Abstract
Seven novel N-alkyl-plinabulin derivatives with aryl groups moieties (nitroquinoline, 1,4-dihydroquinoline, 4-methoxybenzene, and 4-chlorobenzene) have been synthesized via aldol condensation and alkylation in one-pot, and tested for their cytotoxicity against 4 cancer cell lines (KB, HepG2, Lu, and MCF7). Compounds (Z)−3-((6,8-dimethyl-4-oxo-1,4-dihydroquinolin-2-yl)methylene)−6-((Z)−4-methoxybenzylidene)−1-(prop-2-yn-1-yl)piperazine-2,5-dione (
In recent years, plinabulin (NPI-2358), a synthetic analog of 2,5-diketopiperazine, has been reported to be a promising vascular disrupting agent (VDA) thus serving as an anti-cancer drug. VDAs target microtubules—the essential filamentous structure of eukaryotic cells, particularly by inducing rapid depolymerization of microtubules in highly proliferating tumor selective vascular endothelial cells, thus causing tumor vascular collapse. 1 Historically, notable VDAs, such as colchicine and combretastatin, have been found to be effective anti-cancer drugs. 2 On the other hand, plinabulin has a relatively favorable safety profile while still exhibiting colchicine-like tubulin depolymerizing activity. Several plinabulin derivatives showed high cytotoxicity against different cancer cell lines, for example, benzophenone derivatives were evaluated against human HT-29 colorectal cancer cells, while 5-tert-butyl-substituted imidazole analogs were not highly active. 3,4 The benzene ring of the benzoyl group could induce additional π–π interaction, which could be beneficial to antiproliferation. 5,6 Furan-containing derivatives tested against the human lung cancer NCI-H460 cell line exhibited potent cytotoxic activity at the nanomolar level. 7 Of special interest is a novel colchicine-type anti-microtubule compound: KPU-300, an agent endowed with a 2-pyridyl substituent capable of acting as a potent radiosensitizer. 8 Currently, plinabulin is being evaluated in Phase III clinical trial in combination with docetaxel and in phase I/II clinical trial in combination with nivolumab for stage IIIb/IV non-small cell lung cancer (NCT02504489 and NCT02812667). 9 At the same time, there has also been strong interest in the synthesis of plinabulin derivatives to explore options for heightened effectiveness and new applications. This paper reports 7 novel N-alkyl-plinabulin derivatives with aryl group moieties and their cytotoxic activity against some cancer cell lines in vitro.
Results and Discussion
The development of plinabulin derivatives with aryl moieties is a research direction of interest due to the ability of these moieties to alter significantly the hydrophobicity and π-π interaction capacity of the compound. 10,11 In this study, the moieties of choice were nitroquinoline, 1,4-dihydroquinoline, 4-methoxybenzene, and 4-chlorobenzene. The quinoline ring is commonly acknowledged for its antimalarial potency, and the quinine derivative, Chloroquine, has been the most widely used anti-malarial drug since the 1940s. 12 The versatility of this ring system has enabled the synthesis of a large array of derivatives with diverse biological activities, notably the derivatives of quinoline-2-carbaldehyde. 13,14 Promising quinoline-2-carbaldehyde and 1,4-dihydroquinoline-2-carbaldehyde derivatives previously reported in another study were applied in this work. 15 4-Methoxybenzene and 4-chlorobenzene, 2 of the most prevalent substructures in anticancer molecules as scanned against the National Cancer Institute’s 60 human tumor cell lines, were also utilized. 16 The successful synthesis of these derivatives offers useful information for the development of more potent VDAs based on plinabulin. Thus, in continuation of our previous work, we have prepared new biological active plinabulin analogs. 17 The general procedures for the synthesis of the 7 novel N-alkyl-plinabulin derivatives with different aryl moieties is outlined in 1 -4.

Reagent and conditions: (i) 1.5 equiv. R1CHO (2) 1.5 equiv. K2CO3, 1.5 equiv. propagyl bromide, DMF, r.t, 24 hours; (ii) 1.5 equiv. R1CHO (2) 1.5 equiv. K2CO3, 1.5 equiv. MeI, DMF, r.t, 24 hours.

Reagent and conditions: (i) 1.5 equiv. R2CHO (4), 1.5 equiv. K2CO3, DMF, reflux, 24 hours; (ii) 1.5 equiv. R2CHO (4) 1.5 equiv. K2CO3, 1.5 equiv. MeI, DMF, 0→80 °C, 24 hours.

Reagent and conditions: (i) 1.5 equiv. R2CHO (4) 1.5 equiv. K2CO3, DMF, reflux, 24 hours; (ii) 1.5 equiv. R2CHO (4) 1.5 equiv. K2CO3, 1.5 equiv. MeI, DMF, 0→80 °C, 24 hours.
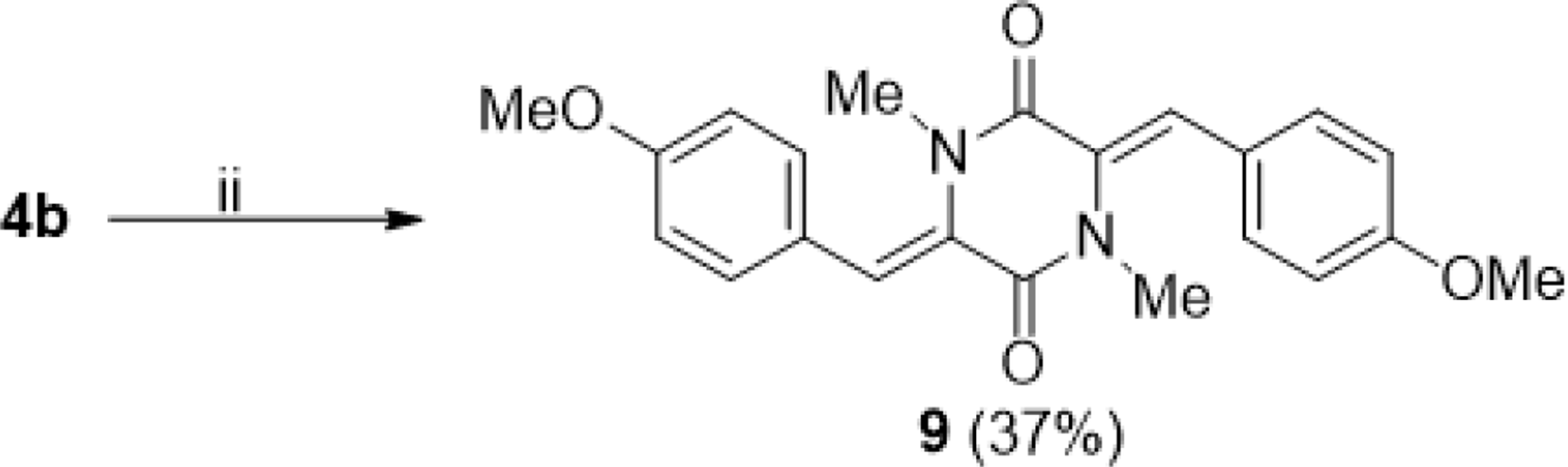
Reagent and conditions: 1.5 equiv. 4-methoxybenzaldehyde (4) 1.5 equiv. K2CO3, 1.5 equiv. MeI, DMF, 0→80 °C, 24 hours.
The N-propargyl-plinabulin derivatives
Similarly, the N-propagyl-N-methyl-plinabulin derivatives
Reaction of 1,4-diacetyl-2,5-diketopiperazine
The cytotoxicity evaluation results of the novel synthesized plinabulin derivatives against the epidermoid carcinoma cell line (KB), hepatoma carcinoma cell line (HepG2), lung cancer cell line (Lu), and breast carcinoma cell line (MCF-7) are presented in Table 1.
Cytotoxicity Evaluation of Novel N-Alkyl-Plinabulin Derivatives.

Ellipticine was used as a positive control.
As the results shown in Table 1, compounds
Experimental
General
All reactions were performed in appropriate oven-dried glass apparatus and under a nitrogen atmosphere. Unless otherwise stated, solvents and chemicals were obtained from commercial sources and used without further purification. Column chromatography was performed using silica gel (60Å, particle size 40‐60 μm). NMR spectra were recorded on a Bruker Advance I spectrophotometer (500 MHz). Chemical shifts (δ) are given in parts per million (ppm) and coupling constants (J) in Hertz (Hz). High resolution mass spectra (HRMS) were recorded on either a Q-executive or a Q-TOF2 instrument, and IR spectra on a Perkin Elmer Spectrum Two.
General Procedure for the Synthesis of Compounds 3, 4a, and 4b
A solution of 1,4-diacetyl-2,5-piperazinedione (
(Z)-1-Acetyl-3-(4-Methoxybenzylidene)-4-(Prop-2-Yn-1-Yl)piperazine-2,5-Dione (3)
91% yield, yellow solid.1H NMR (CDCl3, 500 MHz): 7.38 (1H, s); 7.33 (2H, dd, J = 2.0; 7.0 Hz); 6.94 (2H, dd, J = 2.0; 7.0 Hz); 4.53 (2H, s); 4.31 (2H, d, J = 2.5 Hz); 3.85 (3H, s); 2.62 (3H, s); 2.13 (1H, t, J = 2.5 Hz). 13C NMR (CDCl3, 125 MHz): 171.4; 164.8; 164.3; 160.9; 131.4 (2xC); 129.2; 127.7; 127.1; 124.5; 114.4 (2xC); 72.7; 55.4; 45.1; 33.6; 26.5. HR-ESI-MS: Found m/z 313.1179; calcd. for C17H17N2O4: 313.1183 [M + H]+.
(Z)-1-Acetyl-3-(4-Chlorobenzylidene)-4-Methylpiperazine-2,5-Dione (4a)
86% yield, yellow solid. 1H NMR (CDCl3, 500 MHz), δ (ppm): 7.40 (2H, d, J = 8.5 Hz), 7.38 (1H, s), 7.31 (2H, d, J = 8.5 Hz), 4.54 (2H, s), 2.96 (3H, s), 2.62 (3H, s). HR-ESI-MS: Found m/z 293.0687; Calcd. for C14H14ClN2O3: 293.0687 [M + H]+.
(Z)-1-Acetyl-3-(4-Methoxybenzylidene)-4-Methylpiperazine-2,5-Dione (4b)
88% yield, yellow solid.1H NMR (CDCl3, 500 MHz), δ (ppm): 7.30 (1H, s), 7.28 (2H, dd, J = 2.0, 7.0 Hz), 6.92 (2H, dd, J = 2.0, 7.0 Hz), 4.51 (2H, s), 3.85 (3H, s), 2.96 (3H, s), 2.62 (3H, s). 13C NMR (CDCl3, 125 MHz): 171.5, 165.1, 164.1, 160.6, 131.5 (2xC), 129.8, 125.8, 124.8, 114.1 (2xC), 55.4, 45.3, 34.2, 26.6. HR-ESI-MS: Found m/z 289.1191; Calcd. for C15H17N2O4: 289.1183 [M + H]+.
General Procedure for the Synthesis of Compounds 5a-C and 7
A solution of compound
(Z)-3-((6,8-Dimethyl-4-Oxo-1,4-Dihydroquinolin-2-Yl)methylene)-6-((Z)-4-Methoxybenzylidene)-1-(Prop-2-Yn-1-Yl)piperazine-2,5-Dione (5a)
38% yield, yellow solid.1HNMR (CDCl3, 500 MHz), δ (ppm): 8.00 (2H, d, J = 8.5 Hz), 7.70 (1H, s), 7.37 (1H, s), 7.30 (2H, d, J = 8.5 Hz), 7.29 (1H, s), 6.63 (1H, s), 6.38 (1H, s), 4.96 (2H, d, J = 2.5 Hz), 3.97 (3H, s), 2.77 (3H, s), 2.52 (1H, t, J = 2.5 Hz), 2.42 (3H, s). 13C NMR (CDCl3, 125 MHz), δ (ppm): 162.2, 159.7, 154.4, 152.8, 145.6, 135.8, 135.6, 134.9, 133.5, 133.1, 133.0 (2xC), 131.9, 129.1, 128.5 (2xC), 128.2, 119.7, 118.4, 105.8, 101.9, 77.9, 75.2, 55.7, 54.5, 21.7, 19.0. HR-ESI-MS: Found m/z 454.1753; Calcd. for C27H24N3O4: 454.1761 [M + H]+.
(Z)-6-((Z)-4-Methoxybenzylidene)-1-(Prop-2-Yn-1-Yl)-3-((1,6,8-Trimethyl-4-Oxo-1,4-Dihydroquinolin-2-Yl)methylene)piperazine-2,5-Dione (5b)
43% yield, yellow solid.1H NMR (CDCl3, 500 MHz), δ (ppm): 7.79 (1H, s), 7.44 (1H, s), 7.42 (1H, s), 7.29 (2H, d, J = 8.5 Hz), 6.91 (2H, d, J = 8.5 Hz), 6.87 (1H, s), 6.76 (1H, s), 4.48 (2H, d, J = 2.5 Hz), 4.05 (3H, s), 3.84 (3H, s), 2.83 (3H, s), 2.49 (3H, s), 2.14 (1H, t, J = 2.5 Hz). 13C NMR (CDCl3, 125 MHz), δ (ppm): 162.4, 160.0, 159.1, 145.1, 135.9, 135.7, 133.1, 131.8, 130.9 (2xC), 127.1, 126.0, 122.3, 119.9, 118.4, 114.0 (2xC), 113.2, 102.2, 101.1, 77.7, 72.4, 55.7, 55.3, 35.6, 21.7, 19.1. HR-ESI-MS: Found m/z 468.1947. Calcd. for C28H26N3O4: 468.1918 [M + H]+.
3-((Z)-4-Chlorobenzylidene)-6-((Z)-4-Methoxybenzylidene)-1-(Prop-2-Yn-1-Yl)piperazine-2,5-Dione (5c)
35% yield, yellow solid. IR (KBr) νmax (cm-1): 3336, 3072, 2926, 2851, 2132, 1734, 1683, 1700, 1613, 1583, 1558, 1336, 1179, 1088, 845. 1H NMR (DMSO-d6 , 500 MHz), δ (ppm): 7.43 (2H, d, J = 7.5 Hz), 7.36 (2H, d, J = 7.5 Hz), 7.27 (1H, s), 7.24 (2H, d, J = 8.5 Hz), 7.00 (2H, d, J = 8.5 Hz), 6.98 (1H, s), 4.72 (2H, d, J = 2.5 Hz), 3.84 (3H, s), 2.53 (1H, t, J = 2.5 Hz).13C NMR (DMSO-d6 , 125 MHz), δ (ppm): 159.6, 159.4, 157.8, 134.7, 131.4, 131.0 (2xC), 129.8 (2xC), 129.7 (2xC), 129.2, 126.8, 126.4, 121.3, 115.5, 114.8 (2xC), 78.1, 75.9, 55.8, 46.7. HR-ESI-MS: Found m/z 393.1021. Calc. for C22H18ClN2O3: 393.1000 [M + H]+.
3,6-Bis((Z)-4-Chlorobenzylidene)-1-Methylpiperazine-2,5-Dione (7)
36% yield, yellow solid.1H NMR (CDCl3, 500 MHz), δ (ppm): 7.42‐7.36 (6H, overlapped), 7.23 (1H, s), 7.22 (2H, d, J = 8.5 Hz), 7.00 (1H, s), 2.99 (3H, s).13C NMR (CDCl3, 125 MHz), δ (ppm): 159.2 (2xC = O amide), 134.8, 134.6, 132.2, 131.1, 130.9, 129.9 (2xC), 129.8 (2xC), 128.6 (2xC), 128.0 (2xC), 126.1, 119.7, 116.2, 36.7. HR-ESI-MS: Found m/z 373.0521; Calcd. for: C19H15Cl2N2O2: 373.0505 [M + H]+.
General Procedure for the Synthesis of Compounds 6, 8, and 9
A solution of compound
3-((Z)-4-Chlorobenzylidene)-6-((Z)-4-Methoxybenzylidene)-4-Methyl-1-(Prop-2-Yn-1-Yl)piperazine-2,5-Dione (6)
33% yield, yellow solid. IR (KBr) νmax (cm-1): 3330, 3076, 2926, 2855, 2135, 1715, 1680, 1668, 1600, 1580, 1551, 1333, 1169, 1076, 845. 1H NMR (DMSO-d6 , 500 MHz), δ (ppm): 7.39 (2H, dd, J = 2.0, 8.0 Hz), 7.37 (1H, s), 7.23 (2H, d, J = 8.0 Hz), 7.00 (2H, dd, J = 2.0, 8.0 Hz), 6.97 (2H, d, J = 8.0 Hz), 6.96 (1H, s), 4.75 (2H, d, J = 2.5 Hz), 3.84 (3H, s), 3.03 (3H, s), 2.55 (1H, t, J = 2.5 Hz).13C NMR (DMSO-d6 , 125 MHz), δ (ppm): 159.9, 159.7, 157.7, 131.0 (2xC), 130.5, 129.5 (2xC), 127.0, 124.6, 120.6, 119.9, 117.2, 116.0, 114.9 (2xC), 114.7 (2xC), 78.1, 75.9, 55.8, 46.6, 36.6. HR-ESI-MS: Found m/z 407.1173. Calcd. for C23H20ClN2O3: 407.
(Z)-3-((Z)-4-Chlorobenzylidene)-1,4-Dimethyl-6-((8-Methyl-4-Nitroquinolin-2-Yl)methylene)piperazine-2,5-Dione (8)
29% yield, yellow solid. IR (KBr) νmax (cm-1): 3047, 2924, 2855, 1735, 1680, 1616, 1583, 1487, 1334, 1165, 1089, 819, 756. 1H NMR (DMSO-d6 , 500 MHz), δ (ppm): 8.02 (1H, s), 7.45 (1H, d, J = 8.0 Hz), 7.63 (1H, dd, J = 1.0, 8.0 Hz), 7.38‐7.50 (5H, m, overlap), 7.21 (1H, s), 7.11 (1H, s), 3.02 (3H, s), 2.88 (3H, s), 2.67 (3H, s). 13C NMR (DMSO-d6 , 125 MHz), δ (ppm): 160.8, 160.6, 151.8, 146.7, 141.3, 137.1, 134.7, 132.2, 132.2, 131.6 (2xC), 131.2, 128.3 (2xC), 128.0, 124.4, 123.7, 121.4, 119.1, 118.6, 117.2, 36.0, 35.3, 17.7. HR-ESI-MS: Found m/z 463.1175. Calcd. for C24H20ClN4O4: 463.1168 [M + H]+.
3-((Z)-4-Chlorobenzylidene)-6-((Z)-4-Methoxybenzylidene)-1,4-Dimethylpiperazine-2,5-Dione (9)
37% yield, yellow solid. 1H NMR (CDCl3, 500 MHz), δ (ppm): 7.28 (4H, d, J = 7.0 Hz), 7.15 (2H, s), 6.90 (4H, d, J = 7.0 Hz), 3.84 (6H, s), 3.00 (6H, s).13C NMR (CDCl3, 125 MHz), δ (ppm): 162.9 (2xCO amide), 159.9 (2xC), 131.1 (4xC), 130.1 (2xC), 125 (2xC), 121.7 (2xC), 113.9 (4xC), 55.3 (2xC), 35.1 (2xC). HR-ESI-MS: Found m/z 378.1579, calcd. for C22H22N2O4: 378.1574 [M + H]+.
Cell culture and cell viability assay
The synthesized compounds were evaluated for their cytotoxicity against the 4 hoursuman cancer cell lines, epidermoid carcinoma (KB), hepatoma carcinoma (HepG2), lung cancer cell line (Lu) and breast carcinoma (MCF7) obtained from the American Type Culture Collection (USA). The cells were grown in RPMI 1640 medium supplemented with 10% fetal bovine serum, 100 U/mL penicillin, and 100 µg/mL streptomycin at 37 °C in a humidified atmosphere (95% air and 5% CO2). Exponentially growing cells were used throughout the experiments. The inhibitory effects of the compounds on the growth of the cancer cell lines were determined by measuring their metabolic activity using a 3-[4,5-dimethylthiazol-2-yl]−2,5-diphenyl-trazolium bromide (MTT) assay. Briefly, the cell lines (1 × 105 cells/mL) were treated for 3 days with a series of concentrations of the compounds (in DMSO): 0.125, 0.5, 2.0, 8.0, 32.0, and 128.0 µg/mL. After incubation, 0.1 mg MTT solution (50 µL of a 2 mg/mL solution) was added to each well, and the cells were then incubated at 37 °C for 4 hours. The plates were centrifuged at 1000 rpm for 10 minutes at room temperature, and the medium was then carefully aspirated. Dimethyl sulfoxide (150 µL) was added to each well to dissolve the formazan crystals. The plates were read immediately at 540 nm on a microplate reader (TECAN GENIOUS). All the experiments were performed 3 times, and the mean absorbance values were calculated. The results are expressed as the percentage of inhibition that produced a reduction in the absorbance by the treatment of the compounds compared to the untreated controls. A dose-response curve was generated, and the inhibitory concentration of 50% (IC50) was determined for each compound, as well as for each cell line.
Conclusions
In conclusion, 7 novel N-alkyl-plinabulin derivatives with aryl group moieties (nitroquinoline, 1,4-dihydroquinoline, 4-methoxybenzene, and 4-chlorobenzene) were successfully synthesized via aldol condensation alkylation in one-pot. Preliminary cytotoxicity evaluation of these compounds against the KB, HepG2, Lu, and MCF7 cell lines demonstrated diverse levels of effectiveness and selectivity, thus providing useful information for the development of more potent VDAs based on plinabulin. Compounds
Supplemental Material
Online supplementary file 1 - Supplemental material for Synthesis and Cytotoxic Activity of Several Novel N-Alkyl-Plinabulin Derivatives With Aryl Group Moieties
Supplemental material, Online supplementary file 1, for Synthesis and Cytotoxic Activity of Several Novel N-Alkyl-Plinabulin Derivatives With Aryl Group Moieties by Pham The Chinh, Pham Thi Tham, Duong Huong Quynh, Nguyen Van Tuyen, Dinh Thuy Van, Phan Thanh Phuong, Tran Thi Thu Hang and Phan Van Kiem in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors are indebted to the Vietnamese National Foundation for Science and Technology Development (NAFOSTED), code 104.01-2016.18 for financial support.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
