Abstract
BACKGROUND:
Bladder cancer is one of the most common types of cancer diagnosed each year, and more than half of patients have non-muscle invasive bladder cancer (NMIBC). The standard of care for patients with high-grade NMIBC is Bacillus Calmette-Guerin (BCG). Unfortunately, multiple BCG shortages have limited access to this treatment. Available alternatives using intravesical administration of chemotherapy have some efficacy, but lack prospective validation and long-term outcomes. Development of novel intravesical therapies may provide more active alternatives to BCG for patients with high-grade NMIBC.
OBJECTIVE:
To develop an optimal imidazolium salt for the intravesical treatment of NMIBC and determine preliminary in vitro activity of anthraquinone-substituted imidazolium salts.
METHODS:
The development of the anthraquinone-substituted imidazolium salts was undertaken in an attempt to increase the potency of this class of compounds by incorporating the quinone functional group observed in the chemotherapeutics doxorubicin, valrubicin, and mitomycin. All compounds were characterized by 1H and 13C NMR spectroscopy and infrared spectroscopy. Furthermore, these imidazolium salts were tested for in vitro cytotoxicity by the Developmental Therapeutics Program (DTP) on the NCI-60 human tumor cell line screening. Additional in vitro testing was performed against diverse bladder cancer cell lines (RT112, TCCSUP, J82, and UMUC13) using CellTiter-Glo® assays and colony-forming assays.
RESULTS:
The NCI-60 cell line screening indicated that compound
CONCLUSIONS:
The most active lipophilic anthraquinone imidazolium salt, compound
INTRODUCTION
Bladder cancer is estimated to affect over 80,000 new patients nationally in 2020, with more than 17,000 deaths anticipated [1]. Additionally, bladder cancer is the 4th and 8th most prevalent cancer in men and women, respectively, with the most common stage at the time of diagnosis being non-muscle invasive bladder cancer (NMIBC) [2]. Many therapies are used for NMIBC, but few are effective at elimination of the disease in some patients. The current standard of care for NMIBC is intravesical treatment with Bacillus Calmette-Guerin (BCG). Although BCG is an effective therapy, it is currently underproduced, which puts many patients at risk for cancer recurrence, progression, and subsequent radical cystectomy [3, 4]. Immunotherapy isn’t the only option as intravesical chemotherapy can be used as a treatment for NMIBC. A problem with intravesical chemotherapy is that the treatment has been shown to lead to 62% recurrence in intermediate grade patients within 5 years, according to a study performed by the European Association of Urology [5, 6]. This may be circumvented with a treatment regimen of gemcitabine/docetaxel that has shown a reduction of recurrence, but lacks the evaluation of long term outcomes and prospective validation [7]. Furthermore, there are more difficult cases to treat with high-grade NMIBC such as carcinoma in situ (CIS), which have shown cancer progression when treated with intravesical chemotherapy [8]. We presume that one of the reasons for the reduced efficacy of intravesical chemotherapy is that the activity of the chemotherapeutic used is dependent on prolonged exposure in order to be incorporated into the cell in S-phase (during DNA replication) or mitosis, when most chemotherapies exert their toxicity. Prolonged exposure requires longer intravesical retention of the chemotherapy to achieve a therapeutic effect, which is not practical for most patients.
Due to the numerous BCG shortages and the reduced efficacy of intravesical chemotherapy compared to BCG, novel chemotherapeutics are needed to treat bladder cancer. Development of novel anticancer materials is a necessity to combat bladder cancer and improve patient outcomes. We recently developed an imidazolium salt (
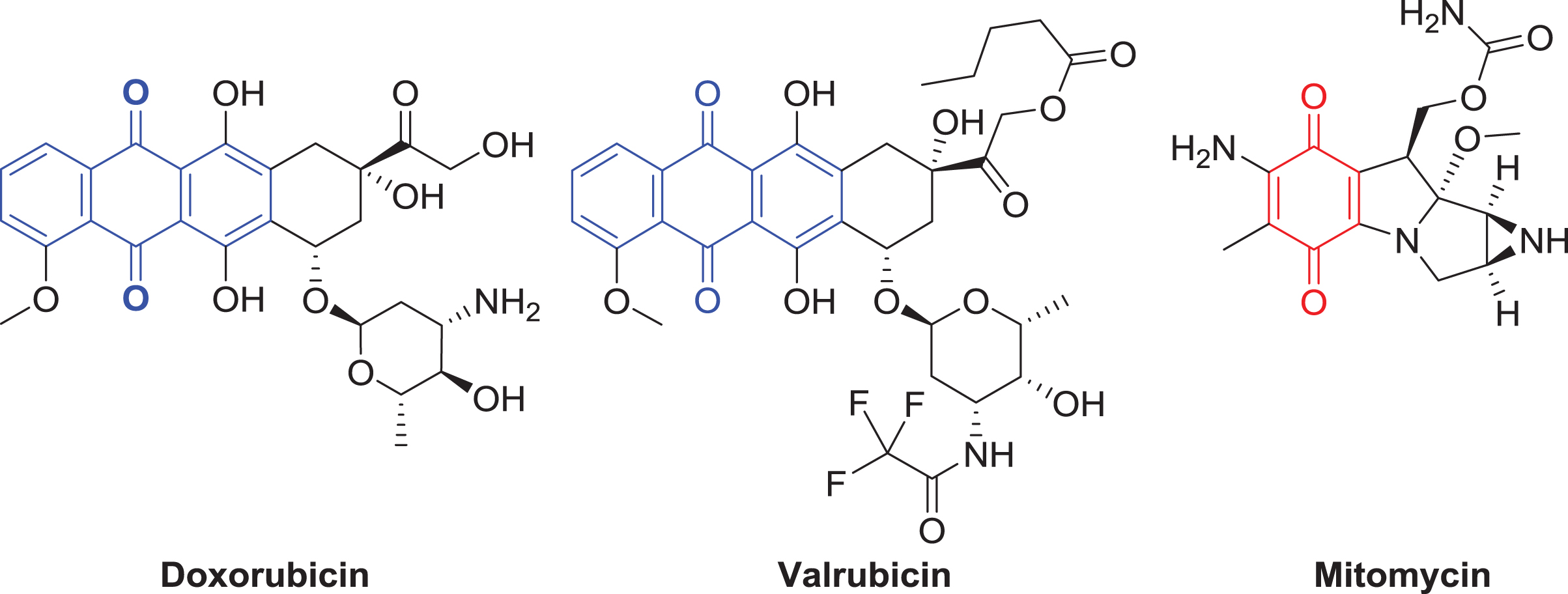
Examples of current chemotherapeutics that contain anthraquinone (3-ring system) and quinone (the basic functional group of a single conjugated cyclic dione) moieties are illustrated above and are highlighted with dashed bond lines. The structures of doxorubicin, valrubicin, and mitomycin are shown.
MATERIALS AND METHODS
General considerations
All reactions were performed aerobically unless otherwise stated. Solvents and chemical reagents were unmodified and purchased from VWR, Fisher Scientific, or Sigma Aldrich. The synthesis of starting material
X-ray crystallographic analysis
Crystals of the compounds were coated in Paratone oil, mounted on a CryoLoop and placed on a goniometer under a stream of nitrogen. Crystal structure data sets were collected on a Bruker Kappa APEX II Duo CCD system equipped with a Mo IμS source and a Cu IμS micro-focus source equipped with QUAZAR optics (λ= 1.54178 Å). The unit cells were determined by using reflections from three different orientations. Data sets were collected using APEX II software packages. All data sets were processed using the APEX II software suite [12, 13]. The data sets were integrated using SAINT [14]. An empirical absorption correction and other corrections were applied to the data sets using multi-scan SADABS [15]. Structure solution, refinement, and modelling were accomplished by using the Bruker SHELXTL package [16]. The structures were determined by full-matrix least-squares refinement of F2 and the selection of the appropriate atoms from the generated difference map. Hydrogen atom positions were calculated and Uiso(H) values were fixed according to a riding model.
DTP NCI-60 cell line screening
The developmental therapeutics program (DTP) treatment protocol is summarized as follows: 60 cell lines ranging from leukemia to breast cancer are treated with a 10μM concentration of the compound. The results are presented as growth percent. Continued growth is quantified from 0 to 100 and lethality is scored from –100 to 0. Further information on the experimental procedure for cell treatment can be viewed at NCI-DTP website (https://dtp.cancer.gov/discovery_development/nci-60/methodology.htm).
CellTiter-Glo® assay
Bladder cancer cell lines (RT112, TCCSUP, J82, and UMUC13) were seeded at 10,000 cells/well in black-walled 96 well plates and incubated for 24 h. Cells were treated with compound
Colony-forming assay
Cells were seeded at 500 cells/well in 6-well plates and 24 h later treated with 25μM, 50μM or 100μM of compound
Synthesis of 2-(bromomethyl)-9,10-anthraquinone (1)
2-(Bromomethyl)-9,10-anthraquinone (compound
Synthesis of 2-((imidazol-1-yl)methyl)-9,10-anthraquinone (2)
Imidazole (0.308 g, 4.52 mmol) was stirred at reflux in acetonitrile (10 mL) with potassium hydroxide (0.281 g, 5.01 mmol). After 15 minutes compound
Synthesis of 1,3-bis((9,10-anthraquinone-2-yl)methyl)imidazolium bromide (3)
Compound
Crystal data for
Synthesis of 1-((9,10-anthraquinone-2-yl)methyl)-3-(naphthalen-2-ylmethyl)imidazolium bromide (4)
Compound
Synthesis of 2-((benzimidazol-1-yl)methyl)-9,10-anthraquinone (5)
Benzimidazole (0.506 g, 4.52 mmol) was stirred at reflux in acetonitrile (10 mL) with potassium hydroxide (0.273 g, 5.01 mmol). After 30 minutes,
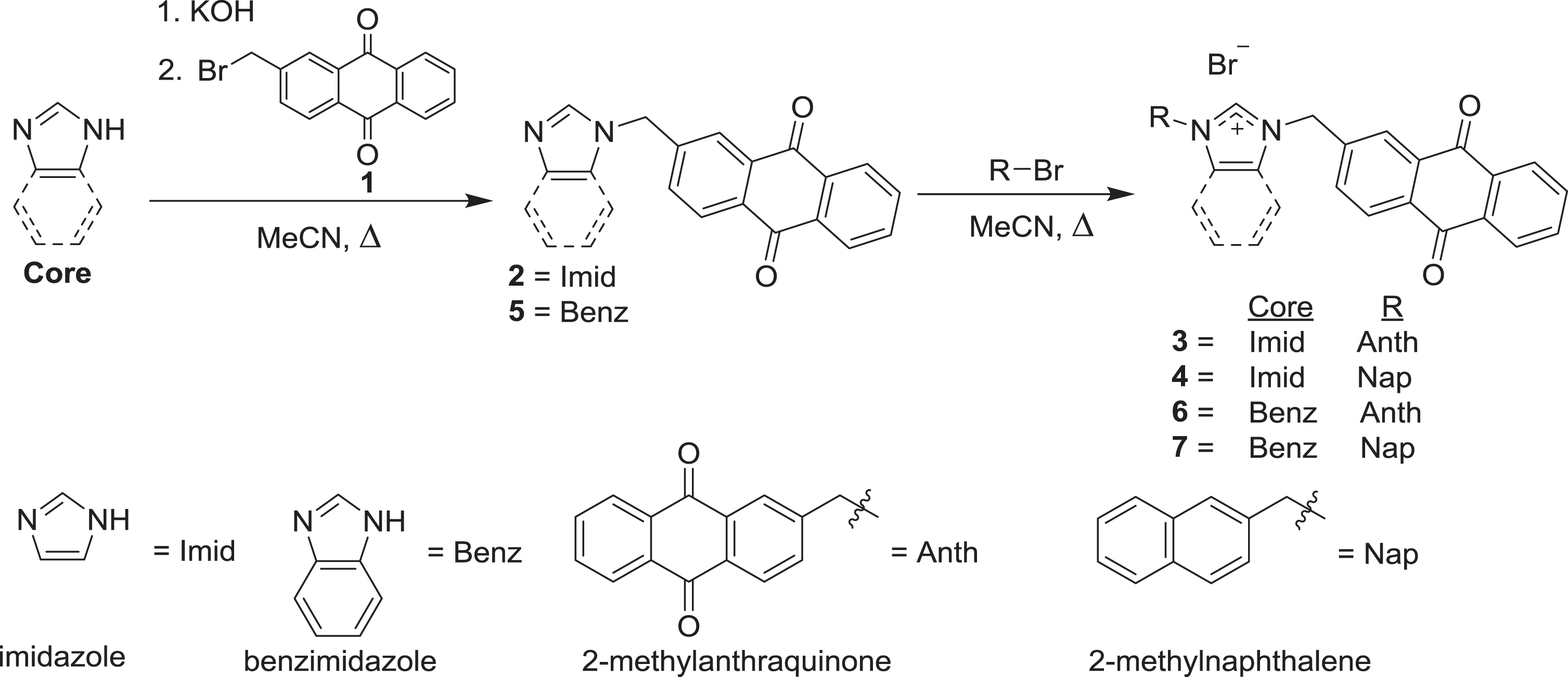
Syntheses of compounds
Synthesis of 1,3-bis((9,10-anthraquinone-2-yl)methyl)benzimidazolium bromide (6)
Compound
Synthesis of 1-((9,10-anthraquinone-2-yl)methyl)-3-(naphthalen-2-ylmethyl)benzimidazolium bromide (7)
Compound
Crystal data for compound
RESULTS AND DISCUSSION
Synthesis and characterization
The syntheses of the anthraquinone-substituted imidazolium salt compounds
The anthraquinone benzimidazolium salts, compounds
To confirm the structures of the compounds discussed above, single crystal X-ray diffraction was used. Crystals suitable for analysis were obtained from vapor diffusion of ethyl ether into N,N-dimethylformamide solutions and only structures for compounds
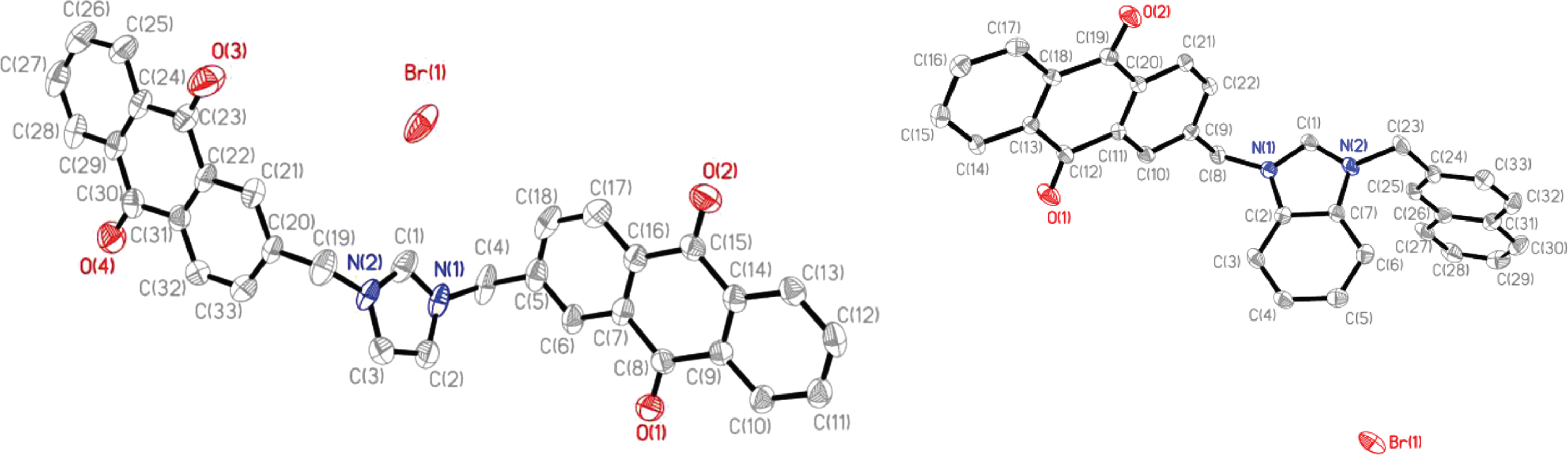
Thermal ellipsoid plots of
In vitro biological evaluation of anthraquinone compounds as anticancer agents
The in vitro biological activity of compounds
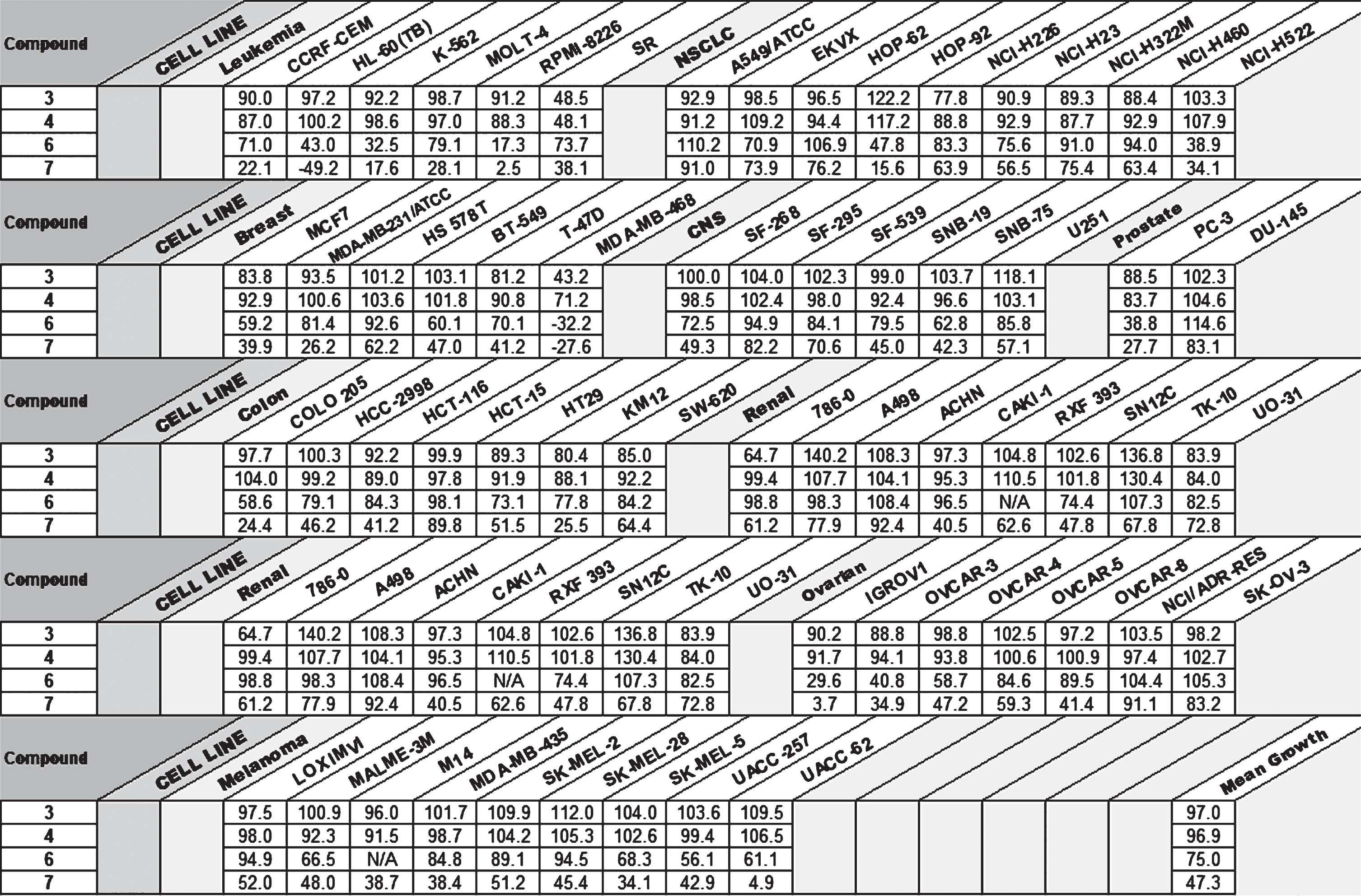
One Dose NCI-60 Cell Line Screening for Compounds 3,4,6, and 7 (GI%)
The asymmetric compounds
With preliminary data indicating the increased cytotoxicity of compound
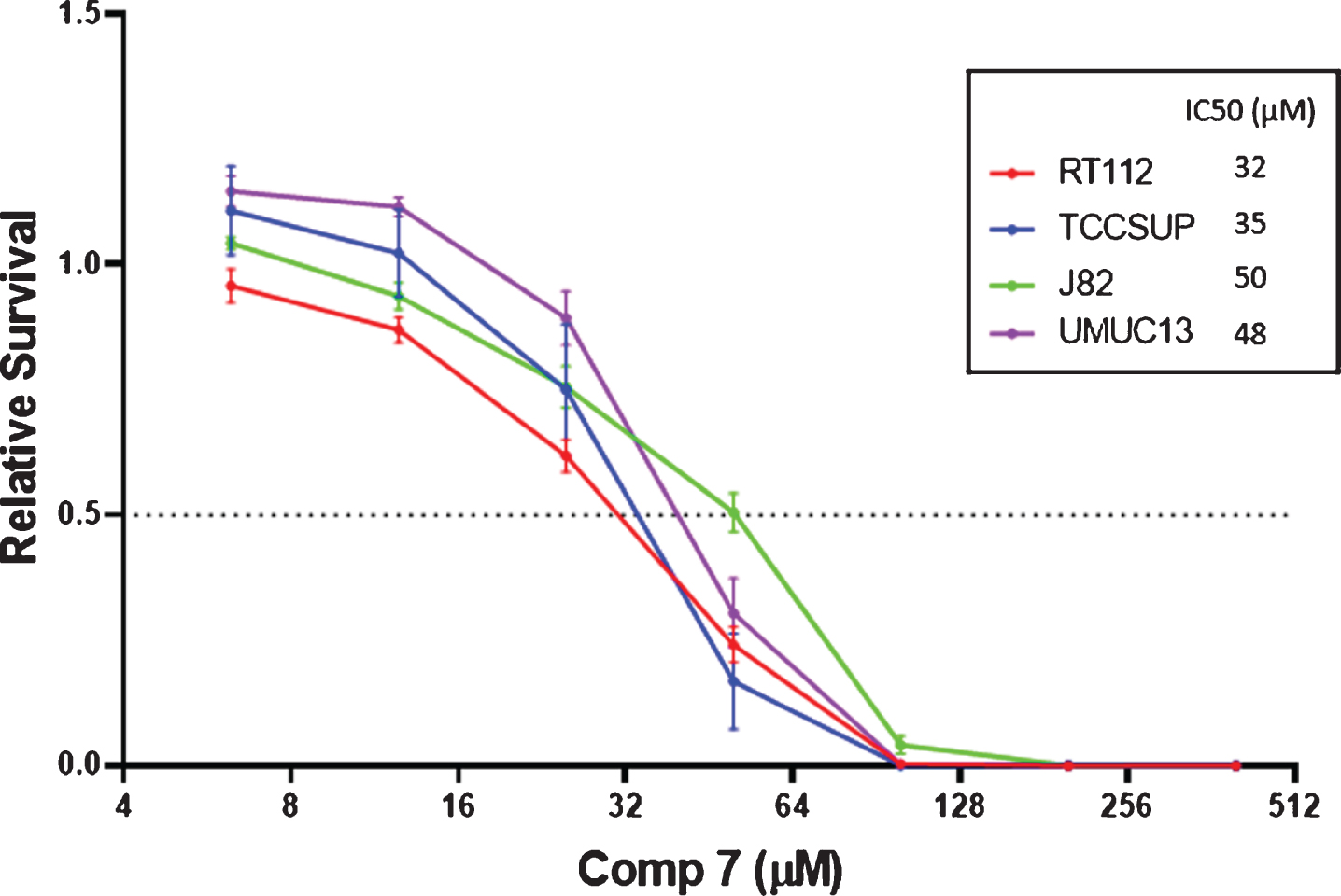
IC50 values determined by the CellTiter-Glo® assay for a 1 h exposure of select bladder cancer cell lines to compound
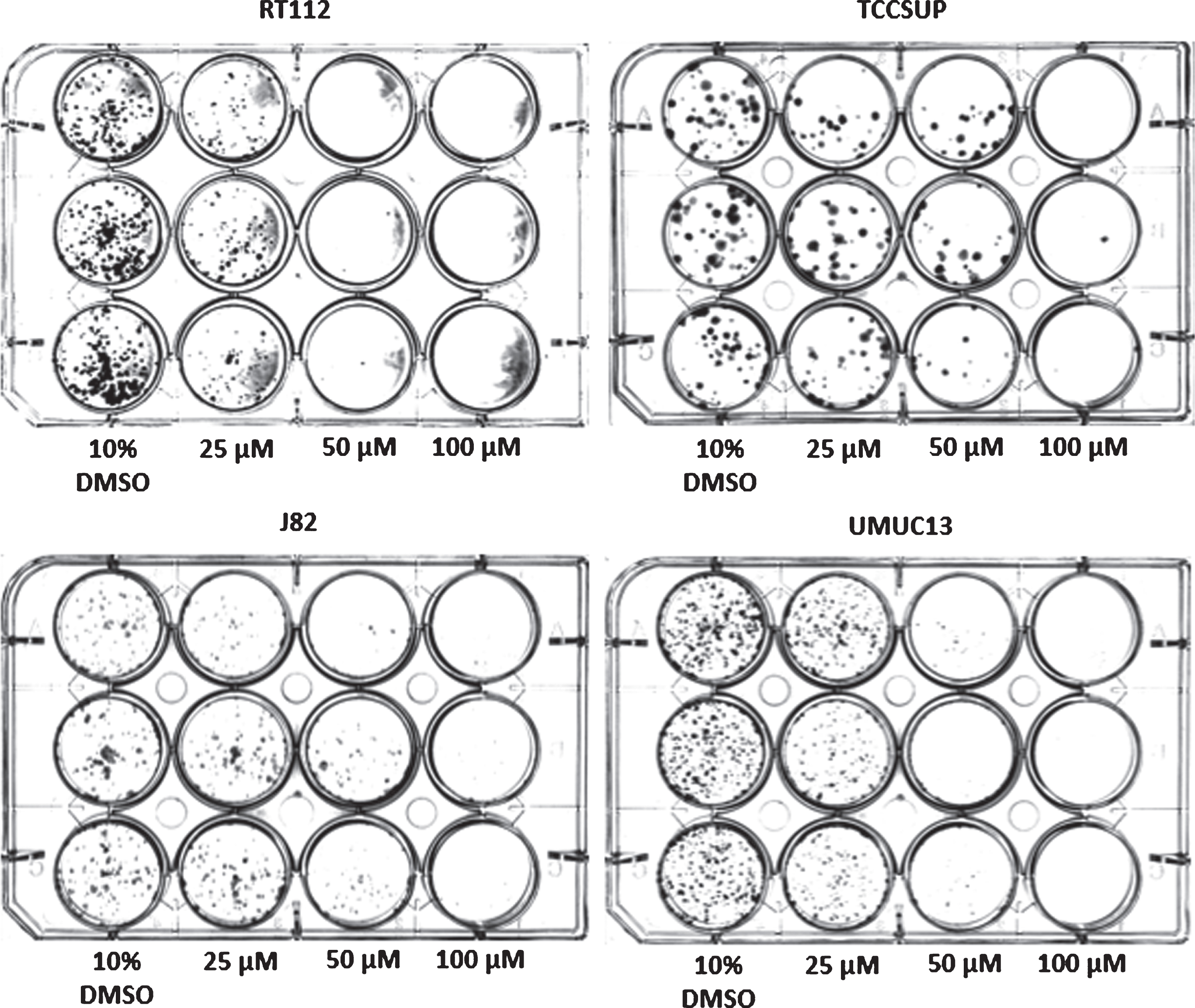
Colony-forming assay of select bladder cancer cell lines 10 days after a 1 h treatment with compound
The long-term cytotoxicity of short-term exposure to compound
CONCLUSION
In this work, a series of anthraquinone-substituted imidazolium salts was synthesized. The bisanthraquinone imidazolium and benzimidazolium salts, when tested by DTP on the NCI-60 cell line screening, yielded little inherent activity against all cell lines at the 10μM test concentration. Even though these compounds are lipophilic, the correlation between the naphthalene substituent and activity is apparent, as compounds
SUPPLEMENTARY INFORMATION
One dose NCI-60 human tumor cell line screening data for compounds
Crystallographic Information File for
Footnotes
ACKNOWLEDGMENTS
The authors would like to thank NCI’s DTP for performing the 60 human tumor cell line screening with their one dose assay for compounds
FUNDING
This work was funded by the following: PHA-BCAN Young Investigator Award, US-Avery Postdoctoral Fellowship, and the Fox Chase Center Core Grant (P30 CA006927). Additional funding for instrumentation was provided by the National Science Foundation (NSF) for NMR spectrometers (Nos. CHE-0341701 and DMR-0414599) and the X-ray diffractometer (CHE-0840446) used in this work. Also supported by Fox Chase Cancer Center Core grant P30 CA006927.
AUTHOR CONTRIBUTIONS
Conceptualization, M.S. and W.Y.; development and structural characterization, M.S.; biological investigation, D.W., U.S., and P.A.; writing-original draft preparation, M.S.; writing-review and editing, M.S., D.W., U.S., P.A., and W.Y. All authors have read and agreed to the published version of the manuscript.
CONFLICTS OF INTEREST
The authors declare no conflict of interest.