (PRE) in improving physical function and pain in patients with knee OA.
METHODS:
Thirty-four patients with knee OA (17 in each group) participated in a single-blind randomized control study. Patients were randomly assigned to receive 24 sessions of either combined PEMF and PRE (treatment group) or PRE only (control group). Patients were evaluated at pre-treatment, post-treatment (2 months), and at 3-month and 6-month follow-ups using the Knee Injury and Osteoarthritis Outcome Score (KOOS), Numeric Pain Rating Scale (NPRS); walking speed and 5-times chair stand test. Mixed ANOVA was used for statistical analysis with Bonferroni adjustments.
RESULTS:
There was no significant group-by-time interaction for any outcome (
CONCLUSION:
Both treatment options, PRE only versus PRE with PEMF, were equally effective in decreasing pain and improving physical function in patients with knee OA. This would suggest that the optimal parameters for PEMF that may show beneficial effects for knee OA when added to PRE training need to be determined.
Introduction
Osteoarthritis (OA) is one of the main causes of disability in the elderly population in the United States [1, 2]. According to Shamekh et al., knee OA was the most prevalent type of OA in the Middle East and North Africa (MENA) region, with an incidence peak in the 45–49 age range. In addition, knee OA, which peaked in the 75–79 age range, was the primary cause of years lived with disability (YLDs) (per 100,000 population) [3]. Although osteoarthritis is not directly fatal, it is believed that compromised muscle strength and functional capacity contribute significantly to morbidity and reduced life expectancy in patients with knee OA [4].
The main goals of knee OA interventions are reducing knee pain, improving physical function and quality of life, and minimizing cartilage deterioration and disease progression. The treatment options for knee OA include non-pharmacological (conservative), pharmacological, and surgical procedures [5, 6]. Current clinical guidelines for treating patients with knee OA recommend starting with conservative non-pharmacological treatment initially, this includes physical exercises that are relatively low in cost and have low side effects [5, 6]. A large body of evidence supports the benefit of physical exercise as a treatment option for patients with OA [7, 8, 9]. Additionally, the American College of Rheumatology recommends using strengthening exercises for treating patients with knee OA symptoms [10, 11]. Yet, patients with knee OA are required to perform progressive resistance exercise training, at their maximum capacity, to ensure an optimal strength gain that leads to better outcomes.
Pulsed electromagnetic field (PEMF) is a non-invasive physical therapy modality recommended by some physicians to treat patients with knee OA. In animal models, PEMF has been suggested to be effective in reducing chondrocyte apoptosis in cartilage [12, 13] and increasing morphogens that stimulate osteogenesis [14, 15]. Furthermore, PEMF has been shown to increase extracellular matrix production and reduce the release of proinflammatory cytokines and chondrocyte apoptosis [16, 17, 18, 19]. Decreased production of proinflammatory mediators has been advocated to be the mechanism via which PEMF modulates chondrocyte death. Despite conflicting evidence regarding the beneficial effect of PEMF [20, 21], a large number of studies suggested that PEMF is beneficial in reducing pain and improving function in patients with knee OA [22, 23, 24]. One systemic review [23] and two meta-analyses of randomized placebo-controlled trials indicated that PEMF is effective in improving function in patients with knee OA at 4–6 weeks and 8 weeks after the initiation of treatment compared to placebo [25, 26], with no adverse events associated with using PEMF [23]. However, the effect size of the PEMF benefit is considerably small. This may suggest that other therapeutic modalities may contribute to improving the outcomes in patients with knee OA such as progressive resistance exercise training. The reported small effect size of the PEMF may have resulted from using different types and dosages of PEMF [27]. While PEMF has been reported to be more effective in managing patients with knee OA than placebo in the short term according to some trials [25, 28, 29, 30, 31, 32], PEMF usage is expensive for patients and the healthcare system. Currently, no study has investigated the effect of adding PEMF to progressive resistance exercise (PRE) training on knee symptoms and functional activities in patients with knee OA. Therefore, the purpose of this study was to examine the additive effect of pulsed electromagnetic field to progressive resistance exercises on pain level, patient-reported measures, and performance-based physical function in patients with knee OA. It was hypothesized that patients with knee OA treated with combined interventions (PEMF and PRE) will show better outcomes in pain and physical function than patients treated with PRE only.
Methods
Study design
In this randomized control trial, patients were randomly assigned to receive an 8-week (3 sessions/week) combined PEMF and PRE (treatment group) or PRE only (control group). Patients with unilateral or bilateral knee OA as defined by the American College of Rheumatology [33], and with documented radiographic evidence of knee OA were recruited from Princess Basma Educational Hospital. Patients with mild to moderate knee OA who were referred for physical therapy by their physicians between April and October 2019 were invited to participate in the study. Patients who accepted the invitation and were willing to participate in the study were interviewed by a licensed physical therapist to evaluate their eligibility. All participants provided informed consented before study participation. The study protocol was registered at clinical trial.gov under trial registration number NCT04106986.
Subjects
Inclusion criteria: All patients aged 40 years or older and who have been diagnosed with mild to moderate unilateral or bilateral Knee OA were eligible to participate in this study. Mild and moderate Knee OA were determined based on physicians’ diagnoses. The physician used the American College of Rheumatology criteria [34] for determining symptomatic knee OA and Kellgren-Lowrence scores for determining the severity (Mild
Exclusion criteria: Patients with current or previous knee surgeries; inflammatory arthritis (rheumatoid arthritis, gout, etc.); Alzheimer’s disease; Parkinson’s disease, or unable to walk unaided for 6 months were ineligible to participate in this study. To maximize the efficiency of our experiment, patients who participated in progressive resistance training or received PEMF treatment in the prior year, or performed exercise regularly more than once a week were also excluded.
Sample size calculation
The sample size was determined using G*Power software v 3.1.0 (University Düsseldorf, Düsseldorf, Germany). The minimal detectable change (MDC) for the primary outcome measure Knee injury and Osteoarthritis Outcome Score (KOOS) from a previous study was used to determine the required effect size for mixed-design analysis [36]. The MDC has been recommended by other studies as a useful metric for sample size determination when the information about the intended sample size is lacking [37, 38]. To detect a change of 20 points between the treatment and control groups with a significant level of 0.05 and 80% power, the power analysis indicated that a total of 32 patients (16 in each group) was needed.
Materials
PEMF treatment was administered using the Pagani magnetic therapy table (PMT) by Electronica Pagani, Italy. The PEMF generator is controlled by a microprocessor that manages 3 independent channels: each channel has 2 outputs to which 4 solenoids and 2 Flexa applicators are connected. The PMT device applied the magnetic fields parallel to the longitudinal axis of the body segment through the solenoids, or perpendicularly to the surface of the treated segment through the Flexa applicators, the patient is in the supine position. PEMF was applied for 30 minutes per session at a 50-Hz frequency, 50-G intensity, and 90-sec interval. The PEMF dosage and duration used in this study replicates the treatment protocol suggested by We et al., which suggested the application of 8 weeks of 30-minute PEMF is beneficial [23].
Treatment protocol
The treatment group (PEMF and PRE) received 3 sessions per week for 8 weeks of 30-minute PEMF and 45 minutes of PRE training on the leg press machine per session [39]. While the control group received only 3 sessions per week for 8 weeks of 45-minute PRE training. The amount of resistance and the number of repetitions for each set of the PRE training were determined based on a modification of the daily adjustable progressive resistance exercise (DAPRE) program described by Knight et al. [40]. The DAPRE program was chosen to ensure that patients performed the PRE at their optimal capacity during each session, thus ensuring optimal strength gains. For each exercise, the patients performed 4 sets of repetitions. During the 1st set, patients performed 10 repetitions at 50% of the established working weight and the 2nd set consisted of 6 repetitions at 75% of the working weight. During the 3rd set, patients performed as many repetitions as possible at 100% of the working weight, and the number of repetitions performed during the 3
After the strengthening exercises, participants performed stretching exercises to minimize muscle soreness, and cold was applied for 10–15 minutes to the knee to minimize knee pain and swelling. The amount of resistance and number of repetitions were recorded for each set and session. To enhance internal validity, all participants were asked not to alter their regularly prescribed OA medication or their physical activities during study participation.
Procedure
Patients were tested at pre-treatment, post-treatment (2 months), and at 3-month and 6-month follow-ups.
Primary outcome measures
The primary outcomes were KOOS, 4-meter (4-m) walking speed, and the 5-times chair stand test. The KOOS is a knee-specific instrument, developed to assess the patient’s opinion about their knee and associated problems [41]. The KOOS evaluates both short-term and long-term consequences of a knee injury. It holds 42 items in 5 separately scored subscales: pain, other symptoms, function in daily living (ADL), function in sport and recreation (Sport/Rec), and knee-related quality of life (QOL). The KOOS total score and KOOS subscales scores were computed and analyzed in this study. The Arabic version of KOOS is valid and reliable for patients with knee OA with excellent internal consistency (Cronbach’s alpha
The time spent walking 4 meters was recorded in seconds (4-m walking speed) [43, 44]. Patients also performed chair-stand tests [45, 46]. In this test, patients were sitting in a chair with a standard height and without armrests, and then they were asked to stand up to the upright position. The test was repeated 5 times and the time required to complete the chair stand test was measured in seconds with the mean time of the 5 trials computed. Both the walking speed and chair stand tests have demonstrated good intra- and inter-reliability, responsiveness, and validity [43, 45, 46].
Secondary outcome measure
The secondary outcome of this study included the numeric pain rating scale (NPRS). NPRS is an 11-point scale comprising a number from 0 through 10; 0 indicates “no pain”, and 10 indicates the “worst imaginable pain”. Patients were instructed to choose a single number from the scale that best describes their pain level [47]. The Arabic version of NPRS was used in this study and it has shown good to excellent test-retest reliability and validity for patients with knee OA [48].
Randomization procedure
The principal investigator (PI) of this study used computer software to produce a sequence of allocation based on simple randomization. Each patient was given a unique code. This code was sequential (e.g., 01,02,03,04) to generate a randomization plan. The randomization was concealed using opaque envelopes. The PI monitored the implementation of randomization and ensured that a single-blinded assessor who conducted the study tests was kept masked and blinded to treatment design throughout the study period. The assessor was a senior physical therapist that works in a different clinic and came only to the study location to complete the testing procedure. The PI also was blinded to patients’ testing results until the study was completed.
Ethical considerations
Institutional Review Board approval from Jordan University of Science and Technology and King Abdullah University Hospital was obtained before conducting the study (approval no. 43/120/2019). Detailed information about the study benefits and risks of participation was explained to eligible patients before they were enrolled in the study. Written informed consent was obtained from all study participants. The trial of this study was registered at clinicaltrial.gov under registration number NCT04106986.
Statistical analysis
Independent
Results
Forty-one patients were screened for eligibility, of which 34 patients participated in this study. Patients were randomized to receive combined PEMF and PRE (treatment group (
Patients’ demographic and baseline characteristics of treatment and control groups
Patients’ demographic and baseline characteristics of treatment and control groups
Mixed ANOVA analysis for KOOS Total Score for Pre-treatment, Post-treatment, 3-month, and 6-month Follow-up of Both Groups (Mean (SD))
Table 1 represents demographic information of the study sample distributed by groups. The average age for PEMF and PRE and PRE-only groups were 59.7
There was no significant time-by-group interaction for the total KOOS score (F
Mixed ANOVA Analysis for KOOS Sub-scales (Symptoms, Pain, Activity of daily living, Sport, and Quality of life) at Pre-treatment, Post-treatment, 3- month, and 6-month Follow-up for Both Groups (Mean (SD))
Mixed ANOVA Analysis for KOOS Sub-scales (Symptoms, Pain, Activity of daily living, Sport, and Quality of life) at Pre-treatment, Post-treatment, 3- month, and 6-month Follow-up for Both Groups (Mean (SD))
Mixed ANOVA Analysis for Walking Gait Speed and 5-time Chair Stand Test at Pre-treatment, Post-treatment, 3- month and 6-month Follow-up for Both Groups (Mean (SD))
Flow diagram of the study.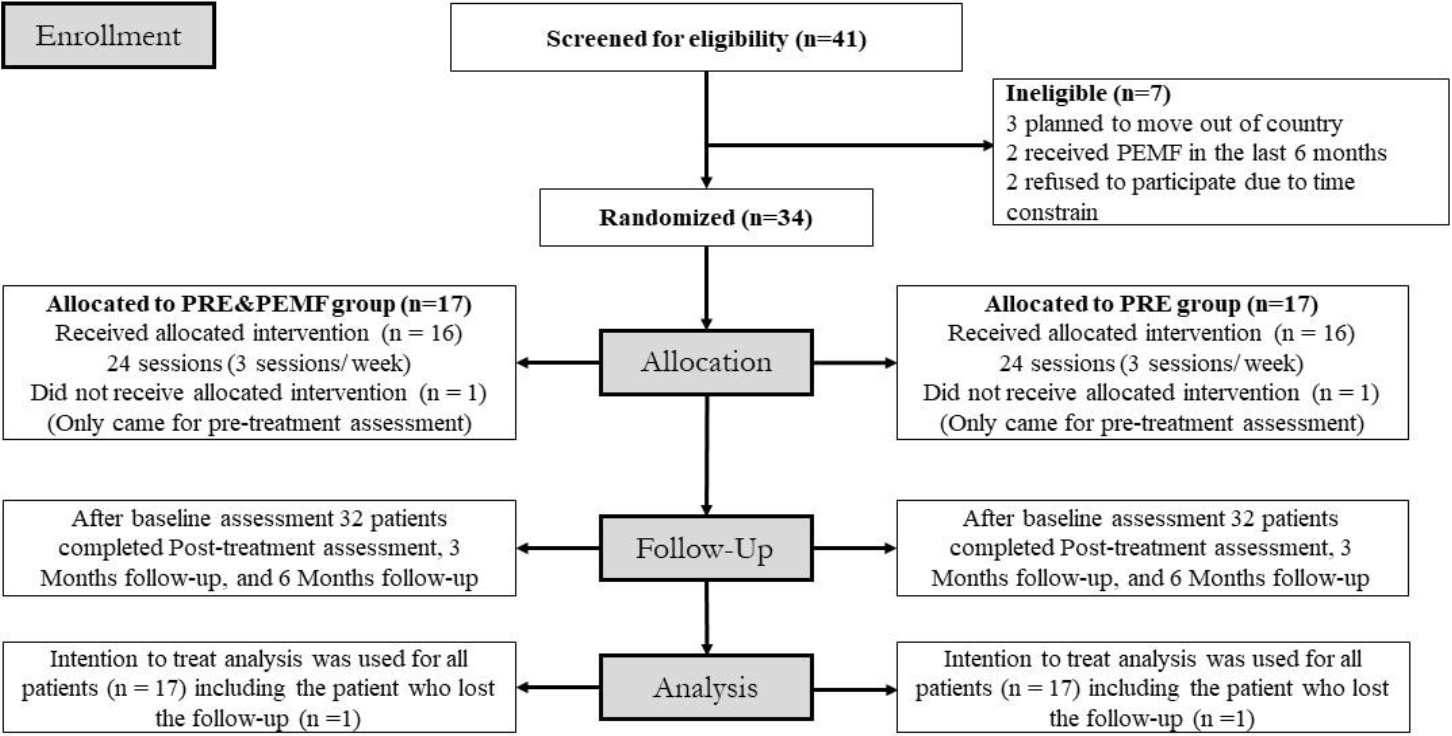
There was no significant group-by-time interaction for all KOOS sub-scales. However, there were significant main effects of time for all KOOS sub-scales (
Mixed ANOVA Analysis for NPRS at Pre-treatment, Post-treatment, 3- month, and 6-month follow-up for both groups (Mean (SD))
NPRS: Numeric Pain Rating Scale;
Regarding walking speed, results showed no significant group-by-time interaction (F
For NPRS, no significant group-by-time interaction appeared for NPRS (F
Discussion
In this randomized controlled study, the effectiveness of combined PEMF and PRE was compared to PRE only on knee pain, patient-reported, and functional measures in patients with knee OA. The findings of this study showed no differences between groups and indicate that adding PEMF to the PRE to treat patients with knee OA induced no further benefit in reducing knee pain intensity or improving functional performance. However, both groups decreased their knee pain, scored higher on patient-reported measures, and improved their performance at all follow-up time points.
The findings of the current study failed to approve our hypothesis. The PEMF is thought to play a significant role in improving the articular cartilage vitality and micro-structures, which may help reduce joint pain and improve the patient’s functions. It is advocated that magnetotherapy induces physiological changes characterized by increased oxygen release from erythrocytes, thereby ensuring tissue oxygenation. Additionally, magnetotherapy helps in accelerating the blood flow, vasodilation, and change in blood ion content at the application area [32]. These changes lead to a decrease in toxins in the damaged area and increases in vital nutrients and endorphins levels, which in turn reduces the nociceptor sensitivity that helps with patient symptoms. Despite the previously mentioned benefits of using PEMF, the optimal treatment protocol of PEMF for knee OA is still unclear.
The findings of the current study are not in agreement with several previous studies that reported on the effect of PEMF in managing patient’s symptoms in patients with musculoskeletal conditions including joint OA [28, 29, 30, 31, 32].
Bagnato et al. reported improvement in pain reduction and physical function in a group of patients with knee OA using PEMF compared to a placebo group [28]. However, patients in Bagnato’s study received PEMF treatment for only 6 weeks, with 12 hours a day as opposed to the intervention group in this study who received PEMF treatment for 8 weeks, 3 times a week, for 30 minutes each. In a meta-analysis study, Wu et al. examined the effectiveness of PEMF compared to sham treatment for knee OA [29]. They reported that patients who received PEMF for 30 minutes improved their knee pain and physical function.
In another study by Thamesborg et al., patients under 65 years old with knee OA who received PEMF treatment improved their knee pain and stiffness during ADL [30]. The contradicting findings between this study and those studies that showed a beneficial effect of PEMF might be attributed to the differences in the time duration of the PEMF treatment sessions. However, in this study, the time duration of treatment sessions was shorter in length which falls within the time range used in the modern physical therapy settings. Another factor attributed to the contradicting findings between studies could be the variation in parameters that were used for PEMF treatments. In which, the frequency, intensity, and duration of PEMF used in our study may have not been the optimal parameters to induce additive benefits in patients with knee OA who received PRE training. Therefore, further work might be needed to investigate whether the PEMF treatment is effective in the treatment of patients with knee OA using different parameters and to determine the appropriate parameters that might provide optimal effects. The current study also included a control group that received the PRE which has a well-determined treatment effect as compared to the sham or placebo treatment used in the previously mentioned studies. Therefore, the PRE may have influenced the findings of the current study as it could overcome the benefit of PEMF in the intervention group. The findings of the current study do not negate the effect of the PEMF treatment for knee OA. However, when PEMF is used in adjunct to the PRE training for managing patients with knee OA, it does not add any additional benefits.
Several studies reached to the same conclusion as our data showed that PEMF was not statistically beneficial [51]. Ozguclu et al. found no additive effect of PEMF when added to traditional physical treatment for managing patients with knee OA that includes hot packs, therapeutic ultrasound, and isometric quadriceps exercises [52]. Ay et al. also found no additive effect of PEMF on a physical therapy program that included hot-pack, transcutaneous electrical nerve stimulation, and isometric quadriceps training program [53]. Additionally, Fary et al. [29] found no significant effect of using PEMF when added to a standard Knee OA treatment program. These studies along with our study support the notion that the benefit of PEMF is diminished when it is used in combination with other treatment modalities for managing patients with knee OA.
This study is the first randomized control trial that investigated the effectiveness of PEMF combined with PRE versus individually tailored progressive resistive exercises. The study showed no statistical differences between the two groups. This would suggest that when the PRE training is used for managing patients with knee OA, adding the PEMF treatment does not improve patients’ symptoms and functions. Therefore, PEMF might not be used in combination with PRE training. Further, the findings of the current study may raise a clinical concern about the use of passive, yet expensive, modalities for treating patients with knee OA, when it would be preferable to encourage them to engage in physical activity and therapeutic exercises.
It appears that strengthening the lower extremity musculature in patients with knee OA may improve the patient’s ability to perform physical activities and lower the knee pain which can contribute to improving the quality of life in the elderly population that suffers from knee OA [54, 55, 56, 57, 58, 59]. Progressive resistance exercise training in the elderly population has been shown to improve the maximum voluntary activation of the muscle fibers and increase the individual muscle fiber area (type I and type IIa) and the total muscle cross-sectional area [60]. This may, in turn, result in long-term relief by allowing patients to perform these exercises in an independent rehabilitation program which is much more cost beneficial.
Our study has some limitations that need to be acknowledged. The sample size was small with 34 patients participating in the study. However, a priory sample size calculation was performed and indicated that a sample of 32 participants would be powerful to detect significant changes between groups. Additionally, a longer follow-up time is needed to determine the long-term benefits of the PRE training program. Furthermore, the groups in the current study were not equal in terms of gender at baseline which may have affected the study results. However, none of the previous studies has shown that patients with knee OA responded differently to treatment based on their gender. Lastly, classifying patients according to the disease severity was not possible due to the small sample size and may have impacted the results.
Future studies with larger sample sizes are needed to allow for more subgroup analysis and comparisons (i.e., comparisons between different disease severity and age subgroups). Additionally, researchers may use different outcome measures including knee ROM, muscle strength testing, and physical activity that require one limb to perform to better determine the effect of PEMF treatment in patients with knee OA. Furthermore, future studies that aim to determine the optimal PEMF parameters and duration are still needed.
Conclusion
This study showed that both treatment groups for knee osteoarthritis, individually tailored PRE versus combined PRE with PEMF, were equally effective in decreasing pain and improving physical function. This would suggest that the currently used parameters for PEMF are not optimal to show beneficial effects in patients with knee OA when added PRE training.
Ethics statement
This study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). The study was approved by the Institutional Review Board of Jordan University of Science and Technology and King Abdullah University Hospital (approval no. 43/120/2019). Written informed consent was obtained from all study participants before study participation.
Author contributions
MY: Concept and design of the trial, analysis, and interpretation of data, drafting of the article, generation of tables, critical revision of the article for the importation of intellectual content, and final approval of the article. AA: Collection of data, drafting of the article, critical revision of the article for important intellectual content, and final approval of the article. ZN: Concept and design of the trial, interpretation of data, drafting of the article, critical revision of the article for important intellectual content, and final approval of the article. SO: Interpretation of data, drafting of the article, critical revision of the article for important intellectual content, and final approval of the article. IA: Analysis and interpretation of data, drafting the article, critical revision of the article for important intellectual content, and final approval of the article. KB: Concept and design of the trial, drafting of the article, critical revision of the article for important intellectual content, and final approval of the article.
Funding
The study was internally funded by the Jordan University of Science and Technology (grant no. 20190071).
Footnotes
Acknowledgments
The authors would like to thank all study participants and Dr. Ali Al-Rjoub and Dr. Waleed Almomani for clinic facilitation and support.
Conflict of interest
All authors certify that they have no affiliations with or financial involvement (consultancies, stock ownership, equity interest, patent/licensing arrangements, etc.) in any organization or entity with a direct financial interest in the subject matter or materials discussed in the article.
Appendix A
Change in working weight for fourth set and next session based upon number of repetitions performed in the DAPRE program
Number of repeti-
tions performed
during 3rd set
4th set
Next session
Decrease by 2kg
No change
5–7 reps
No change
No change
8–12 reps
Increase by 2–5 kg
Increase by 2–7 kg
Increase by 5–7 kg
Increase by 5–10 kg
