Abstract
BACKGROUND:
Bladder cancer surveillance is invasive, intensive and costly. Patients with low grade intermediate risk non-muscle invasive bladder cancer (NMIBC) are at high risk of recurrence.
OBJECTIVE:
The objective of this model is to compare the cost of a strategy to alternate surveillance with cystoscopy and a urine marker, Bladder EpiCheck, to standard surveillance.
METHODS:
A decision tree model was built using TreeAge Pro Healthcare to compare standard surveillance (Standard) with a modified surveillance incorporating Bladder EpiCheck. The model was based on 2 years of surveillance. Outcomes were obtained from literature. Costs were obtained from US and 9 European countries. Sensitivity analyses were performed.
RESULTS:
The efficacy of the model was equivalent in terms of recurrence for each arm with median recurrence rate of 22%. When setting marker price at 200 local currency, the marker arm was less expensive in the USA, Netherlands, Switzerland, Belgium, Italy, Austria and UK by 154€ to 329£ per patient, for a 2-year period. Cost was higher in France, Spain, and Germany by 33–103€. Cost parity was achieved with marker price between 148€ and $421. Marker cost and specificity have the greatest impact on the overall model cost.
CONCLUSIONS:
A strategy alternating the urine marker Bladder EpiCheck with cystoscopy in the surveillance of patients with low grade intermediate risk bladder cancer is cost equivalent in the US and European countries when the marker is priced 148€ –$421, as a result of the marker’s high specificity (86%). Prospective studies will be necessary to validate these findings.
INTRODUCTION
Bladder cancer is the 5th most common cancer and one of the most expensive from diagnosis to death. Approximately 75% of newly diagnosed bladder cancers are non-muscle invasive bladder cancer (NMIBC) and these are managed with risk-based intravesical therapy. Due to high risk of recurrence, patients with NMIBC undergo frequent cystoscopy to allow for earlier detection of recurrence and progression. Frequent cystoscopy is associated with cost and morbidity to patients [1–3]. There are also studies that demonstrate overuse of cystoscopy in low risk patients [4]. For patients with intermediate and high risk NMIBC, patients are often subjected to cystoscopy every 3 months for 1st one to two years [5]. The Bladder Cancer Advocacy Network Patient Survey Network evaluated research prioritization for patients and found that reducing pain associated with cystoscopy was high priority for patients with NMIBC [6]. As such, strategies to reduce number of cystoscopic procedures during surveillance would be viewed favorably if it can be done safely and efficiently.
Urine based tumor markers have been introduced to improve detection and surveillance of bladder cancer but their incorporation into clinical practice has been challenging. Overall, they have a better sensitivity than cytology especially for detection of low-grade cancers [7].
Bladder EpiCheck® (Nucleix Ltd.) is a new promising urine-based test which includes a panel of 15 DNA methylation patterns for the identification of recurrent NMIBC [8]. A recent study including 822 patients with NMIBC found the test had a specificity of 85.8% (95% confidence interval [CI] 83%–89%), a negative predictive value (NPV) of 94.3% (95% CI 92%–96%) for the detection of any cancer and an NPV of 98.8% (95% CI 97%–100%) for the detection of high-grade cancer [9, 10]. The sensitivity of the test was 62.5% (95% CI 51%–73%), 86.4% (95% CI 72.6%;94.8%) and 33.3% (95% CI 19%–51%) overall, high and low-grade cancer, respectively.
For patients with low grade intermediate risk NMIBC, the risk of recurrence is high but risk of progression is very low over a 3 to 6-month period [11]. For these patients, incorporating a urine marker alternating with cystoscopy could reduce the frequency of cystoscopy without jeopardizing oncologic outcomes. In this study, we develop a decision analysis model to compare standard surveillance with a modified surveillance incorporating Bladder EpiCheck. We assessed the cost of each approach in the US and several European countries.
MATERIALS AND METHODS
A decision tree model was built using TreeAge Pro Healthcare (Williamstown, MA) software to compare standard surveillance (Standard) with a modified surveillance incorporating Bladder EpiCheck where cystoscopy is alternated with Bladder EpiCheck test (marker). The model was based on 2 years of surveillance after a transurethral resection of a bladder tumor (TURBT). Table 1 details all model assumptions.
Model assumptions
Model costs and rates of use of cytology and office fulguration
*Rate used in model. ∧Based on mixed payers from over 1,200 cystoscopic procedures at UT Southwestern Medical Center in Dallas. ‡Cytology and pathology costs are included in the overall price of the procedure (cystoscopy and TURBT, respectively).
The base case is a patient with intermediate risk NMIBC according to the AUA guidelines for NMIBC with low grade only disease who is undergoing surveillance [5]. Intermediate risk patients with high grade Ta tumors were not included.
The initial year of the model is shown in supplementary Figures 1 and 2.
Sensitivity analysis
In one-way sensitivity analysis, costs and medical assumptions were individually adjusted across a range of values to assess the impact of one variable on baseline assumptions. A tornado diagram was developed to evaluate the relative impact of different cost components (Fig. 1). Two-way sensitivity analyses were performed evaluating the least costly approach when vary 2 factors over a range (Figs. 2 and 3).
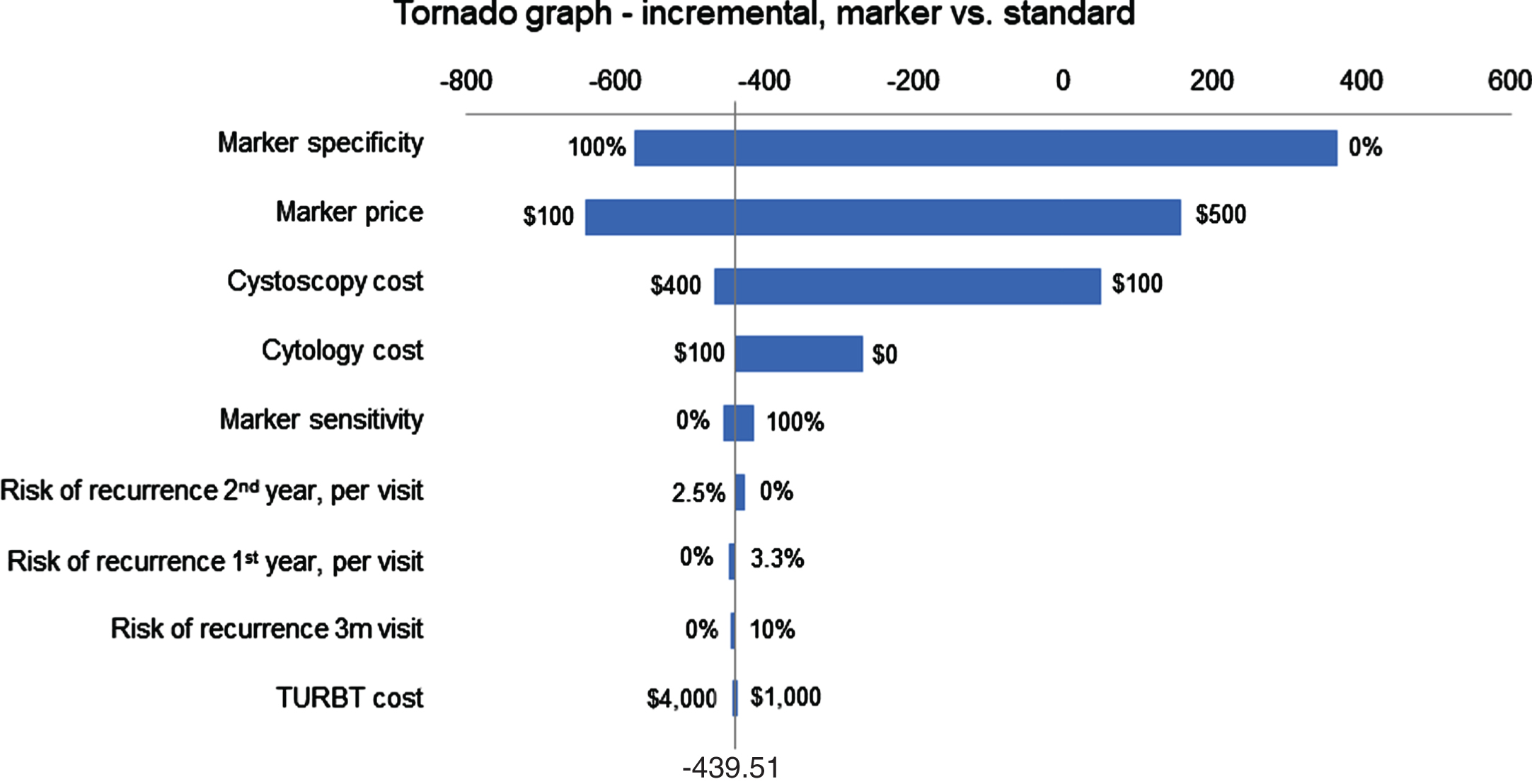
Tornado diagram using U.S. model evaluating incremental cost of Standard and Marker arms.
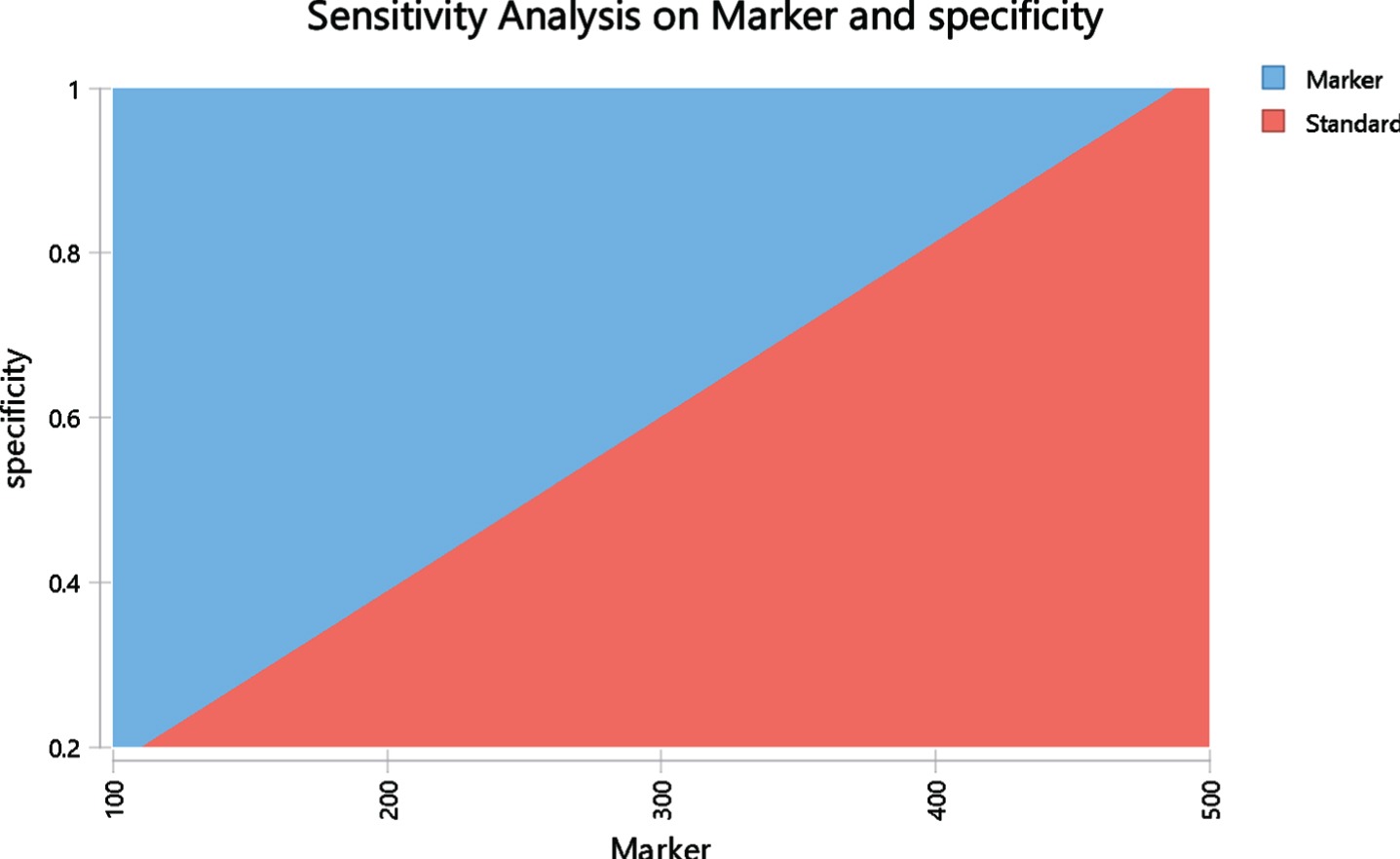
Two-way sensitivity analysis based on US model evaluating cost parity with varying cost of marker ($) and specificity of marker. The red area represents the area where the standard is less costly and the blue area is the area where the marker is less costly. The margin between these areas represent points where the costs are the same in both arms.
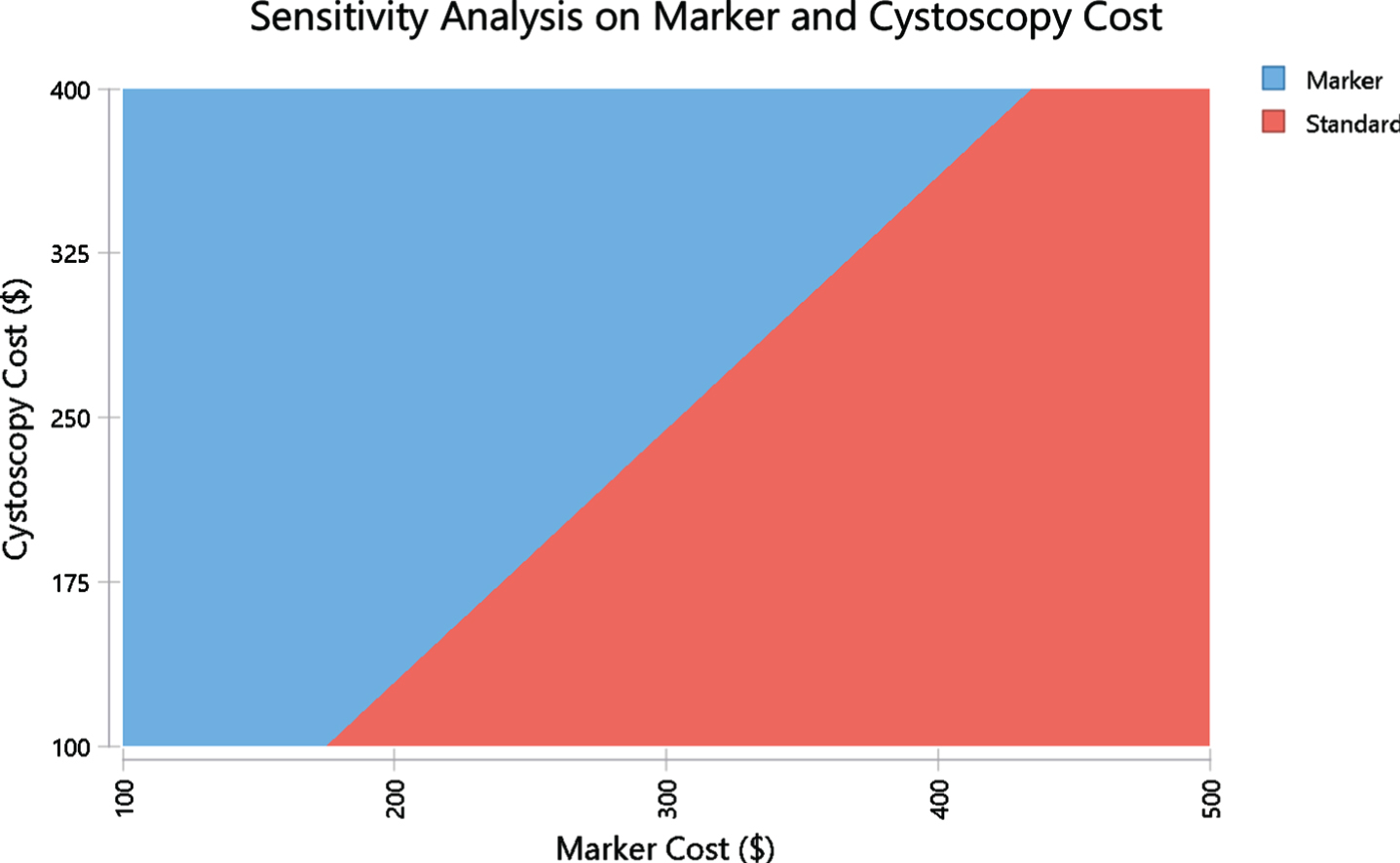
Two-way sensitivity analysis based on US model evaluating cost parity with varying cost of marker ($) and cost of cystoscopy ($). The red area represents the area where the standard is less costly and the blue area is the area where the marker is less costly. The margin between these areas represent points where the costs are the same in both arms.
RESULTS
The efficacy of the model was equivalent in terms of recurrence for each arm with median recurrence rate of 22%. The cost of each arm varied by country (Table 3). When assuming marker cost of 200 local currency, the marker arm was less expensive in the USA, Netherlands, Switzerland, Belgium, Italy, Austria and UK by 154€ to 329£ per patient for the 2-year period. Cost was slightly higher in France, Spain, and Germany by 33–103€ under the same assumptions. The marker cost parity point represents the cost of the marker at which the marker and standard arms are cost equivalent for each country (Table 3), and was between 148€ in Spain and $421 in the USA. The tornado diagram (Fig. 1) shows the impact of different factors on cost of different arms of the model (based on USA costs). Marker cost and specificity have the greatest impact on the overall model cost. A higher marker price leads to increase in marker arm costs. A lower specificity also increases cost since more patients in the marker arm require cystoscopy due to false positive results. The cost of cystoscopy is also an important factor since it impacts the standard arm cost more than marker arm.
Outcomes of model and marker cost parity point
*Standard arm is less expensive than marker arm.
We evaluated the number of cystoscopy procedures in each arm varying the specificity in the marker arm. In the baseline model, the average number of cystoscopy procedures per patient performed over 2 years was 5.2 (Fig. 4). With a specificity of 85.8% for the marker, the average number of cystoscopy procedures was 3.5. Lower marker specificity is associated with a greater number of cystoscopy procedures in marker arm.
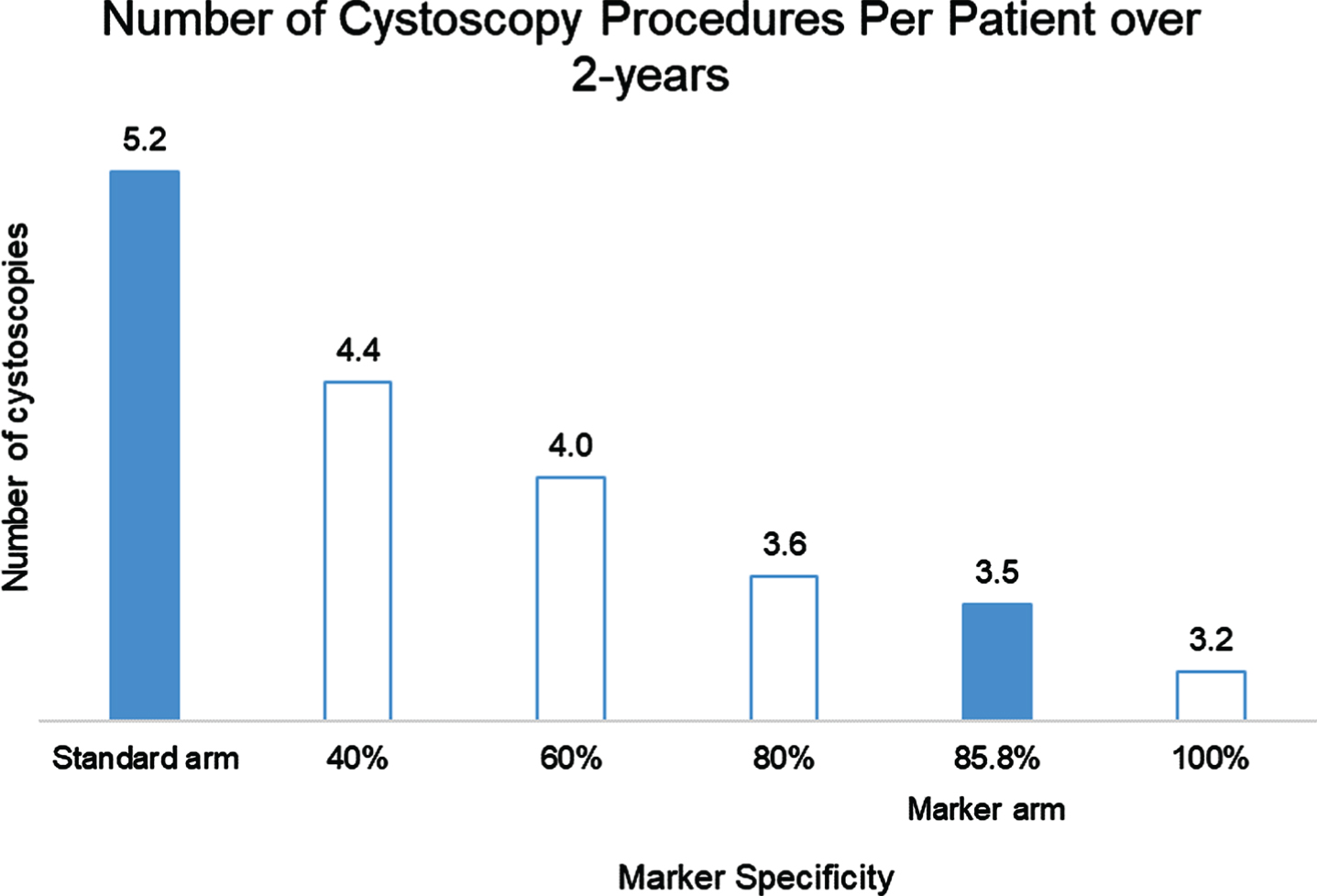
Number of cystoscopy procedures per patient over 2 years in Standard and Marker arms.
One way sensitivity analysis found that in the U.S. model, the marker arm becomes more expensive than the standard arm in case the specificity is below 38.9% or if cystoscopy costs less than $129.4. On the other hand, marker sensitivity had minimal impact on its cost-effectiveness with merely $10 difference in the cost-parity price between sensitivity of 20% and 80%. Two-way sensitivity analyses were performed to assess if marker or standard is more costly when varying marker specificity and price (Fig. 2). The red area represents the area where the standard is less costly and the blue area is the area where the marker is less costly. The margin between these areas represent points where the costs are the same in both arms. For example, when the specificity is 80%, marker is less expensive as long as marker is priced below $395. If specificity drops by 20% to 60% then the maximum marker price for cost-saving drops by ∼25% below $300. A two-way sensitivity analysis was also performed evaluating marker and cystoscopy cost using US model. It demonstrates cost points for cystoscopy and the marker where each arm is less costly. At current marker cost of $200 and cystoscopy average cost of $242, the marker arm is less costly.
DISCUSSION
Low grade non-invasive bladder cancers are common and many of these tumors are categorized as intermediate risk due to frequent recurrences, increased size or multiplicity. These low grade tumors rarely progress to invasive or high grade disease. Most guidelines recommend frequent cystoscopy procedures to identify recurrent tumors but there is both a financial cost and patient discomfort associated with these procedures [12]. The use of urine-based tumor markers to reduce the number of procedures is a meritable goal. In this model, we evaluated the use of Bladder EpiCheck® (Nucleix Ltd.) to alternate with cystoscopy in the surveillance of low-grade non-invasive bladder cancers over a 2-year period of time. We evaluated the cost of a marker-based approach compared to a standard approach in 10 countries. When assuming marker cost of 200 local currency, the marker arm was less expensive in the USA, Netherlands, Switzerland, Belgium, Italy, Austria and UK. Cost was slightly higher in France, Spain, and Germany where reported cystoscopy costs were less than 100€. In these countries cost parity was achieved when marker was priced 148€–184€. The use of Bladder EpiCheck reduced frequency and number of cystoscopies as well as reducing costs over a 2-year period of time. For example, in the US, the average cost benefit per patient was $439 and the number of cystoscopies was 3.47 in the marker arm vs. 5.19 in the standard arm.
When considering an alternative strategy to cystoscopy, the most important consideration is safety. There are several reasons why using a low-grade intermediate risk cohort makes sense. First, progression of disease within 3 to 6 months is exceedingly rare for these patients. Intermediate risk low grade patients have EORTC progression scores of 2–6. These patients are estimated to have a 1% risk of progression at 1 year [11]. In our model, patients even in the marker arm had a cystoscopy every 6 months so missed progression would be very low. It is even rarer for a low-grade Ta tumor to progress to muscle invasive disease so at worst likely could become HG Ta. Furthermore, the sensitivity of Bladder EpiCheck for high grade disease is 86.4% (95% CI 72.6%;94.8%) and an NPV of 98.8% (95% CI 97%–100%) for the detection of high-grade cancer [9]. As such, over a one-year period less than 1 patient per 1,000 patients would have a 3 month delay in diagnosing a high grade cancer. Missing a low-grade cancer has minimal impact on patient outcomes and there are even studies suggesting conservative management of non-invasive low grade tumors [13–15]. Using a marker alternating with cystoscopy has the advantage of reducing patient discomfort while still detecting cancers within reasonable timeframes. In most countries, the marker-based approach was less expensive since the marker was less expensive than cystoscopy especially when one considers the accompanying loss of work. Only in 3 countries where the cost of cystoscopy was reported to be less than 100€ was the standard arm less expensive and marker price had to be reduced to 148–184€ to achieve cost parity.
Sensitivity analyses were performed to determine the cost at which both arms were cost-equivalent. In Table 3, the cost of the marker at which the 2 arms (standard and marker based) were cost-equivalent ranged from 148€ to $421. Two-way sensitivity analyses were performed using the US model to identify the impact of varying marker cost with marker specificity and cystoscopy cost. These can be performed for each country to determine the impact of varying costs and marker performance. The tornado diagram identified the factors that have the highest impact on cost of each arm. While sensitivity had a minimal impact overall, specificity significantly impacts the model since any patient with a positive marker undergoes cystoscopy. Those patients have the cost of the marker and the cost of cystoscopy. As such, a test that results in many false positive results can dramatically increase the cost of a marker arm. Since the Bladder EpiCheck test has a specificity of 85.8%, this is an infrequent event.
There have been several prior models published evaluating the use of markers in surveillance of bladder cancer [16, 17]. These models did not focus on low grade intermediate-risk disease but found that markers could be cost-effective if marker cost was reasonable and that several markers had sufficiently good performance characteristics. There is still need to perform prospective studies to evaluate the safety of such an approach as well as assessing patient and physician acceptance [18]. There is an ongoing study (UroFollow) in which patients with pTa G1/G2 NMIBC, a tumor size less than 3 cm, and with no accompanying pTis are randomized (1:1) to either usual-care with cystoscopy or the marker arm which includes a urine marker and ultrasound [19]. The primary endpoint of UroFollow is to investigate, if marker-guided (noninvasive) follow-up of patients with low- and intermediate-risk NMIBC is equally effective to detect tumor recurrence and progression compared to standard surveillance using cystoscopy.
There are some additional considerations when using a marker in surveillance. While we assumed that white light cystoscopy has a 100% sensitivity and specificity, this is not accurate since there are both false negative results as demonstrated from literature regarding enhanced cystoscopy as well as the risk for false positive findings [20–22] It is also possible that a positive marker result may improve urothelial carcinoma detection rate using cystoscopy as seen in the CeFub trial [23]. There is a risk of finding abnormal lesions by cystoscopy that are not cancerous which could result in unnecessary biopsies adding cost and morbidity to the patient [19, 24]. This was not incorporated into the model but would be reduced in patients in the marker arm.
In this analysis we did not include the indirect costs of cystoscopy’s complications, such as urinary tract infection, urinary retention, etc. which can result in antibiotic treatment in the community and additional loss of work in the mild cases, or prolong hospitalization in the severe ones [25]. This biases the cost result against the marker arm. There are also indirect benefits of avoiding use of office assistants and appointment slots for the urologist while a potential downside of reducing profits from the procedure. We did not assess a strategy of avoiding cystoscopy altogether with longer intervals between surveillance visits. While this would reduce patient visits, there is little data on risks and benefits and impact on quality of life. While a marker adds costs, there is a high sensitivity for high grade disease and it provides some reassurance that a longer interval between cystoscopy is safe.
There are some inherent limitations in developing models. The assumptions used were based on best available literature and cost data acquired from varying countries. The accuracy of cost data is sometimes difficult to ascertain especially in very heterogeneous health care systems. Sensitivity analyses help with analyzing the variance but future studies using real world data will be necessary to determine the actual cost if these types of strategies are implemented. Quality of life is also an important factor in patients with NMIBC. There are benefits to patients with avoiding cystoscopy since it is an invasive procedure but there are potential issues with anxiety among patients who get a marker due to lower sensitivity. However, the impact of these considerations on quality of life are unknown. Future studies should include specific and validated questionnaires to evaluate the impact of a marker based strategy on quality of life. There is also another potential benefit of a urine marker based approach due to the COVID 19 pandemic since use of urine tests may reduce visits to the health care system leading to lower risk of exposure of patients and medical staff. Another limitation of the model is that we did not include the cost of intravesical therapies. Many patients are given intravesical therapies for intermediate risk bladder cancers but rates vary by provider and geography. The cost of intravesical therapies would be equivalent for both arms of the model and would not impact the conclusions.
A strategy alternating the urine marker Bladder EpiCheck with cystoscopy in the surveillance of patients with low grade intermediate risk bladder cancer is cost-equivalent in the US and European countries when the marker is priced 148€–$421. Prospective studies will be necessary to validate these findings.
Footnotes
ACKNOWLEDGMENTS
The authors have no acknowledgments
FUNDING
This analysis was funded by Nucleix Ltd.
AUTHOR CONTRIBUTIONS
ETHICAL CONSIDERATIONS
This study, as a literature review is exempt from any requirement for Institutional Review Board approval.
CONFLICT OF INTERESTS
All authors are consultants for Nucleix Ltd (YL, GG, MM, JM, HM, FP, CP, MR, CS, SS, AW)
Two figures are included in the supplementary material.
