Abstract
Bladder cancer (BC) is a significant global health concern, with non-muscle invasive bladder cancer (NMIBC) comprising 75% of cases at diagnosis. High-risk NMIBC (HR-NMIBC) poses a significant therapeutic challenge due to its high recurrence and progression rates despite Bacillus Calmette-Guerin (BCG) therapy. Radical cystectomy remains the gold standard for BCG-unresponsive cases but is often met with considerable morbidity and patient reluctance. This has driven research into alternative bladder-sparing therapies (BSTs). Emerging BSTs include immune checkpoint inhibitors like pembrolizumab and novel agents such as nadofaragene firadenovec and nogapendekin alfa inbakicept (IL-15). These therapies have demonstrated promising response rates in clinical trials, offering potential for disease management while preserving bladder function. Gene therapies and targeted agents like CG0070 and EG-70 are also gaining traction for their innovative mechanisms. However, most data are derived from early-phase, single-arm studies, necessitating larger, randomised trials for validation. Device-assisted strategies, including hyperthermic and electromotive drug delivery systems, show potential to enhance intravesical therapy efficacy. Despite advancements, challenges remain in balancing efficacy, safety, and cost-effectiveness within diverse healthcare settings. This narrative review highlights the evolving landscape of BSTs for HR-NMIBC, emphasising the need for robust clinical evidence to refine patient selection and optimise outcomes.
Introduction
Bladder cancer (BC) is now the 9th most common cancer worldwide, rising from 10th place in 2020. 1 Mortality in men is nearly 3 times more common than women, with future projections suggesting a sustained rise in the number of deaths in men up to 54 percent and a 2-to-3 fold increase for women by the year 2040. 2 Three-quarters of BC patients present with non-muscle invasive bladder cancer (NMIBC) at diagnosis, inclusive of non-invasive carcinoma confined to the epithelium or mucosa (Ta), tumours infiltrating the lamina propria (T1), with or without carcinoma in situ (CIS/Tis).
The heterogenous landscape of bladder cancer lends itself to multiple definitions for high-risk bladder cancer per the European Association of Urology (EAU) and the American Urological Association (AUA) in conjunction with the Society of Urologic Oncology (SUO). High-risk bladder cancer involves an amalgamation of the following features: T1 disease, high-grade histology, CIS, age of more than 70 years-old, index tumour size of 3 centimetres or more, multifocal tumours, variant histology and lymphovascular or prostatic urethral invasion.3,4
Up to 60% of high-risk NMIBC (HR-NMIBC) may recur after 1 year, necessitating a more aggressive approach to further management and follow-up. 5 In particular, high-risk NMIBC and CIS are associated with a poor prognosis and progression to muscle-invasive disease. Across the spectrum of CIS, clinical outcomes are poorer with diffuse CIS, prostatic urethral CIS, or concomitant CIS in the context of existing T1 disease, with the latter raising the risk of 1-year progression from 10% to 29%, and 5-year progression from 29% to 75%.5,6
Intravesical bacillus Calmette-Guerin (BCG) induction and maintenance is recommended for HR-NMIBC as first-line treatment to mitigate this risk, as based on the SWOG-8507 trial, which demonstrated 60% recurrence-free survival (RFS) at 5 years in patients who received induction and subsequent maintenance BCG for over 3 years. 7 While the exact mechanism of BCG is incompletely understood, it elicits recruitment of natural killer (NK) cells, CD4 and CD8 lymphocytes and granulocytes, causing cytotoxicity to cancer cells via mediation of the humoral immune response. 8 Although 80 percent of high-risk NMIBC (HR-NMIBC) shows an initial response to BCG, almost half still experience high-grade recurrence or progression, with up to 20 percent progressing to muscle-invasive disease within 2 years.9,10 In addition, patients with progressive MIBC fare poorer than those with de novo MIBC, with a 5-year overall survival of 37% versus 49% respectively even after radical cystectomy. 11
Currently, radical cystectomy (RC) remains the standard of care for fit patients with BCG-unresponsive or very high-risk disease. 4 However, RC is associated with significant morbidity, mortality as well as altered body image, and is considered unacceptable by many as treatment despite thorough counselling on the risk of disease progression.12,13 This, alongside the increased recognition of BCG-unresponsive disease as well as the recent worldwide shortage of BCG has led to further inquiry into alternative bladder-sparing therapies (BST), particularly in the context of HR-NMIBC.
Valrubicin, previously approved by the US Food and Drug Administration (FDA) in 1998 for use in NMIBC after BCG, showed complete response (CR) rates of 13.5% and 8% at 1- and 2-years respectively. However, this was limited by a heterogenous study population and a lack of specific treatment indications, particularly in the context of updated definitions of HR-NMIBC or BCG-unresponsive disease. 14 As of July 2023, there are currently three further FDA-approved BST for BCG-unresponsive NMIBC – pembrolizumab, nadofaragene firadenovec, and nogapendekin alfa inbakicept (IL-15), although the role of early adjuvant bladder-sparing therapy in lieu of BCG remains to be defined.10,15,16 This review aims to highlight and provide an overview of emerging bladder-sparing strategies for high-risk NMIBC.
Methods
For the purpose of this narrative review, clinical trials for high-risk NMIBC were queried by the National Library of Medicine (NLM) (www.clinicaltrials.gov) between the years 2000 and 2024. Search criteria agreed by the study team included the keywords “non muscle invasive bladder cancer”, “high risk bladder cancer”, “carcinoma in situ”, “interventional”, “phase 2 and 3”, in conjunction with BCG status (“BCG refractory”, “BCG unresponsive”, “BCG exposed”, and “BCG naïve”). Other novel agents and updated interim trial analysis was also identified via a search of abstracts (including “late-breaking data” and “trials in progress”) and presentations from meetings of the Society of Urologic Oncology (SUO), European Society for Medical Oncology (ESMO), and American Society of Clinical Oncology (ASCO) over the years 2020 to 2024. Relevant trials which were ongoing, as well as completed, were categorised by BCG status for discussion. Trials which were withdrawn, terminated due to poor accrual or suspended were excluded from detailed discussion. This study was exempt from ethics approval as the content for this review comprised existing published data without primary data collection, patient interaction or potential breaches of confidentiality.
Results and discussion
While radical cystectomy remains the standard of care for BCG-unresponsive or very high risk disease as defined by the EAU, the latest recommendations from the IBCG suggest that BCG non-responders may be considered for bladder-sparing therapy in the context of clinical trials, or in highly selected populations following detailed counselling and informed consent. These patients are divided into two groups by disease pathology: CIS with or without papillary disease, versus Ta or T1 disease. Options for the former include intravesical gemcitabine and docetaxel, nadofaragene firadenovec, nogapendekin alfa inbakicept (also known as NAI, N-803 or superagonist IL-15), and pembrolizumab. Options for the latter are similar, but also include hyperthermic mitomycin-C, as well as single-agent chemotherapy. 14 Figure 1 summarises 12-month complete response rates from interim analysis and published data on monotherapy, combination therapy and BCG-specific combination therapy in BCG-unresponsive NMIBC. A more detailed summary of results from recent and ongoing trials into bladder sparing therapy is provided in Table 1.
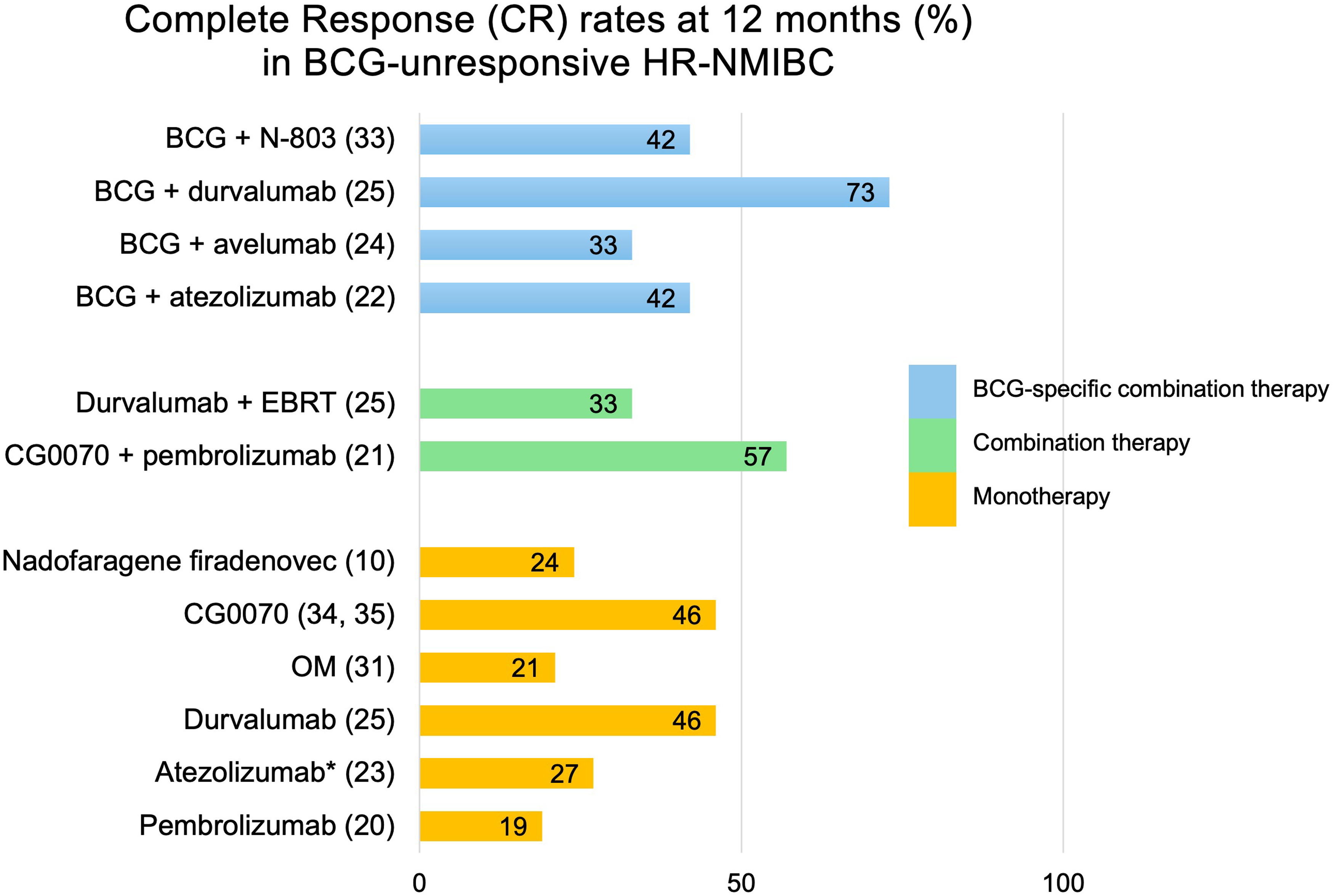
Complete Response rates at 12 months in BCG-unresponsive HR-NMIBC
Results from clinical trials for high-risk non-muscle invasive bladder cancer (including interim analysis).
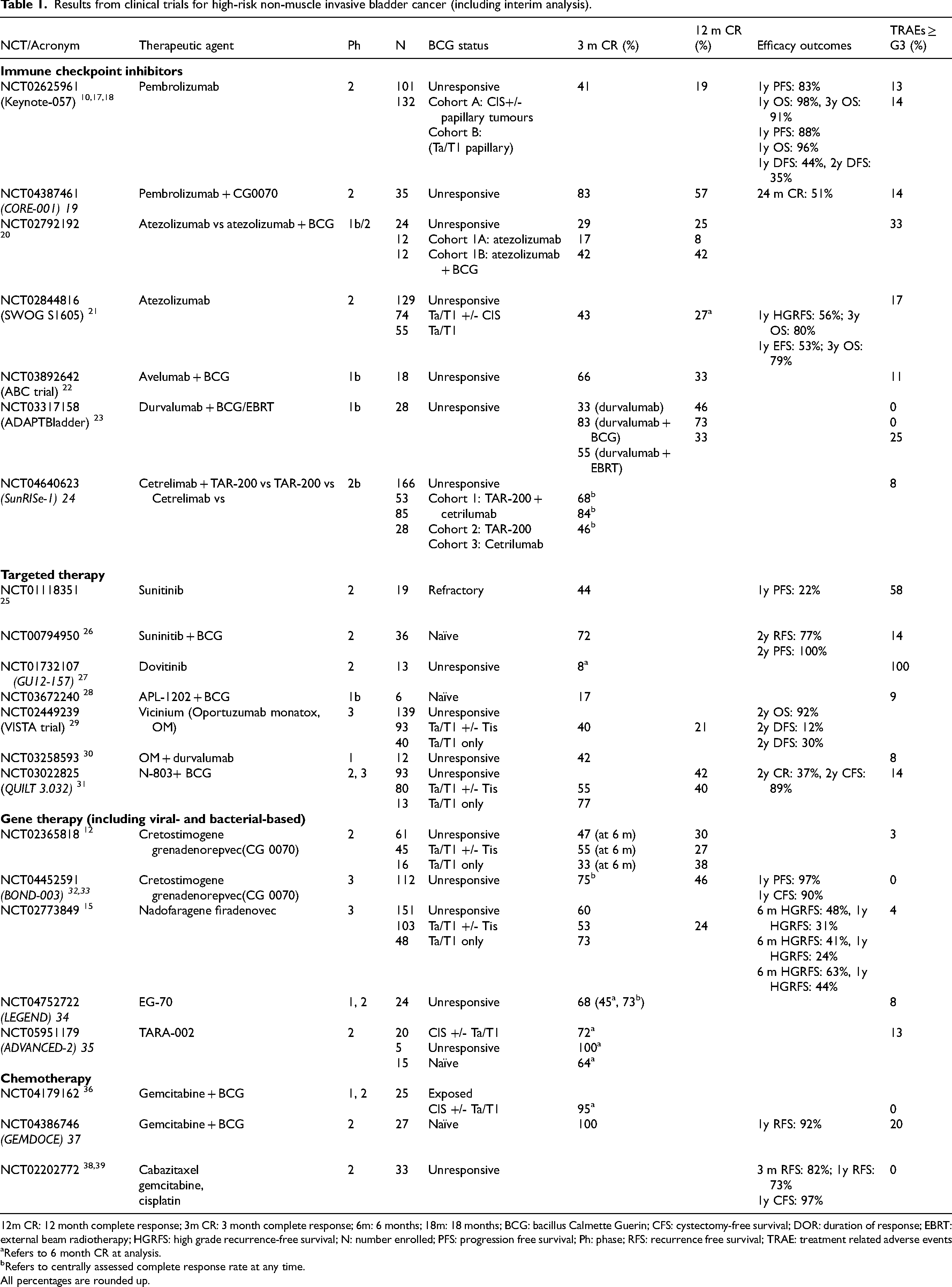
12m CR: 12 month complete response; 3m CR: 3 month complete response; 6m: 6 months; 18m: 18 months; BCG: bacillus Calmette Guerin; CFS: cystectomy-free survival; DOR: duration of response; EBRT: external beam radiotherapy; HGRFS: high grade recurrence-free survival; N: number enrolled; PFS: progression free survival; Ph: phase; RFS: recurrence free survival; TRAE: treatment related adverse events
Refers to 6 month CR at analysis.
Refers to centrally assessed complete response rate at any time.
All percentages are rounded up.
Immunotherapy
In the post-BCG era, current and emerging bladder-sparing strategies include immune checkpoint inhibitors (ICIs), targeted therapies, viral- and bacterial-based therapy and gene therapy. ICIs currently dominate the landscape of evolving research, either as monotherapy or in combination with BCG. There are 3 main immune checkpoints under further investigation in HR-NMIBC: PD1 (programmed cell death-1), PDL1 (programmed cell death ligand-1) and CTLA-4 (cytotoxic T-lymphocyte antigen 4). Inhibition of these checkpoints, particularly in the context of NMIBC with high tumour mutational burden (TMB), is beneficial in upregulating T-cell response against tumour cells.40,41 BCG unresponsive tumours have also demonstrated increased expression of PD1 and PDL1. Correspondingly, PDL1 positivity was virtually absent in BCG responders, forming the basis for further research. 42 A summary of ongoing and completed trials on ICI in high-risk NMIBC is presented in Table 2.
Clinical trials evaluating immune checkpoint inhibitors for high-risk non-muscle-invasive bladder cancer.
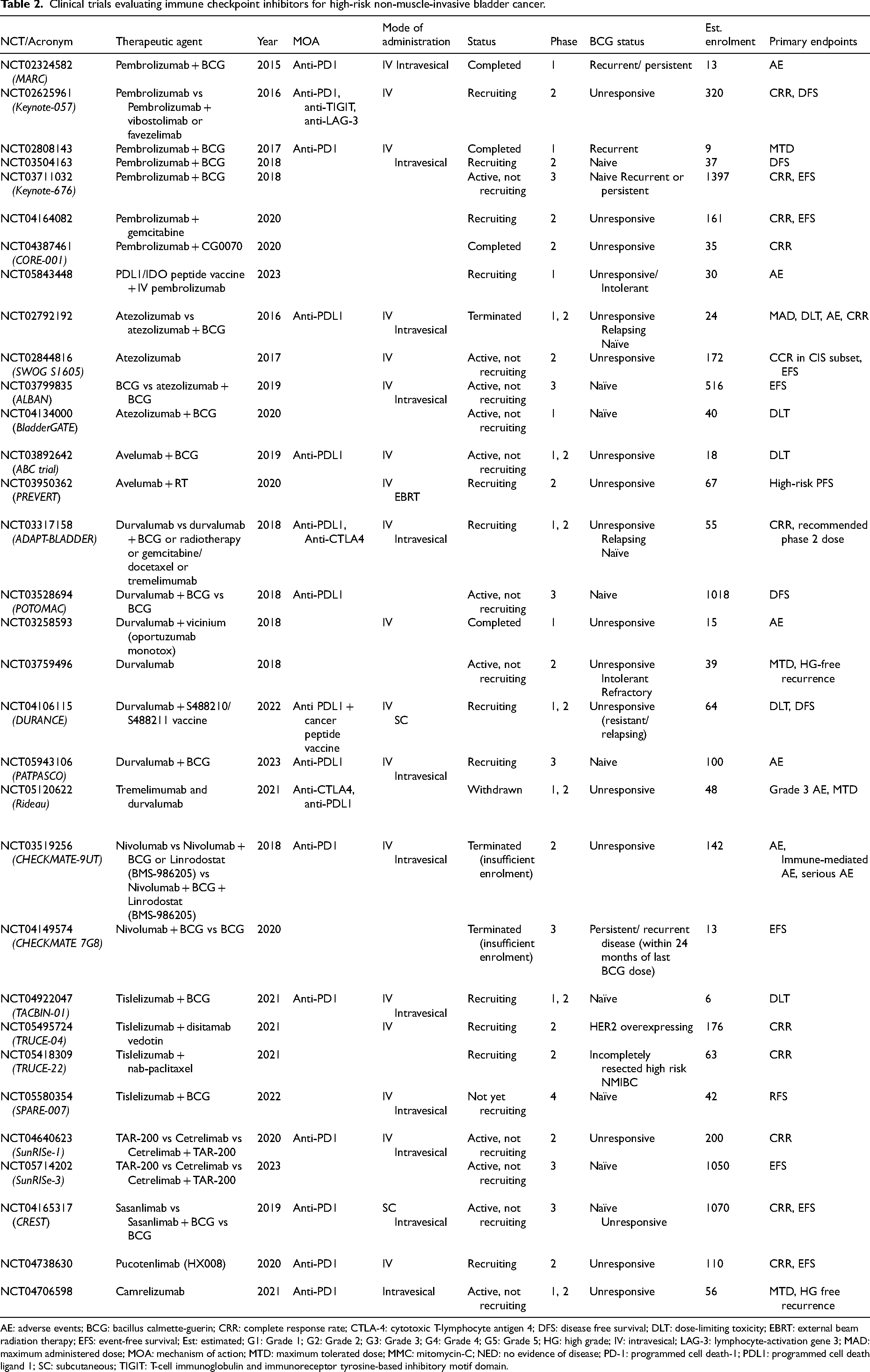
AE: adverse events; BCG: bacillus calmette-guerin; CRR: complete response rate; CTLA-4: cytotoxic T-lymphocyte antigen 4; DFS: disease free survival; DLT: dose-limiting toxicity; EBRT: external beam radiation therapy; EFS: event-free survival; Est: estimated; G1: Grade 1; G2: Grade 2; G3: Grade 3; G4: Grade 4; G5: Grade 5; HG: high grade; IV: intravesical; LAG-3: lymphocyte-activation gene 3; MAD: maximum administered dose; MOA: mechanism of action; MTD: maximum tolerated dose; MMC: mitomycin-C; NED: no evidence of disease; PD-1: programmed cell death-1; PDL1: programmed cell death ligand 1; SC: subcutaneous; TIGIT: T-cell immunoglobulin and immunoreceptor tyrosine-based inhibitory motif domain.
Pembrolizumab
Pembrolizumab, a PD1 inhibitor, has shown the most promise so far in the treatment of high-risk NMIBC via the ongoing randomised Phase II clinical trial KEYNOTE-057 (NCT02625961), which formed the basis of its recent FDA approval. Outcomes from 101 patients in Cohort A (CIS with or without papillary tumours) showed a 41% and 19% complete response (CR) at 3 and 12 months respectively, with 83% progression-free survival (PFS) at 1 year and a favourable safety profile of 13% treatment-related adverse events (TRAEs) which were Grade 3 or higher. Cohort B (Ta or T1, papillary) showed similar TRAEs at 14%, with a 1 year PFS of 88%..10,17,18 The results of combination ICI in Cohort C (pembrolizumab and combination ICI therapy with TIGIT inhibitor vibostolimab or LAG-3 inhibitor favezelimab) are currently pending.
Pembrolizumab has also shown positive results in combination with cretostimogene grenadenorepvec (CG0070) gene therapy in the CORE-001 trial (NCT04387461). This phase II trial of 35 patients with BCG-unresponsive CIS with or without concurrent papillary disease reported 3- and 12-month CR rates of 83% and 57% respectively. AEs were comparable to monotherapy in the Keynote-057 trial with TRAEs ≥3 of 14%. 19 Pembrolizumab is currently under investigation as combination therapy with other agents such as BCG (NCT02808143, NCT03504163, NCT03711032), gemcitabine (NCT04164082), and the PDL1/IDO (indoleamine 2,3-dioxygenase) peptide vaccine (NCT05843448). Of these, 2 are ongoing trials seeking to expand the role of pembrolizumab with combination BCG therapy in BCG naïve disease – Keynote-676 (NCT03711032), a phase III trial exploring CR and event-free survival (EFS) in the context of BCG naïve, recurrent or persistent disease; as well as NCT03504163, a phase II trial awaiting outcomes on disease-free survival (DFS).
Atezolizumab
Atezolizumab is an antibody that blocks PD1/PDL1 pathways. The phase Ib/II trial NCT02792192 has shown favourable outcomes in BCG unresponsive NMIBC with combination therapy with atezolizumab and adjuvant BCG, with a 3 month CR of 42%, sustained at 1 year. Overall CR rates at 1 year were 25% versus 8% for the monotherapy arm. However, almost a third of patients experienced TRAEs ≥ Grade 3, raising concerns regarding long-term safety. 20 The SWOG S1605 trial (NCT02844816) on the other hand, showed a lower frequency of adverse events at 17%, with 27% CR at 6 months in the TaT1 and CIS subgroup with 56% high-grade recurrence-free survival (HGRFS) at 1 year). 21
Ongoing trials include the phase III ALBAN trial (NCT03799835) of 516 BCG naïve patients with the primary endpoint of event-free survival (EFS), and the phase Ib BladderGATE trial (NCT04134000) of 40 patients assessing dose-limiting toxicity (DLT).
Avelumab
Avelumab is an anti-PDL1 inhibitor. Associated studies have focused on BCG-unresponsive NMIBC, with results from the Phase Ib ABC trial (NCT03892642) of 18 patients showed 66% CR at 3 months, halved at 1 year follow-up. The overall incidence of treatment-related adverse effects higher than Grade 3 was 11%. 22 The ongoing phase II, single-arm PREVERT trial aims to assess high-risk progression-free survival (PFS) of avelumab with radiotherapy (60–66 Gy in 2-Gy fractions).
Durvalumab
Durvalumab is a human monoclonal IgG kappa anti-PDL1 antibody which is currently being investigated for both BCG naïve and unresponsive high-risk NMIBC. In the phase Ib ADAPTbladder trial (NCT03317158) of 28 patients, durvalumab in combination with BCG in unresponsive disease showed superior CR at 73% at 1 year, in comparison with monotherapy (46%) or in combination with external beam radiation therapy (33%). 23
Ongoing trials on BCG unresponsive disease include NCT03258593 on HG DFS with durvalumab monotherapy; DURANCE (NCT04106115) which aims to assess ICI with S488210/S488211 cancer peptide vaccine in terms of disease-free survival, as well as NCT03258593, which aims to evaluate safety and efficacy of combined durvalumab and oportuzumab monotox. POTOMAC (NCT03528694) and PATPASCO (NCT05943106) are ongoing trials in the BCG naïve arena, exploring DFS and AE respectively with combined durvalumab and BCG therapy.
Cetrelimab
Cetrelimab is an anti-PD-1 monoclonal antibody currently under investigation in combination with TAR-200, an intravesical drug delivery system which facilitates the continuous release of gemcitabine into the bladder. 43 Results of the SunRISe-1 trial of 166 patients with BCG unresponsive NMIBC showed 84% CR at 5 months with TAR-200 alone, versus 68% at the same interval with combination therapy and 46% with cetrelimab alone. The safety profile was favourable with only 8% of patients experiencing reportable AEs Grade 3 or higher. 24 The SunRISe-3 trial (NCT05714202), a multicentre phase III trial of BCG-naïve patients with HR-NMIBC aiming to assess event-free survival, is currently ongoing with recruitment planned at 254 sites. 44
Sasanlimab
Sasanlimab is an immunoglobulin G4 monoclonal antibody which selectively binds PD-1, thereby blocking the interaction between PD-1 and PD-L1 expressed by cancer cells. 45 Of note, it can be administered via subcutaneous injection every 4 weeks, rendering it a potentially more convenient option compared to its intravenous counterparts. The phase III CREST study (NCT04165317) of sasanlimab in combination with BCG versus BCG alone in BCG-naïve HR-NMIBC is ongoing and aims to assess EFS and CR as primary endpoints. 46 Preliminary updates are positive with respect to EFS with further data awaited.
Characteristics of further trials involving other immune checkpoint inhibitors such as nivolumab, tislelizuma, pucotenlimab and camrelizumab are summarised in Table 2. Of note, the Checkmate-9UT (NCT03519256) and Checkmate 7G8 (NCT04149574) studies were terminated early due to insufficient enrolment. Intravesical camrelizumab has also shown potential in a phase II trial of 14 patients, with a median EFS of 12.7 months at median follow-up of 23.1 months and a 12-month EFS of 50%. TRAEs ≥ grade 3 were comparable with contemporary studies on pembrolizumab at 14%. 47
Targeted therapy
Nogapendekin alfa inbakicept
Nogapendekin alfa inbakicept, also known as NAI or N-803, is a interleukin-15 (IL-15) superagonist complex which enhances the effect of CD8+ T cells and natural killer cells. 48 NAI was theorised to provide a synergistic effect in promoting the action of BCG via by stimulating both adaptive and immune responses, leading to the phase I/IIb trial, QUILT-2.005 (NCT02138734), which is ongoing and due for completion in 2038. Results from interim analysis showed a 100% CR in 7 BCG-naïve patients at 24 months with a lasting duration of response, with patients remaining disease-free for a period of 8.3 to 9.2 years. 49
The phase II/III QUILT 3.032 trial explored efficacy outcomes of NAI and BCG in BCG-unresponsive NMIBC, and demonstrated an any-time CR of 71% in patients with CIS, outperforming other current FDA-approved agents such as nadofaragene (53% CR) and pembrolizumab (41% CR). 59% of patients maintained CR for a minimum of 12 months, with 48% DFS at 2 years and almost 90% of responders were able to avoid cystectomy over the course of this study.16,31 NAI alone was effective in only 2 of 10 participants in that arm, cementing its role in high-risk NMIBC as a promoter of immune response to be used in combination rather than as monotherapy.
86% of reported TRAEs from QUILT 3.032 were grade 1 to 2, with follow-up quality of life (QOL) analysis showing 7 and 9% of patients reported improvement from baseline well-being by weeks 78 and 104. 50
Tyrosine kinase inhibitors
Tyrosine kinase inhibitors (TKIs) target the vascular endothelial growth factor (VEGF) axis, thereby restricting angiogenesis in bladder cancer and resulting in anti-tumour activity. 34 One phase II trial of BCG and sunitinib showed 72% CR at 3 months in patients with BCG-naïve HG-NMIBC. Rates of recurrence and progression were low, with RFS of 77% and PFS of 100% at 2 years. 14% of patients reported TRAEs ≥ G3. 26 Zahoor et al. then explored the utility of sunitinib and BCG-refractory disease in 19 patients in a Phase II single-arm study (NCT01118351). At 3-month follow-up, 44% responded and 50% showed disease progression. Furthermore, 21.1% experienced TRAEs requiring cessation of treatment, with G3 toxicities noted in 58% of patients. Overall, sunitinib was not deemed to significantly improve clinical outcomes in BCG-refractory NMIBC. 25
Dovitinib is a multi-target inhibitor theorised to exert anticancer and anti-angiogenic activity via further inhibition of fibroblast growth factor receptor (FGFR) and platelet-derived growth factor receptor (PDGFR). 51 Another phase II trial (NCT01732107) on dovitinib in BCG unresponsive disease was terminated in view of poor CR of 8% at 6 months and safty concerns, with 100% of patients reporting adverse events. 27
Erdatinifib is a pan-fibroblast growth factor receptor (FGFR) tyrosine kinase inhibitor. FGFR3/2 gene mutations and fusions are found in 60–70% of patients with low-risk NMIBC and in more than 30% of patients with high-risk papillary NMIBC. 52 Following the results of the phase II BLC2001 trial, erdatifinib has established a role in treatment of patients with locally advanced or metastatic urothelial carcinoma with susceptible FGFR mutations who have progressed after initial SOC chemotherapy. 53 THOR-2 (NCT04172675) is a Phase II multicohort trial seeking to expand the utility of erdatifinib in the space of localised, intermediate and high-risk NMIBC with FGFR3/2 alterations. Cohort 1 assessed the response of erdatifinib versus intravesical chemotherapy in 73 recurrent, BCG-treated, papillary-only high-risk NMIBC patients with FGFR3/2 alterations unsuitable or unwilling for RC. RFS at 6- and 12-months was 96% and 77% for erdafitinib, compared to 73% and 41% for chemotherapy, respectively. Median follow up for RFS was 13.4 across both arms and not reached for the erdatifinib arm. 54 The results from Cohort 2 (BCG-unresponsive CIS with or without papillary disease) and Cohort 3 (intermediate-risk NMIBC) are currently pending.
APL-1202
Methionine aminopeptidases 2 (MetAP2) are metalloproteases shown to positively influence angiogenesis and tumourigenesis. 55 APL-1202 is an orally available MetAP2 inhibitor which was previously evaluated in a phase II trial for BCG-relapsing or chemo-unresponsive patients (NCT04498702), reporting a 54.3% RFS at 1 year and a median RFS of 14.7 months. 56 Of the patients studied, 7.3% (3 in 41 patients) had concurrent CIS. There were no reported TRAEs. The ACCRUE Phase III trial (NCT04490993) is currently being conducted to evaluate the safety and efficacy of APL-1202 in combination with intravesical epirubicin hydrochloride in intermediate and chemo-resistant high-risk NMIBC patients.
Epithelial cell adhesion molecule (EpCAM) antibodies
EpCAMs are intercellular adhesion molecules expressed by cancer cells. 57 They are highly expressed in many tumour tissues, including urothelial carcinoma of the bladder, where they are involved in tumorigenesis, bearing a strong association with high-grade, advanced-stage BC with poor overall survival.58,59 In contrast, they are weakly expressed in normal urothelium. Oportuzumab monatox (also known as vicinium) is an intravesical antibody-drug conjugate (ADC)-like recombinant fusion protein of the anti-EpCAM antibody and Pseudomonas exotoxin A. Upon internalisation, it binds to cancer cells, releasing exotoxin A and inducing apoptosis. 60
The phase III single-arm VISTA trial (NCT02449239) of oportuzumab monatox monotherapy was conducted in 139 patients with BCG-refractory NMIBC with complete response as its primary endpoint. The CIS cohort comprised 93 patients, and showed a 40% CR at 3 months with a median duration of response of 9.4 months, with half of responders remaining free of CIS at 1 year. 2-year overall survival across CIS and non-CIS cohorts was 92%..29,61 Investigations into combination therapy are underway, with a phase I trial (NCT03258593) investigating oportuzumab monatox and durvalumab showing 42% CR at 3 months in 12 patients, and 8% incidence of TRAEs Grade 3 or higher. 30
Catumaxomab (CAT), an EpCaM monoclonal antibody, has also been tested in 2 patients with recurrent BCG NMIBC with a good safety profile and cystoscopically confirmed recurrence-free intervals of 32 and 25 months. 62 The phase I CATUNIBLA trial (NCT04819399) followed in BCG-naïve high-risk NMIBC patients. Patients with intermediate and high risk NMIBC after index TURBT underwent 6 weekly intravesical CAT instillations, followed up 2nd TURBT and adjuvant BCG instillation as standard of care. Initial results showed CR as best response in 7 patients, with no recurrence in the cohort receiving a higher dosage of 70 μg (versus 50 μg).. 63 Further interim analysis of 27 patients showed overall CR for of 96%, with a high CR rate of 69% in the CIS subgroup before BCG treatment. Recurrence rate was 15% at a mean follow-up of 13.5 months. 64 Overall, results are encouraging for both agents for patients with high-risk NMIBC.
Enfortumab vedotin
Enfortumab vedotin (EV) is an antibody-drug complex targeting Nectin-4, which is highly expressed on bladder cancer cells. Systemic use of EV in EV-301 has shown an OS benefit over chemotherapy in patients with locally advanced or metastatic urothelial carcinoma following previous platin-based or PD1/PDL1 immunotherapy, making it a third line option in treatment sequencing. 65 EV-104 (NCT05014139) is an ongoing phase 1, multicentre trial in patients with high-risk BCG unresponsive NMIBC who are decline or are ineligible for RC. Primary outcomes include adverse events and dose-limiting toxicities and this study is due for completion by 2028. 66 An overview of trials on targeted therapy in high-risk NMIBC is provided in Table 3.
Clinical trials evaluating targeted therapies for high-risk non-muscle-invasive bladder cancer.
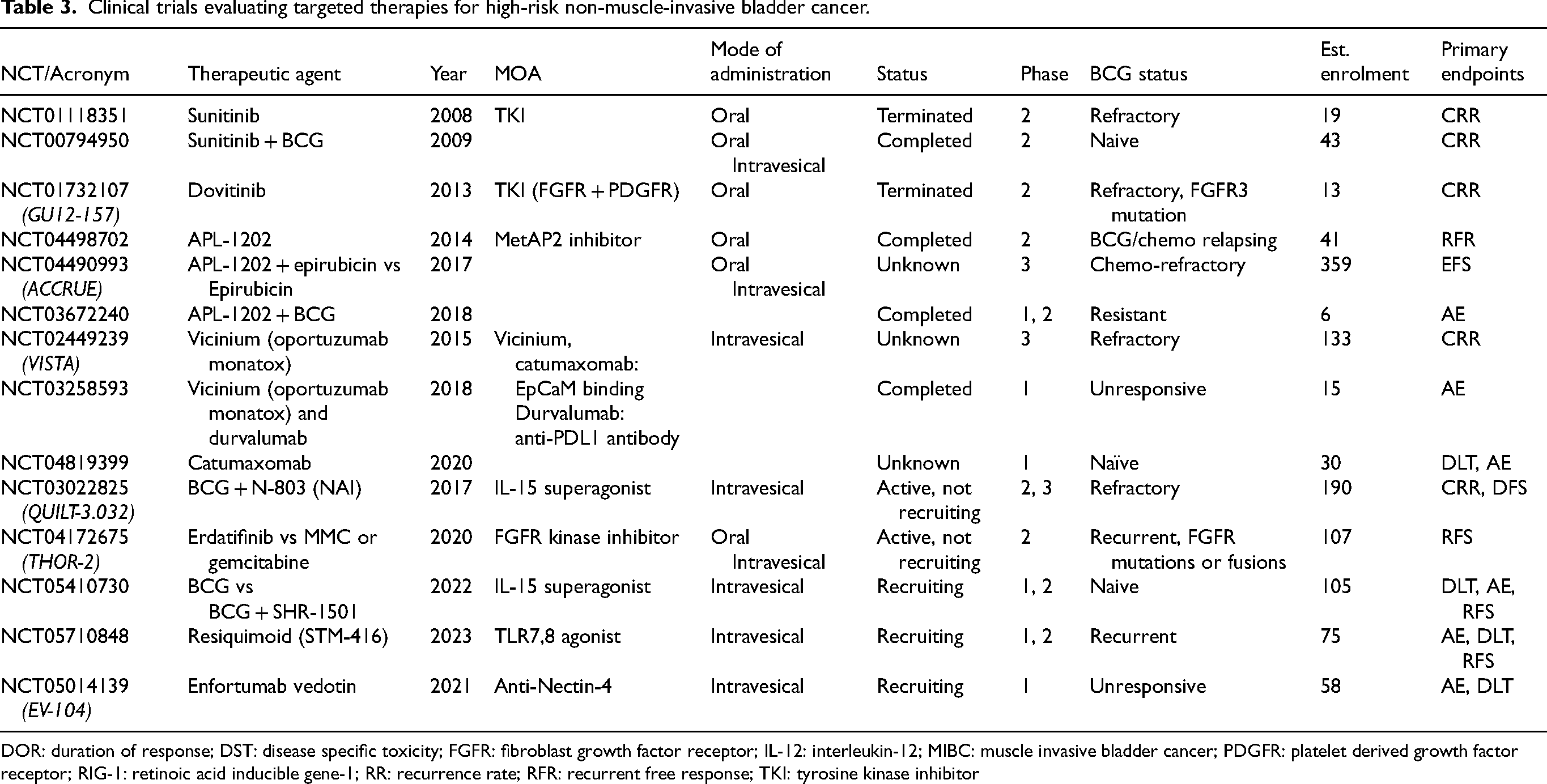
DOR: duration of response; DST: disease specific toxicity; FGFR: fibroblast growth factor receptor; IL-12: interleukin-12; MIBC: muscle invasive bladder cancer; PDGFR: platelet derived growth factor receptor; RIG-1: retinoic acid inducible gene-1; RR: recurrence rate; RFR: recurrent free response; TKI: tyrosine kinase inhibitor
Gene therapy (including viral- and bacterial-based)
Nadofaragene
Nadofaragene firadenovec is an adenovirus vector-based intravesical gene therapy which delivers interferon-α to urothelial cells, limiting proliferation and encouraging apoptosis via tumour necrosis factor (TNF)-related apoptosis-inducing ligand (TRAIL) activation. 67
As of December 2022, it is the first gene therapy approved for urological cancers by the US FDA. Benefits of this therapeutic agent include a convenient treatment schedule of 3-monthly instillations, with intravesical administration facilitating direct contact between the gene therapy vector and cancer cells.
Intravesical nadofaragene has been tested in a phase III study (NCT02773849) of 151 BCG-unresponsive patients with the benefit of recently published extended 5-year follow-up. CR at 3 months was 73% for the non-CIS cohort and 53% for the CIS cohort, with 24% of CIS patients remaining disease-free at 1 year. HG RFS at 1 year in the CIS and non-CIS cohorts was 24% and 44% respectively, and 13% and 33% at 57 months follow-up. 5-year cystectomy-free survival (CFS) of 43% in the CIS cohort and 59% in the Ta/T1 cohort. Overall drug tolerability was good with only 4% of significant TRAEs during initial reporting, with no grade 4 or 5 AEs at 5-year follow-up. 68
CG0070
The retinoblastoma (Rb) pathway is essential in regulating cell proliferation. Rb pathway defects result in increased proliferation and invasion from suppressed p53 and caspase-3 signalling, which inhibits apoptosis. CG0070 (also known as cretostimogene grenadenorepvec) is an oncolytic serotype-5 adenovirus which is selective for Rb-pathway defective tumours and exerts anticancer effects via granulocyte macrophage colony-stimulating factor gene and E2F-1 gene signalling. 69 CG0700 monotherapy was previously assessed in a phase II study of 61 patients with BCG unresponsive disease (NCT02365818). Overall CR was 47% at 6 months, and 55% and 33% respectively in the CIS and non-CIS cohorts, suggesting that patients with CIS may stand to benefit the most from CG0070 therapy. 12 The risk profile of CG0070 also appears favourable, with only 3% of patients experiencing TRAEs ≥ G3. Interim results from the ongoing phase III BOND-003 trial (NCT04452591) in BCG-unresponsive disease demonstrated a 75% CR at any-time, and 46% CR and 97% PFS at 1 year.32,33 The promising 3 month CR rates, as well as its potential additional therapeutic yield in CIS, led to the study of combination CG0070 and pembrolizumab for BCG unresponsive disease in the phase II CORE-001 trial (NCT04387461), which showed a 51% CR at 2 years, with a side effect profile comparable to pembrolizumab monotherapy at 14%. 19
EG-70
EG-70 is a novel non-viral gene therapy which encodes for interleukin-12 (IL-12) and activators of the immune receptor retinoic acid-inducible gene-1 (RIG-1) which in turn triggers adaptive and innate immune responses respectively. 70 It is administered intravesically to ensure a localised immune response and targeted delivery to urothelial cancer cells within the bladder. The LEGEND study (NCT04752722) in BCG unresponsive patients reported CRs of 68% at 3 months, 45% at 6 months and 73% at any time during the study. 34 Phase II of this trial is currently in the recruitment phase and aims to expand the study population to BCG unresponsive, BCG naïve and those who have received incomplete BCG therapy, with the primary endpoints of adverse events and complete response rates.
TARA-002
TARA-002 is an edited strain of Streptococcus pyogenes that targets toll-like receptor 4 (TLR-4) and is an intravesical therapy currently under investigation in both BCG-unresponsive and naïve high risk NMIBC. ADVANCED-1 (NCT05085977) is a phase Ia trial assessing dose-limiting treatment, adverse events and the recommended phase II dose; ADVANCED-2 (NCT05951179) is a phase I/II trial which currently recruiting with the endpoint of assessing complete response in high grade disease. Interim results from the ADVANCED-2 trial showed an overall CR of 72% at 6 months, and 100% CR in the BCG-unresponsive cohort as compared to 64% in the BCG-naïve cohort, suggesting an increased benefit in BCG unresponsive disease. 35 Estimated study completion is in the year 2030. A summary of salient active and ongoing trials in the space of gene therapy is provided in Table 4.
Clinical trials evaluating gene therapy (including viral- and bacterial-based) for high-risk non-muscle-invasive bladder cancer.
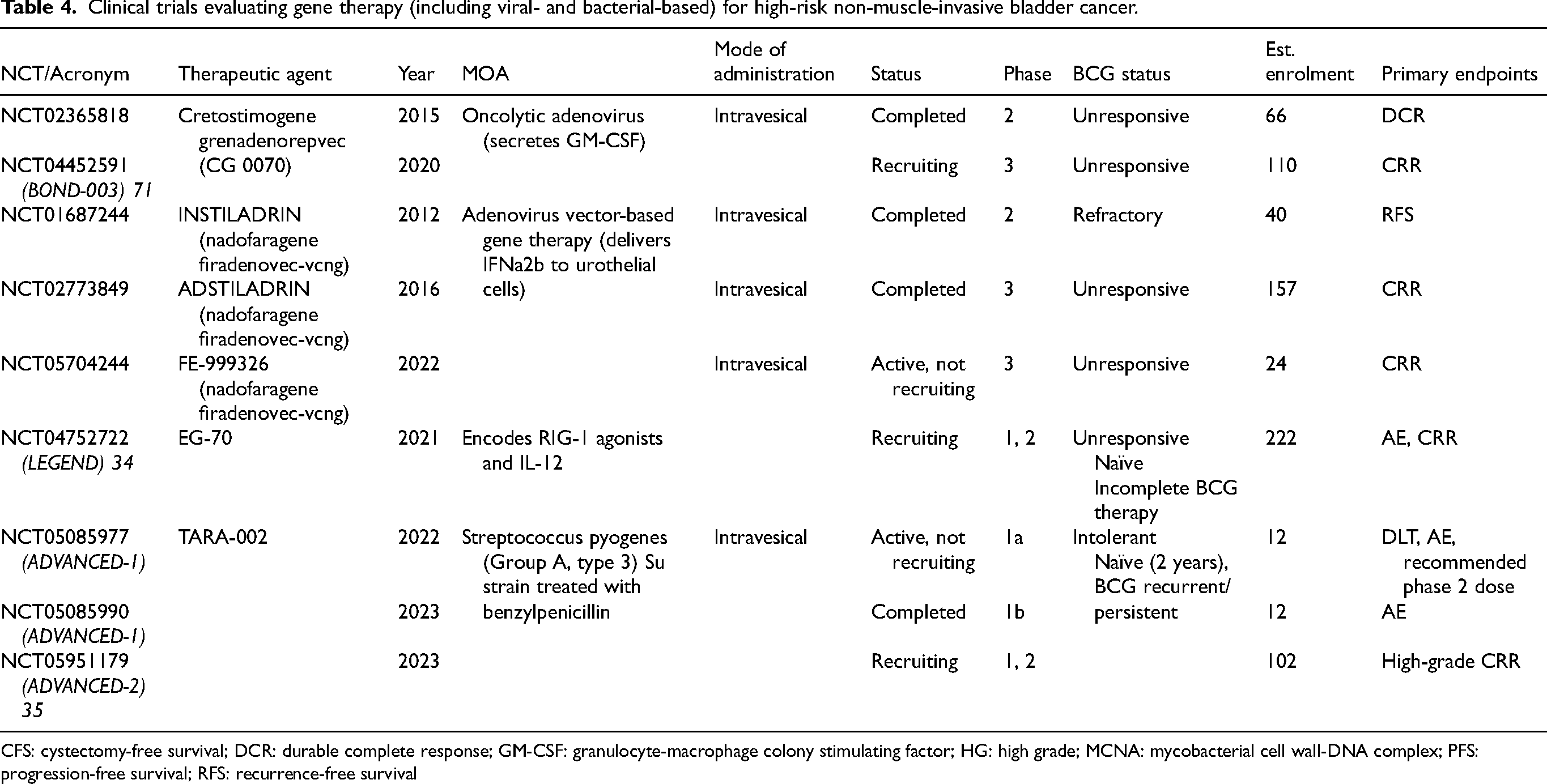
CFS: cystectomy-free survival; DCR: durable complete response; GM-CSF: granulocyte-macrophage colony stimulating factor; HG: high grade; MCNA: mycobacterial cell wall-DNA complex; PFS: progression-free survival; RFS: recurrence-free survival
Chemotherapy
Intravesical chemotherapy has emerged as the de facto alternative to BCG pending further research and validation of novel agents. Options include mitomycin-C (MMC) or gemcitabine (GEM), with a previous randomised phase III trial concluding superior recurrence-free survival with gemcitabine (72%) versus MMC (61%) in recurrent bladder cancers across contemporary risk groups. 72 Intravesical gemcitabine was also compared head-to-head with BCG in a phase II trial of patients with high-risk NMIBC and failure following 1 course of BCG, showing no significant difference in the mean time to first recurrence (P = 0.09) or progression requiring radical cystectomy (P = 0.12). However, the GEM cohort showed superior 2 year RFS of 19%, as compared to the BCG cohort at 3%, resulting in intravesical gemcitabine emerging as a main contender for standard single-agent intravesical chemotherapy in high-risk NMIBC.14,73
Currently, combination therapy favours sequential intravesical GEM followed by docetaxel due to positive results in patients with previously defined BCG failure, with 1- and 2-year high grade RFS of 65% and 52% respectively. 74 More recently, McElree et al. compared combination gemcitabine and docetaxel versus BCG in patients with high-risk NMIBC, showing superior high grade RFS (hazard ratio, 0.57; 95% CI, 0.33–0.97; P =0 .04) and overall RFS (hazard ratio, 0.56; 95% CI, 0.34–0.92; P = 0.02) than treatment with BCG. 75 Although clinical outcomes from combination GEM and docetaxel appear promising, it must be noted that there is a paucity of level I evidence, and that the prior studies discussed are of a retrospective nature. At this time, there are 2 trials seeking to address this deficit in providing high quality evidence to support alternative primary adjuvant therapy. The GEMDOCE trial (NCT04386746) recently reported the results of their phase 2 single-arm study of 25 patients with BCG naïve high risk NMIBC. This study achieved 100% CR at 3 months, and 92% RFS at 12 months. 2 patients experienced T1HG recurrences during follow-up, but none progressed to muscle-invasive disease or required radical surgery. 37 In addition, the ongoing phase IIIb BRIDGE trial (NCT05538663) seeks to compare BCG versus GEM/docetaxel in BCG naïve NMIBC patients, with a primary outcome of event-free survival. Secondary outcomes include safety, toxicity, progression-free survival, cystectomy-free survival and cancer- and bladder-cancer-specific changes in quality of life. 76
Other prospective data on combination therapy in the arena of high-risk NMIBC includes gemcitabine and BCG in a phase I/II trial (NCT04179162) of BCG-exposed patients, with a 6 month CR of 95%. Further combinations under investigation include cabazitaxel, gemcitabine and cisplatin in a phase II trial (NCT02202772) of BCG unresponsive NMIBC, showing a 1 year recurrence-free survival of 73%..38,39 A summary of chemotherapeutic agents under investigation in high risk NMIBC is summarised in Table 5.
Clinical trials evaluating intravesical chemotherapy and other novel agents for high-risk non-muscle-invasive bladder cancer.

Methods to enhance administration of intravesical chemotherapy
Hyperthermic intravesical chemotherapy
Recent years have also seen hyperthermic intravesical chemotherapy (HIVEC) gain traction as a candidate for bladder sparing therapy. Mitomycin-C is often the agent of choice, with its ability to destroy circulating and residual tumour cells. Hyperthermia is thought to increase urothelial permeability and allow for better drug absorption, reduce angiogenesis, cause release of heat shock proteins and induce cell death via denaturation of proteins and cytoplasmic structures.77,78 A meta-analysis by Zhao et al on HIVEC versus BCG in intermediate- and high-risk NMIBC reviewed 5 studies, including 4 randomised trials, ultimately showing no significant differences in 24 to 36 month recurrence rates, 24 to 36 month progression rates, or adverse events between treatment arms. 79 The 2019 phase III, multi-centre, randomised HYMN trial investigated the role of a radiofrequency-induced thermo-chemotherapy effect (RITE) versus a second course of BCG in recurrent BCG-exposed NMIBC. Results showed no significant difference in DFS between treatment arms, or in 3 month complete response rates. Notably, disease-free survival was significantly lower in the RITE arm than in control in the CIS subgroup, suggesting that recurrent non-CIS NMIBC may instead be a more appropriate target group for this treatment. 80
The role of heated intravesical chemotherapy in intermediate risk NMIBC was further queried in the HIVEC-I and HIVEC-II trials, which utilised the COMBined Antineoplastic Thermotherapy (COMBAT) BRS system to heat intravesical fluid to 43 degrees Celsius (°C) at a constant flow-rate via a 16F 3 way Foley catheter. Patients in HIVEC-I were divided into 3 cohorts: control, 43°C at 30 min, and 43°C at 60 min. 24-month recurrence-free survival per protocol was 77%, 83% and 80% respectively. While 30-min hyperthermia at 43°C appeared superior to control, initial results were not statistically significant (P = 0.59). 81 The HIVEC-2 trial soon followed, showing comparable DFS at 24 months in treatment (61%) versus control (60%). 82 While these trials concluded that chemohyperthemia could not be recommended as a superior to standard administration for intermediate risk NMIBC, they laid the groundwork for further interrogation of HIVEC in high-risk NMIBC. Kastner et al. studied HIVEC in BCG naïve and unresponsive high-risk NMIBC, demonstrating overall RFS at 1- and 2-years as 67% and 40% respectively. In contrast to other studies on hyperthermia in intermediate risk NMIBC, no statistical differences were noted in RFS across BCG exposure or CIS status. 77 Although initial results for heated intravesical chemotherapy in HR-NMIBC appear promising, they must be interpreted with caution having been concluded from a retrospective cohort study alone.
EMDA
Electromotive Drug administration of Mitomycin-C (EMDA-MMC) is a device-assisted therapy that utilises electrical current to direct and accelerate movement of the drug towards the target tissues. EMDA-MMC presents an alternative method of enhancing the efficacy of intravesical chemotherapy, this time via a combination of iontophoresis, electrophoresis and electroporation, resulting in higher drug penetration. A phase II study of 26 patients with BCG refractory NMIBC reported DFS of TaG3, T1G3, CIS and TaT1GS with CIS of 75, 71.4, 50 and 25%, respectively at 3 year follow up, reflecting poor response in the high-grade and CIS subgroups. 38.4% of patients had also undergone radical cystectomy by the end of 3-year follow up. 83 Sequential BCG and EMDA-MMC was also explored in BCG unresponsive high risk NMIBC, showing 1- and 2-year PFS of 58.3% and 48.9% respectively and a 1-year CR of 44%. Despite these promising findings, the population assessed within the confines of this retrospective study showed 15% cancer-specific mortality at 2 years, suggesting that very close follow-up was mandatory for high-risk patients undergoing this treatment. 84
TAR-200
As previously discussed, TAR-200 is a novel drug delivery system encouraging sustained local release of gemcitabine into the bladder whilst limiting systemic toxicity. TAR-200 has previously been investigated alongside the immunoagent cetrelimab, with TAR-200 monotherapy demonstrating an 84% CR at 5 months, far outperforming cetrelimab alone (46%) or as part of combination therapy (68%). 24 Following on from these promising results, the ongoing phase 3 SunRISe-5 trial aims to study TAR-200 monotherapy versus GEM or MMC in BCG-unresponsive high-risk NMIBC. This phase 3 study aims to enrol 250 patients and is estimated to complete in 2031. 85
NDV01
NDV01 is a novel controlled release intravesical formation for BCG refractory NMIBC. This involves the gradual release of gemcitabine (1000 mg) and docetaxel (40 mg) over a 10-day period at a steady state, allowing for localised continuous exposure of bladder cancer cells while minimising the side effects of systemic exposure. This phase II trial (NCT06663137) aims to enrol 70 patients, with the primary endpoints of CR and safety, and secondary endpoints of duration of response, CR in the CIS subset, and event-free survival at 12 months. 86
Trimodality treatment
Although the majority of research efforts in chemotherapy for HR-NMIBC are centred on intravesical chemotherapy, a recent trial has also explored the extended role of trimodality treatment outside of muscle-invasive bladder cancer. This phase II trial of 34 patients explored the role of trimodality treatment radiation therapy (61.2Gy in 34 fractions) and radiosensitizing chemotherapy (cisplatin, mitomycin or 5-fluorouracil) in patients with BCG-unresponsive T1 tumours who would otherwise have been candidates for radical cystectomy (NCT00981656). 87 OS at 3 years was 69% with a corresponding freedom from cystectomy rate of 88% for the same time interval; however 52.9% of patients experienced TRAEs ≥ G3 at a median follow-up of 5.1 years.
Photodynamic therapy
TLD-1433 is a novel, water-soluble photosensitizer selective for bladder cancer cells which is administered intravesically. Following activation by diffused green laser light, it generates oxygen radicals leading to immune cascades and eventual cell death. 88 Currently, TLD-1433 photodynamic therapy is being investigated in a phase II study of BCG-unresponsive NMIBC (NCT03945162). Endpoints assessed are CR, duration of CR as well as safety with an estimated enrolment of 125 patients and planned study completion in 2025. Interim analysis of 44 patients showed an any-time CR of 55.3% with DOR of 21% CR at 450 days. 6 TRAEs ≥3 were reported but were deemed to lack direct collection to photodynamic therapy. 89
Conclusion
In patients with recurrent or BCG unresponsive high-risk NMIBC, therapeutic options are limited and radical cystectomy still remains the standard of care for oncological control. However, there is an apparent paradigm shift away from the morbidity and mortality associated with cystectomy, as influenced by some patients’ preference for a bladder sparing approach, fitness for surgery, and emerging bladder preserving strategies in the face of the era of BCG shortage.
While BSTs are currently only advised within the setting of clinical trials, FDA approval of the use of pembrolizumab, nadofaragene firadenovec and NAI signify the changing treatment landscape of high-risk NMIBC. The majority of current data, although promising, is primarily a result of phase II or single-arm trials, and requires further validation in multi-arm, multi-centre, randomised studies. Recent years have seen an emphasis on PD1 and PDL1 immune checkpoint inhibitors dominating this field of research, leaving room for further interrogation of other immune checkpoints such as CTLA-4, LAG3 and TIGIT. Other agents such as CG0070, EG-70, TARA-002 and TAR-200 have also shown promise in achieving early complete response to treatment, but warrant further investigation.
With the expanding armamentarium of potential therapeutics into high-risk NMIBC also comes future challenges to be addressed, such as determining optimal treatment regimens and sequencing, long-term efficacy and tolerability, ease of access to treatment and financial implications outside of clinical trial settings. The burden of treatment and cost-effectiveness of these novel agents must also be evaluated thoroughly within each healthcare context in order to assure the delivery of sustained clinically effective and value-based care for our patients.
Overall, we expect there to be limited uptake in these novel and expensive bladder sparing treatments worldwide, and radical cystectomy remains the mainstay gold-standard treatment for high-risk NMIBC with the best survival outcomes for the patient. The challenge for industry players is how they can reduce their profit margins in order for mass market permeation to urologists to adopt these currently “unconventional” bladder sparing treatments to patients with high-risk NMIBC. Industry partners also have to balance and justify their business cases, given their substantial investments to the research and development pipeline. As for policy makers (e.g. Ministry of Health) or hospital administrative leaders, the challenge is how they can justify innovative state-of-the-art and novel therapies such as those discussed in this paper for high-risk NMIBC patients who desire bladder sparing treatments, while maintaining equitable disbursements of monetary subsidies (i.e. the classic ‘shopping spree’ problem in health economics). Key stakeholders need to sit at the same table to discuss a concerted strategy for each country, and a National Committee or Working Group for NMIBC treatments can be considered, if not already present.
Footnotes
ORCID iDs
Author contributions
Conceptualisation: CG, ZWC, JL.
Methodology: CG, JL.
Data curation: CG.
Writing: original draft preparation: CG, JL.
Writing: review and editing: all.
Visualisation: CG, JL.
Supervision: YLC, DY, ZWC, SY, JL.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: PLC has been involved in advisory boards with Astrazeneca, Amgen, Merck and Astella. The remaining authors CG, YLC, ZWC, DC, EV, SEKY, ZL, TJ, JLK, SHK, DZPY and JJL have no other disclosures or conflicts of interest related to this work.
