Abstract
Kidney-sparing surgery (KSS) for upper urinary tract urothelial carcinoma (UTUC) is a promising alternative to radical nephroureterectomy, especially for low-risk cases. However, due to the established risk of ipsilateral UTUC recurrence caused by the implantation of floating neoplastic cells after endoscopic resection, adjuvant endocavitary (endoureteral) instillations have been proposed. Instillation therapy may be also used as primary treatment for UTUC. The two most studied drugs that have been evaluated in both the adjuvant and primary setting of endocavitary instillation are mitomycin C and Bacillus Calmette-Guerin.
The current paper provides an overview of the endocavitary treatments for UTUC, focusing on methods of administration, novel formulations, oncologic outcomes (in terms of endocavitary recurrence and progression), as well as on complications. In particular, the role of UGN-101 as a primary chemoablative treatment of primary noninvasive, endoscopically unresectable, low-grade, UTUC has been analysed. The drug achieved a complete response rate of 58% after the induction cycle, with a durable response independently of the maintenance cycle. The cumulative experience on the role of UUT instillation therapy appears encouraging; however, no definitive conclusions can be drawn about its therapeutic benefit. Given the current state of the art, any decision to administer adjuvant endoureteral therapy for UTUC should be carefully weighed against the potential adverse events.
Nevertheless, newer investigations that improve visualization during ureteroscopy, genomic characterization, novel drugs and innovative strategies of improved drug delivery are under evaluation. The landscape of KSS for the treatment of the UTUC is evolving and seems promising.
INTRODUCTION
Upper urinary tract urothelial carcinoma (UTUC) accounts for 5% of all urothelial cancers, with an estimated annual incidence of 1-2 cases/100000 [1]. The standard treatment for nonmetastatic UTUC remains radical nephroureterectomy (RNU), possibly in combination to lymph node dissection and perioperative chemotherapy [2].
Nowadays, kidney-sparing approaches have emerged as a valid alternative for selected low-risk cases or in patients not eligible or not accepting radical treatment. However, the accurate UTUC staging and grading remains challenging since the actual standards of tumor detection, i.e computed tomography urography and ureteroscopic biopsy show an overall low concordance with the pathological staging and grading with up to a third of patients with low-grade disease on biopsy having high-grade tumours on radical nephroureterectomy. The limited tissue available from biopsy specimens and the UTUC intratumoral heterogeneity together with the insufficient staging accuracy of the computed tomography urography hinder in the daily practice the possibility of a tailored treatment strategy for patients with UTUC.
With these limitations, in terms of staging, the low-risk category is represented by a unifocal tumor, less than <2 cm in size, combined to negative urine cytology for high-grade UTUC, low-grade ureteroscopic biopsy and no invasive aspect on computed tomography [2, 3].
For such cases, kidney-sparing surgery (KSS), which usually includes either partial ureteral resection, endoscopic tumor ablation or photodynamic therapy reduces the morbidity associated with radical surgery (e.g., loss of kidney function), without compromising oncological outcomes [4]. This option should therefore be discussed in all low-risk cases, irrespective of the status of the contralateral kidney as well as for selected high-risk patients (i.e. those with severe renal insufficiency or having a solitary kidney) [2].
However, KSS has a risk of ipsilateral recurrence ranging between 15–90% [5, 6] because of the potential implantation of floating neoplastic cells after endoscopic resection in the upper urinary tract [7]. Thus, according to the bladder paradigm, adjuvant endocavitary (endoureteral) instillations have been proposed that aim to reduce both the recurrence and progression risk of the UTUC.
The aim of the current paper is to provide an overview of the current adjuvant endocavitary treatments for UTUC, focusing on methods of administration, novel formulations and oncologic outcomes in terms of endocavitary recurrence or progression.
MATERIALS AND METHODS
A narrative review was carried out due to the overall low quality of the available evidence. A literature search was performed using the PubMed and MEDLINE electronic databases. The search was limited to English articles published until July 2022. Searched terms included: “UTUC” OR “urothelial carcinoma” OR “upper urinary tract” AND “instillation” OR “intracavitary treatment” OR “Mitomycin” OR “Bacillus Calmette-Guerin” OR “UGN-101”. The reference list of reports was also checked for additional publications.
Studies reporting on Ta/Tis/T1 UTUC that received endocavitary instillation with adjuvant or primary chemoablative intent with ≥5 patients were included. UTUC recurrence, CSS and OS as well as complications were recorded. Cytology response was documented in case of preinstillation positivity.
Patients with higher stage disease, involvement of the bladder or urethra were excluded.
RESULTS
Instillation therapy may be adopted both as primary treatment and as adjuvant therapy after KSS for UTUC.
Most historical studies have described small, retrospective, uncontrolled series of patients undergoing therapy with thiotepa [8], mitomycin C (MMC) [9, 10] and Bacillus Calmette-Guèrin (BCG) [11], which are the two most studied drugs to be evaluated both in the adjuvant, as well as primary setting of endocavitary instillation.
The major challenges of adjuvant endocavitary treatment are the absence of storage capacity of the UUT and the continuous downward flow of urine that removes the instilled drug from the urothelial surface [5, 12].
The instillation methods have been historically three [12, 13] (Fig. 1): 1) antegrade, through a nephrostomy tube, 2) retrograde, through a ureteral mono-J catheter, 3) retrograde, through a double-J stent by filling the bladder and creating a vesicoureteral reflux.
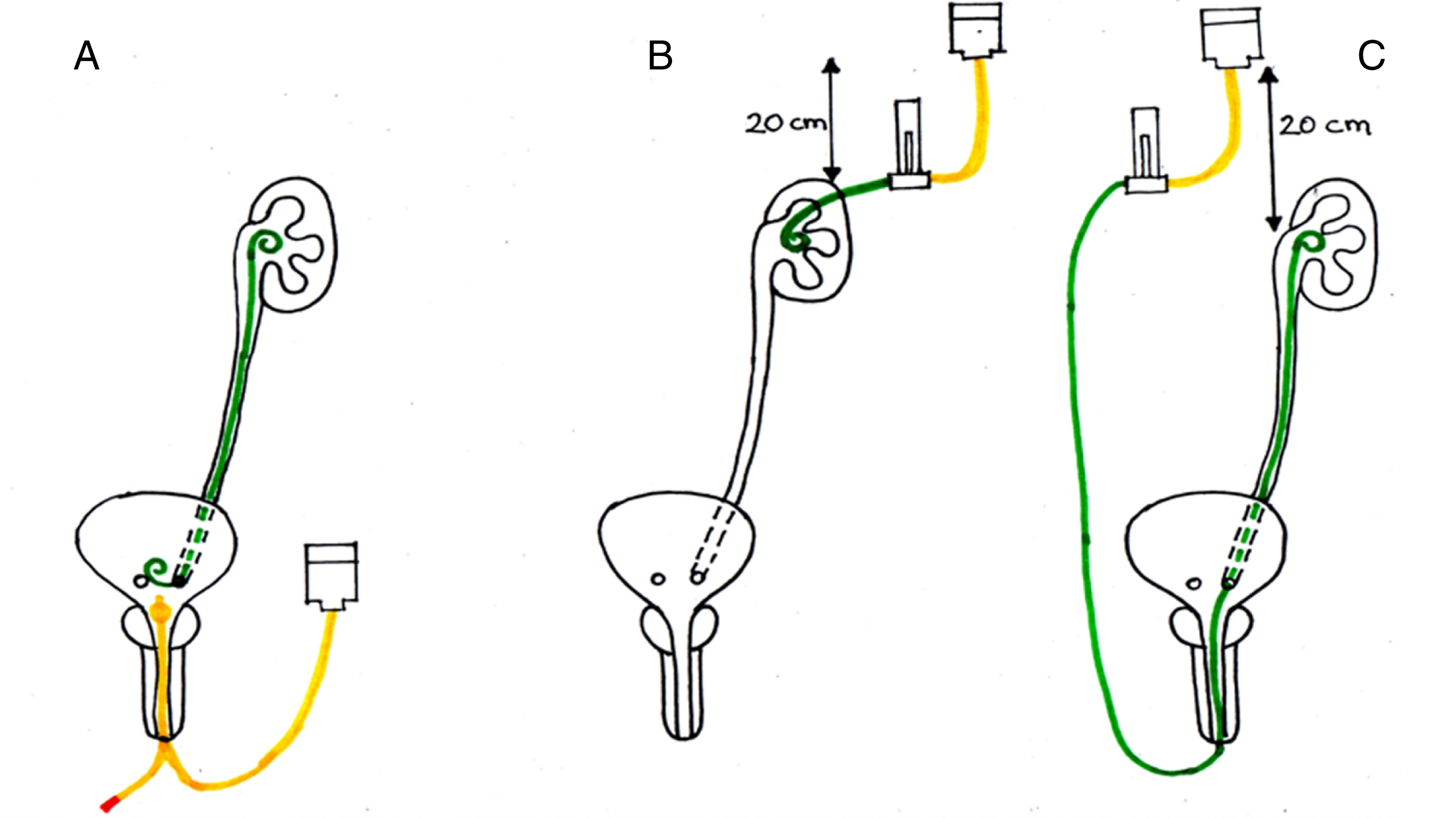
Primary options for upper tract instillation and main limits of each technique. A. Retrograde injection via double-J stent. Following the placement of the DJ, a cystogram is performed with the patient in Trendelenburg position, to determine the volume necessary to clearly visualize the entire UUT. The intravesical instillation of BCG is performed with the patient in the Trendelenburg position, held in position for 15–30’ and voided 30–120’ after instillation. Main limit: the absence of reflux in about 44% of cases limits the treatment efficacy. B. Percutaneous antegrade instillation via nephrostomy tube. It requires a pre-placed nephrostomy that should be maintained during duration of therapy; continuous drug-solution flow for 2 hours, control of flow, drug solution at 20 cm above the kidney. Main limits: Patient needs to accept the external tube for the duration of therapy. Theoretical risk of tumor seeding. C. Retrograde injection via Single-J stent. Patient in Trendelenburg, 1-hour continuous infusion with the solution placed at 20 cm above the patient. Main limit: the presence of the single-J inside the ureter hinders the adequate contact of the drug to the mucosa. There is no control of the flow rate, so intrapyelic pressures may be high or low – if too high, they may lead to systemic absortpion and bacterial sepsis, if too low, the treatment is ineffective. D. Under development (non represented in the figure): biodegradable or bilayer swellable drug-eluting stents.
The antegrade technique may be hindered by the theoretical risks of retroperitoneal tumor dissemination or drug extravasation, however, in principle it would protract the time of interaction between the urothelium and the drug. It should also entail a lower intrarenal pressure, thereby reducing the likelihood of intrarenal intravasation. The retrograde mono-J technique requires repeated ureteral manipulations, increasing the potential risk of stenosis. Finally, the technique in which the drug is administered via double-J would seem unreliable, as the drug may not come in contact with the renal pelvis [14–17]. Other studies suggest that retrograde infusion by an open-ended ureteral catheter is an ideal approach for endocavitary therapy compared to the antegrade and refluxing technique [18, 19]. However, we could not find studies comparing the different approaches. Despite this, there seems to be consensus on some elements of UUT drug administration. This should be given at a low pressure and only when an active urinary infection has been excluded [13]. When a nephrostomy tube is chosen for delivery, the drug should be instilled by gravity in order to avoid excessive intrarenal pressures, which may lead to complications because of systemic absorption and bacterial sepsis [13].
UGN-101 represents an innovative drug formulation for the primary chemoablative treatment of UTUCs. It is composed of RTGeltrademark, a sustained-release hydrogel polymer-based formulation [20] containing 4 mg MMC per ml/gel; in a cold condition UGN-101 is liquid, but at body temperature, it becomes a gel. Thus, when administered through a ureteral catheter, it adheres to the upper tract urothelium, slowly dissolving with urine and gradually releasing MMC in the UUT over a few hours. This formulation enhances the time of exposure of UUT to MMC increasing its pharmacological activity [20].
In the animal model, UGN-101 proved to be a safe and effective method to deliver MMC in the UUT [21, 22]. UGN-101 was also validated in a human cohort by the Olympus trial, i.e. a prospective, open-label, single-arm trial, aimed at assessing the efficacy, safety, and tolerability of UGN-101. The drug was retrogradely instilled in the UUT of 71 patients with a primary noninvasive, endoscopically unresectable, low-grade, UTUC [23], achieving a complete response (CR) (i.e. negative ureteroscopy and negative cytology) in 41 (58%) after the induction cycle. During follow-up, only 6/41 patients (15%) had a recurrence of urothelial carcinoma, while there were not cases of progression to high-grade or invasive cancer.
A recent update of the Olympus trial [24] evaluated the durability of response to primary chemoablation of low-grade UTUC using UGN-101. The 12-mo CR rate was 56% (23/41), with 6/12 (50%) patients receiving no maintenance treatment and 17/29 (59%) patients receiving ≥1 maintenance instillation (median 6, range 1–11). Thus, there was no clear association between maintenance cycle and durability of response while increasing the number of UGN-101 instillations appeared to increase the incidence of urinary adverse events (AE). The authors concluded that the durability of response to UGN-101 is clinically meaningful independently of the maintenance therapy, offering a KSS alternative for patients with low-grade UTUC.
More recently, Rosen et al. [25], described their early outcomes for a cohort of patients who have undergone UGN-101 administration via nephrostomy. The tube was inserted in an adjacent calyx avoiding its direct placement into the tumor; instillations proceeded weekly for a total of 6 instillations. CR was observed through follow-up ureteroscopy in 4/8 (50%) of patients. Three patients reported 5 AE, mostly of grade 1.
Overall, 33 eligible reports were identified. All included reports were nonrandomized observational case series.
Studies reporting on the use of BCG as first-line therapy for the treatment of UUT-CIS
Failure rate = persistent disease or UUT recurrence or progression after treatment. *Includes series of both Cis and papillary UTUC; the rate of AEs for Cis only groups cannot be extrapolated.
Following the report of Herr on intravesical BCG in a patient after resection and reconstruction of a pelvic kidney with pyelovesical anastomosis for persistent CIS of the renal pelvis in 1985 [29], Studer et al reported the outcomes of the first series of UUT BCG instillation for the cure of CIS [30]. In that report, 8 patients for a total of 10 renal units (RU) were treated with antegradely instilled BCG for a duration of 6 weeks. Failure rate was 20% while in 1 patient treatment was stopped prematurely after severe septicemia.
Since that study, 14 case-series reporting on the outcomes of BCG (antegrade or retrograde instillation) for the primary treatment of UUT-CIS and including more than 240 RU have been identified [14–16, 31–41]. The duration of the treatment is usually 6 weeks. Various doses and dilution volumes ranging between 40–250 mL have been used; the solution is administed slowly (usually 1 ml/min). After a mean/median follow-up ranging between 10–58.3 months, the failure rate greatly varies between 0–57%.
The largest experience of BCG primary treatment for UUT-CIS is from Tomisaki et al. [16]. Studying the efficacy and tolerability of the drug in 52 RU, the authors reported a CR rate of 90% (47/52), with 5-year recurrence- and progression-free survival rates of 60.2% and 74.2%, respectively. The second largest experience is from Giannarini et al. [41] and concerns the transnephrostomic (antegrade) instillation of BCG. In a total of 42 RUs the urothelial carcinoma recurrence occurred in 17/42 (40%) and progression in 2/42 (5%) cases. Radical nephroureterectomy (RNU) was performed in 2/42 (5%).
In a 2019 meta-analysis [12], the outcome of BCG instillation treatment in patients with UUT CIS was examined on a total of 226 patients. During a median follow-up of 31 months, 177 (84%) had a full cytology response, 72 (32%) developed UTUC recurrence and 38 (17%) experienced disease-progression, respectively. In the quantitative analyses that comprised 182 patients, the pooled estimates for cytology response, UTUC recurrence and progression were 84% (95% CI 75–92%), 34% (95% CI 27–41%) and 16% (95% CI 11–22%), respectively, with no statistically significant differences among the instillation approaches. Among patients having initial full cytology response, the cumulative rates for UTUC recurrence and progression were 10% (95% CI 5–16%) and 25% (95% CI 8–33%), respectively. Data on mortality was not available.
In the majority of the evaluated studies, the diagnosis of UUT CIS has been usually performed by positive selective urine-cytology (2-3 series) and negative findings in the computed tomography (CT)-urography scan, combined to a negative bladder barbotage cytology and negative multiple random biopsies of the bladder mucosa and prostatic urethra. Thus, the basic parameter for local tumor chemoablation for these studies was the cytology response.
Shapiro et al. [39] presented their outcomes on the retrograde instillation of BCG combined to interferon–α2B into the UUT via ureteral catheter in patients with biopsy-proven CIS. Following a 6-week induction course, the patients were submitted to both selective urinary cytology and rebiopsy of the UUT. Eight out of eleven patients experienced a CR (i.e negative cytology and biopsy). Two patients had negative biopsy but positive cytology and were offered a second induction course, achieving CR. The last patient had persistence of lesions after induction and was offered RNU. Total kidney preservation rate was 91%, while no treatment related events were observed.
In 2018 Horiguchi et al. [15] compared the oncological outcomes of 58 patients with UUT CIS submitted to RNU (N = 20) versus topical BCG instillation therapy (N = 38). Groups were similar in baseline characteristics. BCG was infused by retrograde catheterization (mono or double-J stent, 6-week course). No significant differences were found in the progression-free (PFS), cancer-specific (CSS) and overall survivals (OS) between the groups. The multivariate analysis revealed that topical BCG therapy did not worsen the prognosis of these patients. Surgery-related AE were observed in 3 patients (15%) vs 35 patients (92%) of the BCG group; however, no significant difference in severe AE was observed between groups. The authors concluded that BCG therapy for UUT-CIS might be a useful alternative for patients ineligible for RNU.
Although BCG as first-line therapy for UUT CIS seems promising, as it achieves favorable clinical outcomes and acceptable AE, several variables remain to be standardized, such as the optimal route of delivery, the formulation with optimal exposure, the strain, the instillation schedule, the volume of BCG solution and the duration of infusion. Furthermore, research is limited due to small patient cohorts, varying follow-up, and no standardized methodology to assess response; in fact, the diagnosis of UUT-CIS in many of the evaluated studies was usually based only on the presence of positive urinary cytology and negative radiological findings, without confirmatory URS. As a consequence, the response rates were determined by restitution of normal urine cytology. The diagnosis of CIS could be therefore biased by limitations in the diagnostic accuracy of radiological imaging and urine cytology.
Studies reporting on the us of BCG in the adjuvant setting for UUT Ta-T1 papillary disease or UUT-CIS
BCG has been also used after the endoscopic treatment of UUT papillary disease as described in 10 studies [11, 41–49]. In this adjuvant setting, small case-series report recurrence rates ranging between 13–59%. In [41], recurrence occurred in 13/22 RUs (59%) and progression in 9/22 (41%) with Ta/T1 tumors. RNU was performed in 5/22 (23%) with Ta/T1 tumours. According to the data of this study, patients treated in the primary setting with BCG for UUT-CIS tended to have better RFS (p = 0.42) and significantly better PFS (p < 0.01) and RNU-free survival (p = 0.05) compared with those treated with the same drug in the adjuvant setting after ablation of Ta/T1 tumours.
Recently, Jamil et al. [50] designed a single-centre phase-II, non-randomised and unblinded safety and efficacy study of pembrolizumab+BCG for subjects with pathologically documented non-muscle invasive high-risk UTUC unfit or unwilling to be treated with RNU and submitted to endoscopic ablation (trial registration number NCT03345134). The study plans to enroll 20 subjects that will receive treatment with 200 mg of intravenous pembrolizumab every 21 days, starting 2 weeks from the initial endoscopic resection and continuing for 6 weeks after the final dose of BCG. The outcomes are awaited.
Studies reporting on the use of MMC in the adjuvant setting for UUT Ta-T1 papillary disease
*In this study the authors do not provide separated outcomes between BCG and MMC. **Overall recurrence rate for patients with and without adjuvant MMC; a further subanalysis demonstrated that the 5-year upper-tract recurrence-free survival did not differ significantly between these groups (i.e no benefit of adjuvant MMC).
A total of 8 studies reporting on the role of MMC in the adjuvant setting for a total of 124 patients have been identified [10, 51–55]. Total ipsilateral UUT recurrence rate ranges between 14–68.5%.
Regimens for instillation of MMC vary, with doses of 40 mg being dissolved in 30, 40 or 1000 mL of saline or 5 mg in 20 mL saline with a slow infusion velocity of 1 mL/min.
In 2017 Metcalfe et al. [53] reported their outcomes of the first study on adjuvant MMC utilizing both an induction (six weekly instillations) and maintenance (once monthly for at least 3 months or 3-weekly instillations) course. 27 patients with 28 RUs received adjuvant topical therapy with MMC using 40 mg in 20 mL saline instilled either via ureteral catheter or via nephrostomy tube over 2 hours. The recurrence rate was 39% at a median follow-up of 19 months. The 3-year PFS was 80%, with 18% of patients ultimately undergoing RNU.
In 2020 Gallioli et al. [56] provided the first prospective non-randomized trial addressing the safety and feasibility of an adjuvant single-dose retrograde UUT instillation of MMC (ASDM) performed within 6 h after endoscopic ablation of low-grade UTUC. UTUC recurrence was evaluated with second-look URS and CT scan/URS every 6 months. At a median follow-up of 18 months (IQR 10–29), the recurrence rate was 23.5% and 55.5% in the ASDM group and controls, respectively (p = 0.086). Mean RFS was 28.8 months in the ASDM group vs 18.8 months in controls (p = 0.067) with ASDM being associated with a 7.7-fold lower risk of UTUC recurrence (HR = 0.13; 95% CI 0.03–0.65; p = 0.01) on multivariate Cox regression.
Adjuvant chemotherapy with the use of MMC is feasible; however the small numbers of patients of the published studies as well as the variable selection criteria, do not allow for definite conclusions to be reached. Since the ASDM seems to reduce the risk of recurrence in patients affected by low-grade UTUC, its use should be tested in a randomized controlled trial.
The most common AE of BCG as primary treatment for UUT-CIS are represented by fever and irritative symptoms while the most threatening complications of the drug are its dissemination and septicaemia due to gram-negative bacteria. Deaths during instillations are rare, however they have been reported in several studies, caused by E. coli sepsis [41], a non-BCG-related septicaemia [49], and interstitial pneumonia possibly following BCG dissemination [38]. Other severe AEs have been described, such as (febrile) kidney tubercolosis [15, 32], ureteral stenosis requiring ileal substitution of the ureter [32] and bladder contraction [42]. In 2021, Schembri Higgans J et al. [57] reported the first case of systemic BCGosis following intra-renal instillation that required long-term antituberculartreatment.
Deaths have been described after use of BCG in the adjuvant setting as well [40, 49]. A testicular granuloma requiring orchiectomy has been also reported [49].
To minimize the risk of bacterial sepsis, patients must be evaluated for active infection before UUT instillation therapy, and only a low-pressure delivery system should be used. Ureteral integrity should be also verified by a retrograde or antegrade pyelogram prior to instillation while the BCG therapy should be avoided early after surgery.
The most common AE of UGN-101 included ureteral stenosis, urinary tract infection, hematuria, flank pain, dysuria, transient creatinine increase, hydronephrosis and frequency; they were usually transient and mild to moderate in severity [23, 24]. In the Olympus trial final report [24], ureteric stenosis was the most frequently reported AE in the safety population, occurring in 31/71 patients (44%); however, it was reported in 19/29 patients (66%) who received ≥7 instillations of UGN-101 (ie ≥1 maintenance instillation) compared with 12/42 patients (29%) who received ≤6 instillations of UGN-101. These differences reflect the increased risk of AEs associated with increased exposure to MMC and an increased number of procedures.
MMC instillations in the UUT are mainly related to local AE such as dysuria, cystitis or hematuria [58]. Systemic AE should not be expected since there is only a scarce absorption of the drug through the urothelium that accounts for only 1–6% of the total administered dose [58]. However, this rate could be increased in patients with an extensive area of tumor resection or for those who receive MMC instillations shortly after tumor ablation. Pulmonary and hematological toxicity is rare after intravesical administration and even rarer following UUT-MMC administration. Rodriguez-Reyes presented a case of interstitial pneumonitis linked to myelosuppression in a patient with left kidney pyelocaliceal urothelial carcinoma who received five instillations of MMC through a ureteral catheter [58].
Another commonly reported AE following UUT-MMC instillation is ureteral stricture [52]; however, the cause of the ureteral strictures is unknown. It could be related to endoscopic resection, multiple ureteroscopies, MMC itself, delivery method, or the additive effect of all these factors. Regardless of the definitive cause of the ureteral stricture, observation for onset of hydronephrosis and renal failure is recommended to prevent irreversible renal deterioration.
Rare cases of death due to MMC extravasation during upper tract instillations have been also described [45].
Concerning the ASDM [56], Clavien grade≤II complications occurred in 32% (8/25) and 30.7% (8/26) of the ASDM and control group, respectively (p = 0.9). Two Clavien III complications occurred in the ASDM group: bladder hematuria after concomitant transurethral resection of bladder and obstructive kidney failure in a single-kidney patient. The administration of MMC at the time of the UTUC ablation also portrays a risk of drug extravasation, with potentially catastrophic consequences.
EVIDENCE SYNTHESIS AND FUTURE DIRECTIONS
At the state of the art, the literature does not provide any firm statement on the efficacy of adjuvant topical therapy in the treatment of papillary UTUC. Reports are limited to retrospective, non-randomised small cohort studies with limited follow-up that mainly assess the use of BCG, with only a minority assessing MMC. The control (i.e. non-treatment) arms is frequently not included and consequently, it is difficult to establish any therapeutic benefit from such studies. On the other hand, suboptimal drug concentrations and modes of delivery may have hindered the outcomes of adjuvant therapy for UTUC. Concerning the ASDM, although promising its efficacy should be tested in a randomized controlled trial. As a conclusion, any consideration to administer adjuvant treatment should be carefully assessed against the potential side-effects.
On the contrary, the outcomes on the use of BCG as first line treatment for UUT-Cis seem encouraging with positive cytology responses of up to 100% in the short term. However, failure rates (persistent disease or UUT recurrence or progression after treatment) of up to 57% during follow-up have been reported. The efficacy of the treatment should be evaluated by adequately designed studies with biopsy-proven Cis in order to overcome the inherent flaws of the presumptive diagnosis of Cis that -in the current literature- was mainly obtained by the presence of positive urine cytology combined to negative radiological findings.
Improvements in diagnostics of UTUC both in terms of intraoperative endoscopic visualization and genomic characterizzation of UTUC [59] is crucial for adequate patient selection for KSS and follow-up after surgery. Moreover, innovative forms of drug delivery such as biodegradable [60] or bilayer swellable drug-eluting stents have also been developed to improve drug delivery and apposition with the urothelium [61]. Table 4 summarizes the main axes of investigations for KSS for UTUC.
UTUC: upper tract urothelial carcinoma; TERT: telomerase reverse transcriptase; FGFR3: fibroblast growth factor receptor 3; DNA: DeoxyriboNucleic Acid
CONCLUSIONS
Although the cumulative experience on the role of UUT instillation therapy appears encouraging, definitive conclusions about the therapeutic benefit of this therapy are not easily reached. Possible reasons for this include (1) insufficient numbers to show clinical significance; (2) different tumor biology from that of the extensively studied bladder counterparts; (3) a nonstandardized and possibly inadequate delivery system that, unlike in the bladder, does not allow uniform delivery of the agent with adequate dwell time to enable a clinical response and 4) usual absence of control (non-treatment) arm for comparison. Thus, in the absence of a strong positive evidence, any consideration to administer adjuvant therapy should be carefully assessed against the potential AEs. Further comparative studies of adequate design are necessary to clarify: 1) the role of UGN-101 as a primary chemoablative treatment of primary noninvasive, endoscopically unresectable, low-grade, UTUC 2) the role of the adjuvant single-dose retrograde UUT instillation of MMC and 3) the role of BCG as first line treatment for biopsy proven UUT-Cis. Newer investigations that improve the visualization during ureteroscopy, genomic characterization, novel drugs and innovative strategies of improved drug delivery are under evaluation. The landscape of KSS for the treatment of the UTUC seems promisingly evolving.
Footnotes
ACKNOWLEDGMENTS
We thank Dr. Lorenzo Dutto, Urologist in the Department of Urology of the Queen Elisabeth’s University Hospital (Glasgow, UK) for his kind help with the linguistic revision of the manuscript.
FUNDING
The authors report no funding.
AUTHOR CONTRIBUTIONS
ADA: conception; performance of work; interpretation of data; writing the article.
M.K. performance of work; interpretation of data.
C.K. performance of work; interpretation of data.
G.G: conception; performance of work; interpretation of data.
ETHICAL CONSIDERATIONS
This study, as a literature review is exempt from any requirement for Institutional Review Board approval. No human or animal research was involved in the elaboration of this manuscript.
CONFLICT OF INTEREST
ADA., M.K., C.K. have no conflicts of interest concerning the manuscript.
G.G. is an Editorial Board Member of this journal, but was not involved in the peer-review process nor had access to any information regarding its peer-review.
