Abstract
BACKGROUND:
Many variables may affect the cost of open radical cystectomy (RC) care, including surgical approach, diversion type, patient comorbidities, and postoperative complications.
OBJECTIVE:
To determine factors associated with changes in cost of care following open radical cystectomy (ORC) for bladder cancer using the National Inpatient Sample (NIS).
METHODS:
Patients in the NIS with a diagnosis of bladder cancer who underwent ORC with ileal conduit from 2012–2017 using ICD-9-CM and ICD-10-CM codes were identified. Baseline demographics including age, race, region, postoperative complications, and length of stay were obtained. Univariable and multivariable logistic regression were used to identify factors associated with cost variation including demographics, clinical characteristics, surgical factors, and discharge quarter (Q1-Q4).
RESULTS:
5,189 patients were included in the analysis, with 4,379 at urban teaching hospitals. On multivariable regression analysis, female sex [$1,734 ($1,024–2,444)
CONCLUSIONS:
Significant differences in cost of ORC with ileal conduit exist with respect to patient sex, medical comorbidities, and discharge timing. These differences may relate to greater disease burden in female patients, patient complexity, and variation in postoperative care in academic programs.
INTRODUCTION
The direct medical costs of cancer care in the United States (US) alone were estimated at approximately $125 billion in 2010 [1]. Accounting for evolving population demographics and epidemiological trends in cancer, this price tag is expected to rise to over $155 billion within the next few years. Bladder cancer imposes a significant financial burden on the health care system and the individual, as it is one of the costliest cancers to treat on a per patient basis with an estimated cost of care of $6 billion in 2020 and a majority of this cost is attributed to non-muscle invasive cancer [1–3]. For patients with muscle-invasive disease, the most expensive component of this cost often comes from the hospitalization event for radical cystectomy (RC) and urinary diversion [4].
Many variables may affect the cost of RC care, including surgical approach, diversion type, patient comorbidities, and postoperative complications. Regional variations in cost of RC have also been reported, which suggests that cost of living and other geographic differences in healthcare delivery may also impact cost [5]. It is documented that minimally-invasive techniques are costlier than open RC (ORC) by nearly $4,000 [6]. Similarly, the cost of continent urinary diversions increases inpatient charges by over $4,500 [7]. The primary purpose of this study is to identify differences in cost of RC hospitalization attributable to patient, hospital type, and temporal characteristics. Specifically, our study aimed to characterize differences in cost and complications across teaching and non-teaching hospitals.
MATERIALS AND METHODS
Data source
Patients with bladder cancer undergoing ORC were identified in the National Inpatient Sample (NIS) of the Health Care Utilization Project (HCUP) from 2012 to 2017 [8]. This was deemed IRB exempt by our institution. The NIS dataset is the largest national all-payer inpatient database in the US and samples from hospitals comprising 97%of all discharges in the US. NIS data is de-identified and does not contain protected health information or specific hospital identifiers, and qualified for exemption from the institutional review board at our institution.
Patient selection and outcomes
Each NIS dataset from 2012 to 2017 was queried for the International Classification of Disease (ICD)-9 or ICD-10 diagnosis codes for bladder cancer as defined in
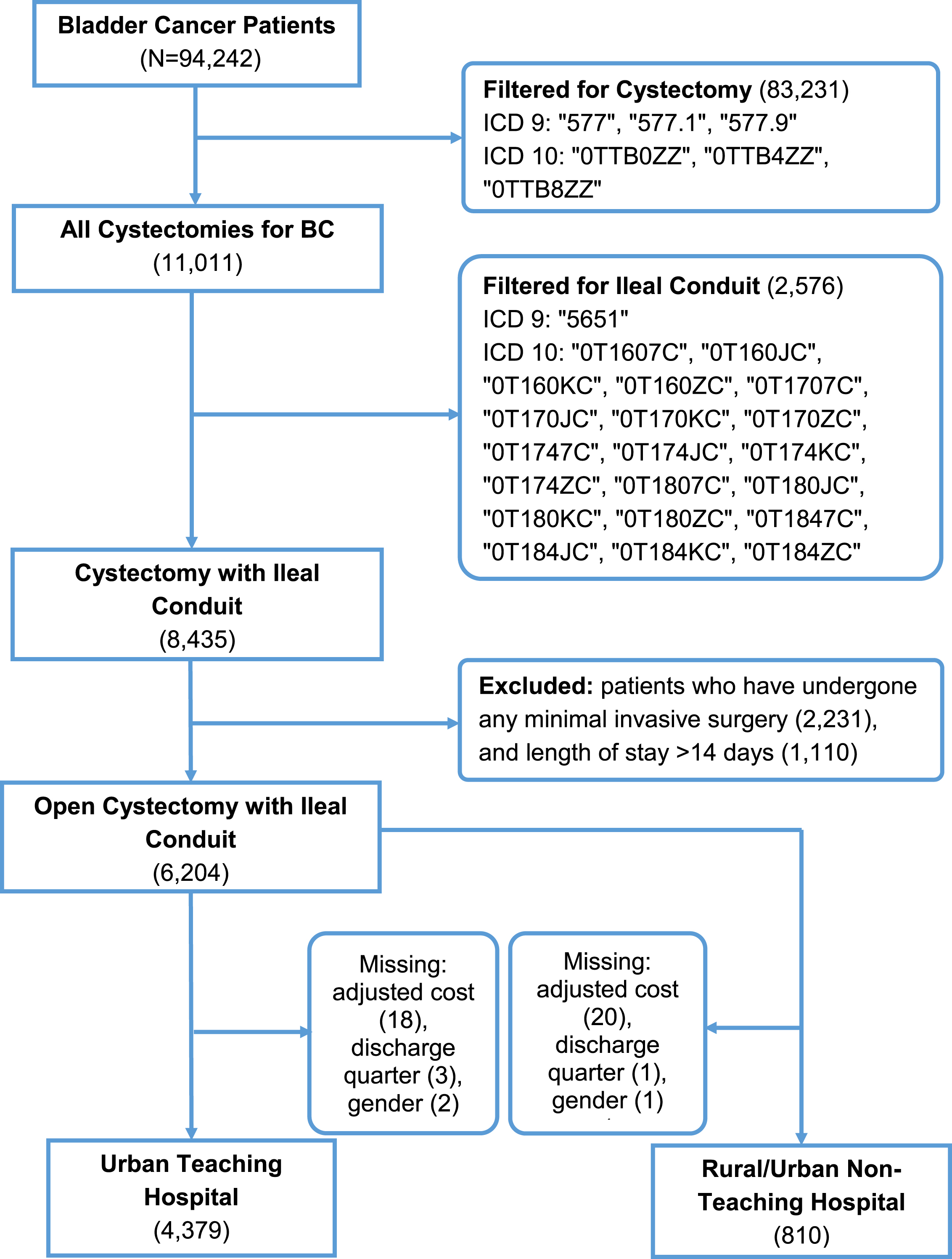
Patient selection flowchart from the NIS data from 2012–2017.
Our primary outcome was the adjusted total cost per hospitalization, calculated by multiplying the charges that hospitals billed for services in the NIS data by the cost-to-charge ratio (CCR), a validated method by the Agency for Healthcare Research and Quality and used by previous studies to estimate to estimate actual hospitalization cost [6–8]. Adjusted hospital cost and hospital charges were rounded to the nearest dollar.
Covariables
The following clinically relevant variables were identified from the NIS data. Patient demographic information, including age (years; continuous), sex (male, female), race (white, African American, Other/Unknown) and insurance type (Medicaid, Medicare, private, self-pay, unknown) was obtained. Similarly, length of stay (days; continuous) and hospital region in the US (East North Central, East South Central, Mid-Atlantic, Mountain, New England, Pacific, South Atlantic, West North Central, West South Central) were identified. The Elixhauser comorbidity score, a method developed by Elixhauser et al. in 1998 used to estimate the burden of a patient’s comorbidities using ICD-9 and ICD-10 codes, was utilized as a measure of patient medical complexity [8, 12]. Discharge quarter was defined as date of discharge and categorized as: Q1 –January to March, Q2 –April to June, Q3 –July to September, and Q4 –October to December. Finally, ICD-9 and ICD-10 diagnosis and procedure codes were used to identify patients who experienced any inpatient complications (yes/no).
Statistical analysis
Descriptive statistics were reported as medians (interquartile range) for continuous variables and numbers (percentages) for categorical variables. Q2 was used as the reference discharge quarter as it was the closest to the median adjusted total cost. Univariable and multivariable logistic regression of all previously described features were used to identify predictors associated with greater adjusted hospital cost. As an additional sensitivity analysis, we also conducted a linear regression analysis for patients at urban non-teaching or rural hospitals. All statistical analyses were performed using R (version 3.6.3, The R Foundation, Vienna, Austria).
RESULTS
Of the 4,379 patients analyzed in the primary study population, the median age was 70 years with 80.0%White and 78.6%male patients, and there was a relatively equal distribution of patients by teaching vs non-teaching hospitals. (
Patient demographics at urban teaching hospitals
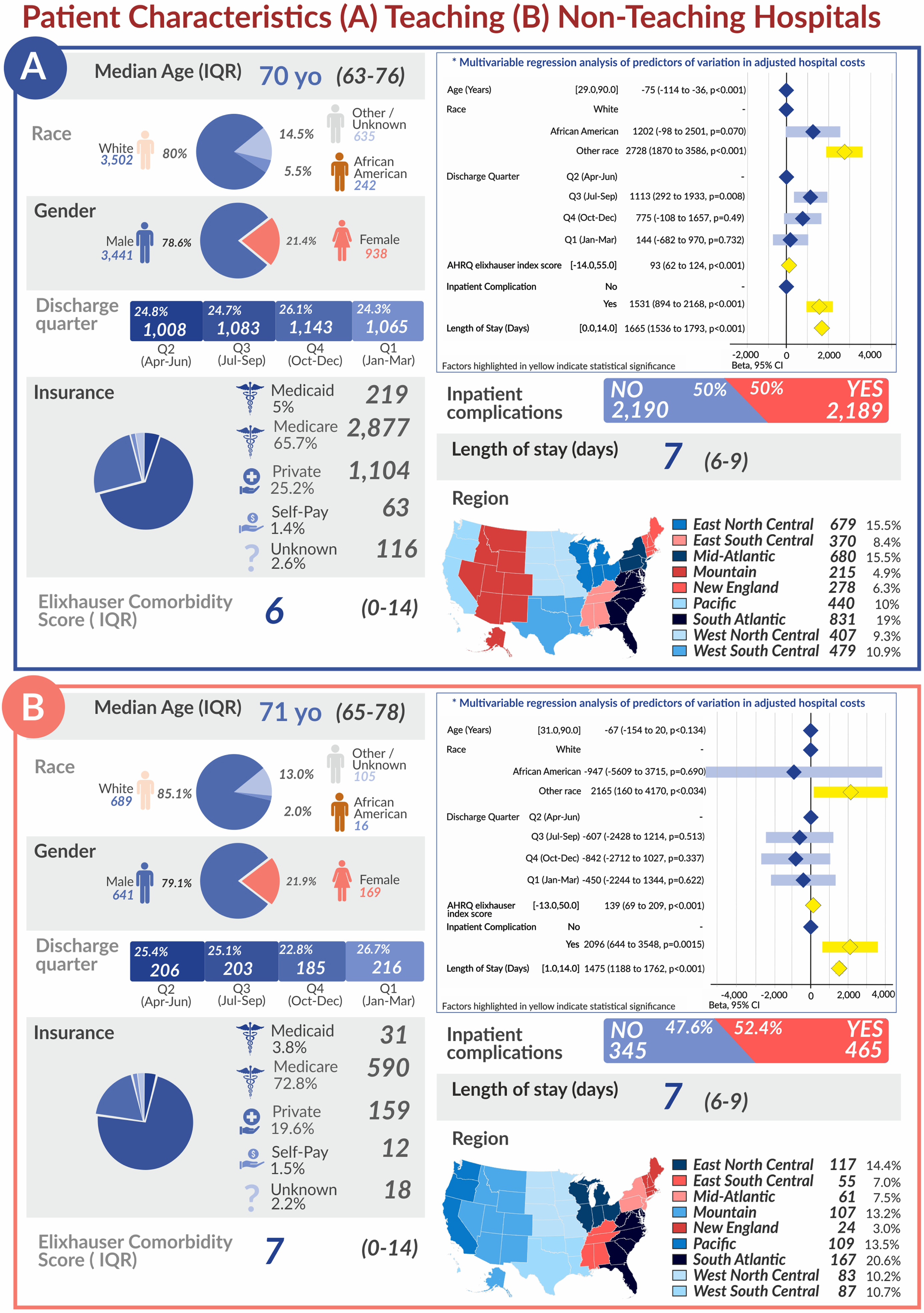
Patient Characteristics (A) Teaching (B) Non-Teaching Hospitals
Linear regression analysis of patient and hospital factors associated with cost variation at urban teaching hospitals
Among the 810 additional patients who underwent ORC at an urban non-teaching or rural hospital, the median age was 71 years with 85.1%White and 79.1%male patients (
COMMENT
RC is the gold standard for treatment of non-metastatic muscle-invasive bladder cancer and select patients with high risk non-muscle invasive bladder cancer [13]. Proposed factors affecting cost of RC can be attributed to patient, surgeon, and hospital characteristics [10, 15]. Higher volume surgeons and hospitals, generally defined as those with greater than ten RC per year, have better outcomes postoperatively and report lower rates of mortality and complications [11, 15]. Though these outcomes can be attributed to surgical technique and hospital resources, the greatest variability in index hospitalization cost of RC has been ascribed to postoperative complications.
Interestingly, this study found disparities between cost of ORC based on patient sex with a $1,734 increase in cost per hospitalization at urban teaching facilities and $2,172 increase at non-teaching and rural facilities for female compared to male patients. The increased cost may stem from differences in the actual surgery performed, such as need for anterior exenteration. Prior research by C
The current study also showed increased adjusted cost of ORC in patients with greater medical complexity, as measured by the Elixhauser comorbidity score, and patients with any complication during admission for ORC. This is congruent with existing literature that found patients with greater comorbidities tend to have greater morbidity, higher rates of readmission, and higher 90-day costs following RC [21, 22]. Though the difference in cost on multivariable regression was marginal for each single point increase in score ($93 for urban teaching hospitals and $139 for urban non-teaching and rural hospitals), a patient with several comorbidities may easily increase the cost of ORC care by several hundred dollars. Managing complex patients may pose a challenge to trainees and lead to greater use of consulting services and diagnostic tests. Complication rates following cystectomy remain high at around 50%following RC regardless of surgical route [6, 23]. However, enhanced recovery after surgery (ERAS) protocols can limit reactive measures and over-management of patients by reducing variance in the perioperative period and lower complication rates [23–25]. Its effect on overall costs appears mixed however, with an average cost savings of $4,488 per RC hospitalization compared with standard management reported by some groups, and no difference in cost reported at another single specialty cancer center [23, 26].
Our results also showed a borderline statistically significant difference in cost of ORC between Q2 and Q3 at urban academic centers. While it is difficult to pinpoint all factors influencing this result without additional study, one possibility relates to trainee experience as there is frequently a large of trainees at academic hospitals during Q3. Our analysis of non-teaching and rural hospitals, where fewer trainees are present, did not show a significant cost difference between discharges in Q3 compared to Q2. The “July Effect” has been used to describe the transition period at the start of each academic year when the average experience level of a teaching hospital declines suddenly [27]. Little data exists on how trainee involvement affects perioperative management, but overutilization of laboratory and imaging tests by trainees can increase cost of care [28, 29]. Interventions such as cost reminders within the electronic health system, teaching, and audit/feedback can reduce avoidable testing. ERAS protocols may be another method of reducing variability in diagnostic testing.
Our data showed a statistically significant decrease in cost by $75 with each single year increase in age in the primary analysis population. This change appears counterintuitive when taken in isolation, but may not capture the variability in patient health status at different ages and seems less clinically significant compared to other factors previously mentioned. Finally, there was a wide range of ORC care costs across different regions of the US. It is beyond the scope of this study to identify regional practice patterns and patient populations that may account for these differences and NIS data does not link patients to specific hospitals, but this may be an interesting basis for future study.
This study has several limitations, including retrospective design and the use of NIS data. The NIS is a cross-sectional dataset and does not divulge differences in operative technique (such as organ-sparing, lymph node dissection, or trainee involvement), whether ERAS protocols were utilized, or severity of disease. This data does not include long-term complications following the index hospitalization. Though we limited the primary analysis to urban teaching hospitals, the NIS does not link patient data to specific surgeons or hospitals which may affect cost depending on surgical volume. Other factors may account for temporal differences in adjusted cost such as timing of initial presentation and seasonal referral patterns by primary care providers. Different hospitals may also have different coding practices that do not allow direct comparison of costs involved, and coding errors may cause discrepancies in data. However, we also consider NIS data to be a strength of this study as it is the largest all-payer inpatient observational database, which provides reliable cost information and is used to guide healthcare policy.
CONCLUSIONS
We conducted an analysis of over 5,000 representative patients in the US to identify clinical predictors of cost variation during hospitalization for ORC and ileal conduit. This study demonstrates that mean adjusted total cost following ORC at urban academic institutions is significantly higher in patients who are female, have greater comorbid conditions, have any inpatient complication following ORC, and those discharged in July to December. Female patients appear to be disproportionately affected by medical complications following ORC which may increase cost of care by nearly $2,000. Implementation of and training on ERAS protocols may help reduce variability in costs while preserving high quality care.
Footnotes
ACKNOWLEDGMENTS
The authors have no acknowledgments.
FUNDING
The authors report no funding
AUTHOR CONTRIBUTIONS
Conceptualization -Helen H Sun MD, Adam Calaway MD MPH; Data Analysis -Amr Mahran MD MS; Investigation -Amr Mahran MD MS, Megan Prunty MD; Methodology -Laura Bukavina MD MPH, Amr Mahran MD MS; Project Administration -Adam Calaway MD MPH,Supervision-Lee Ponsky MD, Laura Bukavina MD MPH; Visualization -Ilaha Isali MD, Laura Bukavina MD MPH; Writing-Original Draft-Helen H Sun MD, Laura Bukavina MD MPH, Ilaha Isali MD; Writing-Review & Editing-Kevin Ginsburg MD, MS; Sarah Markt ScD MPH; Adam Calaway MD MPH, Laura Bukavina MD MPH
CONFLICT OF INTEREST
Helen H. Sun, Megan Prunty, Ilaha Isali, Amr Mahran, Kevin Ginsburg, Sarah Markt, Lee Ponsky, Adam Calaway and Laura Bukavina have no conflicts of interest to report.
