Abstract
BACKGROUND:
Endoscopy-assisted breast conserving surgery (E-BCS) was developed over 10 years ago as a method for breast cancer treatment with the potential advantage of less noticeable scarring. However, the evidence supporting its superiority over conventional breast conserving surgery (C-BCS) remains unclear.
OBJECTIVE:
This study aims to compare the outcomes of E-BCS and C-BCS for the treatment of early breast cancer.
METHODS:
A comprehensive search for relevant articles was performed using specific keywords in Medline, Scopus, ClinicalTrials.gov, and Cochrane Library PubMed up to October 17th, 2022. Clinical trials that compared E-BCS with C-BCS in early breast cancer patients were collected.
RESULTS:
Our analysis of nine studies revealed that E-BCS was associated with shorter incision length [Mean Difference (MD) −6.50 cm (95% CI −10.75, −2.26), p = 0.003, I2 = 99%] and higher cosmetic score [MD 2.69 (95% CI 1.46, 3.93), p < 0.0001, I2 = 93%] compared with C-BCS. However, E-BCS had a longer operation time [MD 35.95 min (95% CI 19.12, 52.78), p < 0.0001, I2 = 93%] and greater drainage volume [MD 62.91 mL (95% CI 2.55, 123.27), p = 0.04, I2 = 79%]. There was no significant difference in blood loss volume (p = 0.06), drainage duration (p = 0.28), postoperative complications (p = 0.69), or local recurrence (p = 0.59) between the two groups.
CONCLUSION:
Our study suggests that E-BCS offers a shorter incision length and better cosmetic outcome compared with C-BCS in the treatment of early breast cancer. However, E-BCS requires a longer operation time and has greater drainage volume. Further studies are needed to confirm these findings.
Introduction
Breast cancer is one of the most prevalent cancers worldwide, with a rising incidence over the past few decades. It affects both genders, but is more frequent in women [1]. According to GLOBOCAN 2020 data, there were 2,261,419 new cases of female breast cancer, accounting for 11.7% of all new cancer cases, and 684,996 deaths due to breast cancer, accounting for 6.9% of cancer-related deaths. Breast cancer is ranked as the second leading cause of death in women after lung cancer [2]. In 2020, breast cancer became the most prevalent cancer in women, with 7.8 million women alive diagnosed within the last 5 years [3]. The incidence of breast cancer increases with age, reaching a peak of 40–50 years in Asian countries and 60–70 years in Western countries [4]. Despite the decline in breast cancer mortality rate by 43% from 1989 to 2020 due to earlier detection through screening, increased awareness, and better management strategies, the decline is currently slowing [5].
To reduce the burden of breast cancer and improve the quality of life for patients, various innovations have been made, including better and less-invasive surgical techniques. Breast-conserving surgery (BCS) combined with postoperative radiotherapy has become the standard therapy for early-stage breast cancer due to its good long-term outcomes and safety, which are not inferior to total mastectomy [6]. The development of oncoplastic surgery has improved aesthetic outcomes after BCS. Endoscopy-assisted BCS (E-BCS) techniques focuses on necessary yet tiny incisions made in discrete or hidden places and it has been developed to achieve superior cosmetic results without sacrificing safety or preventing immediate breast reconstruction from being done through the same incision. However, E-BCS has some drawbacks, including a restricted operative field, lengthier operating time, and higher expenses associated with the use of disposable equipment [7–9].
Endoscopy assisted-BCS was developed more than 10 years ago [7]. In 2001 and 2002, the first case series of E-BCS and endoscopic-assisted nipple-sparing mastectomy (E-NSM) were described, both showing viability and acceptable aesthetic results. There were a total of 28 investigations on endoscopic-assisted breast surgery (EABS) throughout the following two decades, with 15 research on E-BCS and an additional 14 studies on E-NSM. Cohort studies comparing conventional and E-BCS or E-NSM showed comparable surgical and short- or medium-term oncologic results [9]. Several studies have demonstrated that E-BCS for breast cancer has a good survival rate and low recurrence outcomes with satisfactory cosmetic results [8,10,11]. However, none of these studies included a comparison group, so clear evidence about whether E-BCS may offer long-term results as good as a conventional technique but with better cosmetic outcomes cannot be obtained through the current literature.
This study aims to analyse the efficacy and safety of using E-BCS for breast cancer compared with conventional BCS (C-BCS). It seeks to provide evidence regarding whether E-BCS offers long-term outcomes as good as conventional techniques with better cosmetic outcomes. The study hopes to help reduce the burden of breast cancer and improve the quality of life for patients through less-invasive surgical techniques.
Materials and methods
Eligibility criteria
A systematic review and meta-analysis study was conducted according to Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) and Meta-analysis of Observational Studies in Epidemiology (MOOSE) guidelines [12,13]. Records were included in this study if fulfilling the inclusion criteria as defined by using the PICOS formula: P – Population: adult patients (age >18 years old) with breast cancer diagnosis in any stages who were candidates/eligible for surgical procedures; I – Intervention: underwent endoscopic breast conserving surgery (E-BCS); C – Control: underwent conventional breast conserving surgery (C-BCS); O – Outcome: reporting at least one of these outcomes: operation time, intraoperative blood loss volume, drainage duration, drainage volume, cosmetic score, incision length, postoperative complications, and local recurrence rate; S – Study design: clinical trials (either randomized or non-randomized) and observational studies (cohort, case-control, cross-sectional design); Presentation as a full-text article (which included preprints).
Articles were excluded if contain one or more exclusion criteria as follows: (1) articles with no control/comparison group; (2) non-English language articles; (3) studies on pregnant women or pediatric populations; (4) case-reports, letters to editors, correspondence; (5) unpublished study or abstract; and (6) non-primary research.
Search strategy and study selection
All studies published in English language until October 17th, 2022 were searched on the following databases: Medline (via PubMed), Scopus, ClinicalTrials.gov, and Cochrane Library. The keywords for literature searching included “(endoscopic OR endoscopy OR laparoscopy OR laparoscopic OR minimally-invasive) AND (conventional OR open) AND (breast-conserving surgery OR BCS OR partial mastectomy) AND (breast cancer OR breast neoplasm OR breast carcinoma OR mammary cancer OR mammary neoplasm OR mammary carcinoma)”. In the first step, all identified records were screened on titles/abstracts by the two authors to identify studies eligible for inclusion. Additional evaluation of references from found eligible studies was also conducted to search for more potential articles. Duplicate articles were removed. Then, the process was followed by full-text screening of the selected records based on the inclusion and exclusion criteria. If any discrepancies were found, they were resolved through discussion. Screening software Covidence was used to facilitate screening and remove duplicates.
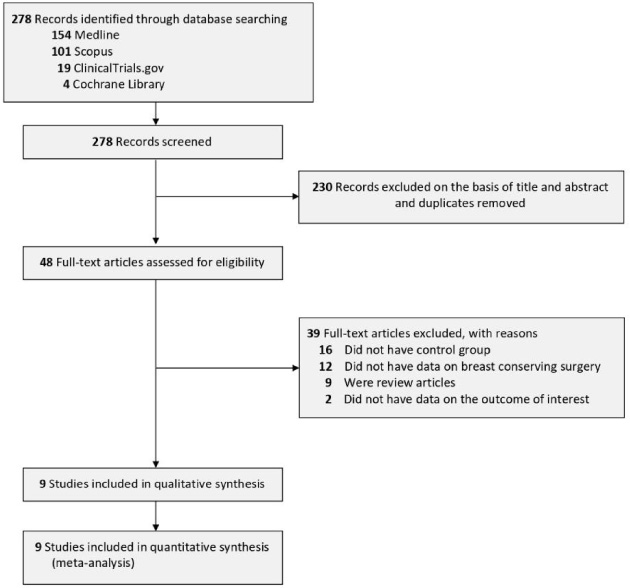
PRISMA diagram of the detailed process of selection of studies for inclusion in the systematic review and meta-analysis.
The process of data extraction was performed independently by two authors. The following items were extracted from each article: first author, year of study, sample size, study design, tumor location, tumor size, cancer stage, cancer type, number of participants in each study group, age, chemotherapy, as well as the outcome of interest.
Two researchers independently assessed the quality of each observational study involved in this study by using the Newcastle–Ottawa Scale (NOS). The assessment process included reviewing the comparability, selection, and outcome of each study, then each research was assigned a total score beginning with zero until nine. Research is graded good if it scores ≥7 [14].
Statistical analysis
The continuous variable outcomes were pooled using the Inverse-Variance formula with random-effect models, regardless of heterogeneity to obtain the mean difference (MD) with its standard deviations (SD). Meanwhile, the dichotomous variable outcomes were pooled by using the Mantel-Haenszel formula with random-effect models to obtain the odds ratio (OR) along with its 95% confidence interval (95% CI). Cochran Q test was used to identify the heterogeneity of the results and it was quantified using the I-squared statistics (I2; Inconsistency). The I2 statistic of >50% was considered statistically significant between-study heterogeneity; it means that at least half of the total variability among effect sizes is due to true heterogeneity between studies [15]. Data presented as medians and interquartile ranges or as medians and minimum-to-maximum ranges were converted to means and standard deviations for meta-analysis pooling by using the formula by Wan X et al. [16]. A funnel plot was adopted to qualitatively assess publication bias [17]. All analyses in this study were conducted using the Review Manager 5.4 software from Cochrane Collaboration.
Results
Study selection and characteristics
The study selection process is illustrated in Fig. 1. In total, 278 records were identified from four databases with the search strategy. After removing the duplicates and screening for titles and abstracts, 48 studies were eligible for the full-text assessment. Of these 48 full-text articles, 39 articles were further excluded because they did not match our inclusion and exclusion criteria. Sixteen articles did not include any control or comparison groups, twelve articles did not have the specified data on the breast conserving surgery (BCS), nine articles were review articles, and two articles did not have data on the outcome of interest, therefore resulting in the final number of 9 studies [17–25] which include a total of 2,725 patients with breast cancer for the analysis. Out of 9 studies, seven have retrospective cohort design, and the remaining two articles were prospective cohort study. Sample sizes ranged from 70 to 824. The distributions of tumor location varied among included studies, but the majority of tumors were located in the upper outer and upper inner quadrants of the breast. Mean tumor sizes were also varied, starting from 1.5 to 2.5 cm. Most of the included studies only recruited patients with grade 0 – grade II breast cancer with the domination of estrogen receptor-positive (ER+) and progesterone receptor-positive (PR+) types. More details about the characteristics of each included study can be seen in Table 1.
Quality of study assessment
Newcastle Ottawa Scale (NOS) was used to evaluate the quality of observational studies. All of the included cohort studies have good quality according to NOS with the total score ranging from 7 to 8, therefore were deemed fit to be included in the analysis. The summary studies’ quality assessment is presented in Table 2.
Endoscopy-assisted breast conserving surgery (E-BCS) vs conventional breast conserving surgery (C-BCS)
Operation time
Nine studies (n = 1,979) reported on the operation time outcome. Our meta-analysis showed that E-BCS was associated with significantly longer operation time when compared with C-BCS [MD 35.95 min (95% CI 19.12, 52.78), p < 0.0001, I2 = 93%, random-effect models] (Fig. 2A).
Baseline characteristics of the included studies
Baseline characteristics of the included studies
Newcastle-Ottawa quality assessment of observational studies
a (1) representativeness of the exposed cohort; (2) selection of the non-exposed cohort; (3) ascertainment of exposure; (4) demonstration that outcome of interest was not present at start of study. b (1) comparability of cohorts on the basis of design or analysis, (maximum two stars). c (1) assessment of outcome; (2) was follow-up long enough for outcomes to occur; (3) adequacy of follow up of cohorts.
Seven studies (n = 890) reported on the blood loss volume-outcome. Our meta-analysis showed that the blood loss volume did not differ significantly between E-BCS and C-BCS procedures [MD 9.22 mL (95% CI −0.56, 19.00), p = 0.06, I2 = 83%, random-effect models] (Fig. 2B).
Drainage duration
Three studies (n = 364) reported on the drainage duration outcome. Our meta-analysis showed that the drainage duration did not differ significantly between E-BCS and C-BCS procedures [MD 0.32 days (95% CI −0.26, 0.91), p = 0.28, I2 = 59%, random-effect models] (Fig. 2C).
Drainage volume
Three studies (n = 364) reported on the drainage volume-outcome. Our meta-analysis showed that the drainage volume was significantly higher in E-BCS than in C-BCS procedures [MD 62.91 mL (95% CI 2.55, 123.27), p = 0.04, I2 = 79%, random-effect models] (Fig. 2D).
Incision length
Three studies (n = 452) reported on the incision length outcome. Our meta-analysis showed that the incision length was significantly shorter in E-BCS than in C-BCS procedures [MD -6.50 cm (95% CI −10.75, −2.26), p = 0.003, I2 = 99%, random-effect models] (Fig. 2E).
Cosmetic score
Three studies (n = 283) reported on the cosmetic score outcome. Our meta-analysis showed that the E-BCS procedure was associated with a significantly higher cosmetic score than the C-BCS procedure [MD 2.69 (95% CI 1.46, 3.93), p < 0.0001, I2 = 93%, random-effect models] (Fig. 2F).
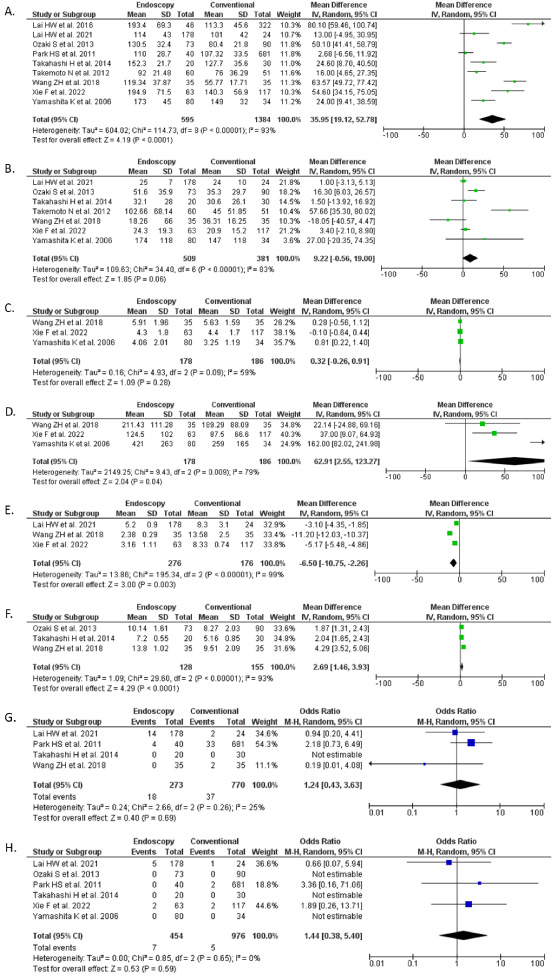
Forest plot that demonstrates the comparison between endoscopy-assisted breast conserving surgery (E-BCS) with conventional breast conserving surgery (C-BCS) for the management of early breast cancer in terms of operation time (A), blood loss volume (B), drainage duration (C), drainage volume (D), incision length (E), cosmetic score (F), post-operative complications (G), and local recurrence rate (H).
Four studies (n = 1,043) reported on the postoperative complications outcome. Our meta-analysis showed that the postoperative complications did not differ significantly between the E-BCS procedure and the C-BCS procedure [OR 1.24 (95% CI 0.43 – 3.63), p = 0.69, I2 = 25%, random-effect models] (Fig. 2G).
Local recurrence
Six studies (n = 1,430) reported on the local recurrence outcome. Our meta-analysis showed that the local recurrence rate did not differ significantly between the E-BCS procedure and the C-BCS procedure [OR 1.44 (95% CI 0.38 – 5.40), p = 0.59, I2 = 0%, random-effect models] (Fig. 2H). The follow-up period for the studies included varies from 3.3–68.6 months [18], 12–30 months [20], 9–40 months [17], 25 months [21], 43 months [24], and 26.2–31.1 months [23].
Publication bias
Publication bias detection using either funnel plots or statistical tests is not reliable if there are fewer than 10 studies when compared with whenever there are more than 10 included studies [26,27]. As in this study the number of included studies in each outcome of interest was fewer than 10 studies, then the test for publication bias was not performed.
Discussion
Our systematic review and meta-analysis aimed to summarize the latest data from nine studies that compared endoscopy-assisted breast conserving surgery (E-BCS) to conventional breast conserving surgery (C-BCS) for the management of early breast cancer.
The surgical procedure mentioned in our studies that is being compared (endoscopic VS conventional), is breast conserving surgery. Other surgery procedures that are also mentioned in the studies, including nipple-sparing mastectomy, skin-sparing mastectomy [18,19], extirpation for benign lesions, wide excision for suspected malignant lesions, modified radical mastectomy [25], and robotic breast surgery [19], are all excluded from the analysis. Several reconstruction methods used following the BCS procedures are partial breast reconstruction, omentum flap, lateral intercostal artery perforator (LICAP) flap, intercostal artery perforator flap [19], subcutaneous flap [21], and skin [17,20,22,23,25].
The axillary staging used in the studies pooled shows similar techniques. Sentinel lymph node biopsies were carried out through 2.5 cm [17,18,20,22–25] to 3 cm [21] incisions in the axillary area using radioisotope and dye methods [1–8]. If the sentinel lymph node is positive, axillary lymph node dissection (ALND) with removal of level I and II lymph nodes was performed. In one study, ALND was performed with complete resection of level I, II, and III lymph nodes [20].
Our pooled analysis showed that E-BCS was superior to C-BCS in terms of shorter incision length and higher cosmetic score. However, the E-BCS group had significantly longer operation times and drainage volumes. On the other hand, the blood loss volume, drainage duration, postoperative complications, and local recurrence rate did not differ significantly between the two groups.
The duration of operation is often influenced by the total number of cases or the type of procedure [25,28], and E-BCS has been noted to take longer time than conventional BCS [29]. Several factors that have to be taken into account for longer operation time are different reconstruction methods [18] and surgeon skills [24]. Another study shows that longer operation time in E-BCS may be caused by the procedures done by breast surgeons who have not yet been proficient in endoscopic breast surgery [20]. Nevertheless, a retrospective study of breast cancer outcomes using E-BCS reveals that 15 cases were required to drastically reduce the surgery time from a mean operation time of 208 ± 53 minutes to 121 ± 37 minutes utilizing the learning curve analysis with the CUSUM approach. With more case experience gained after overcoming the first learning curve, the operating time continued to decrease [11]. Cumulative SUM (CUSUM) graphs are used to track how far individual sample results stray from the desired value. The process aims to achieve this desired value. The figure shows a curve that is created by adding each of these variances, case by case, to a value of zero. Cases are represented by dots along the timeline-based x-axis of the curve. Every point along the curve, therefore, represents the sequential observation of cumulative performance over time [30].
One of the difficulties with E-BCS is the use of expensive disposable equipment. Hybrid E-BCS requires less disposable equipment than traditional E-BCS, hence, it is more cost-effective than the latter [20]. With a change of techniques used in E-BCS, the cost might be reduced [21]. However, in countries where the cost of medical treatment is borne by the patients or by insurance companies that make a profit, it is crucial to take into account the higher operation cost caused by the longer time and greater effort (surgeon skills, operative instruments, etc.) [24].
Seroma development is one of the most typical risks associated with breast conserving surgery. Drains are commonly placed into the incision during breast surgery to avoid this [31]. The amount of tissue that had been removed and how well the space had been filled were taken into consideration when deciding whether to insert the drain [17]. From the pooled data, blood loss volume was found to be higher in the E-BCS group, although not significantly, while drainage volume was significantly higher in the E-BCS group. These issues may be connected to the learning curve and/or limits of the existing endoscopic tools, which are impacted by training and technology advancements, respectively [32].
E-BCS has a significantly higher cosmetic score than C-BCS. The questionnaires used in these studies mainly evaluate the condition of the breast, nipple, and skin, adapted from the Japanese Breast Cancer Society classification [17,20,23]. The cosmetic evaluation was performed by experienced professional medical staff in two studies [17,20] based on the method recommended by the Japanese Breast Cancer Society (JBCS) and by patient’s satisfaction [23].
There are several complications found in E-BCS group, though not life-threatening, such as delayed healing of the areolar wound, partial ischemia of the nipple-areolar complex, complete necrosis of the nipple-areolar complex, and other minor and wound-related complications [19]. Another study reported partial areolar necrosis, partial skin flap necrosis, and postoperative bleeding, all of which were cured with conservative treatment [20]. Park HS et al. reported prolonged seroma with inflammation, cellulitis, bleeding or hematoma, and skin wound necrosis in 10% of the patients who underwent E-BCS. The same complications were also found in the C-BCS group in addition to fat necrosis and suture granuloma, although in smaller numbers compared to the E-BCS group [21]. However, the postoperative complications did not differ significantly between the C-BCS and E-BCS groups.
The rationale behind the endoscopic procedure itself is to offer a minimally invasive procedure to the patients. Unlike conventional breast surgery, which requires a large incisions in the skin to enable good visualization of structures and insertion of several instruments, the endoscopic procedures can be performed successfully with only small incisions (usually ∼2.5 cm) [32]. Based on these rationales, it is not surprising if E-BCS was associated with a shorter incision length and a better cosmetic score than C-BCS, therefore providing better visual results and greater satisfaction, especially for those who are more concerned with their appearance.
The effect of the BCT incision is essential for obtaining positive cosmetic outcomes. The location, size, and management of the surgical site all have an impact on the result. The position and depth of the tumor must be taken into consideration when the surgeon plans the incision. Results are improved by highlighting the surgeon’s role in patient selection and procedure optimization. The incision should be set up to accommodate a prospective mastectomy inclusion in the future. For the upper breast, curvilinear or transverse incisions are advised, but for the lower breast, curvilinear or radial incisions are preferred. Careful planning, oncoplastic resection, and intraoperative margin analysis are all beneficial to get a sufficient surgical margin without having to re-excise. The incision in BCT aims to provide both an aesthetic outcome that is acceptable and efficient tumor management [33].
The inclusion criteria for tumor size in these studies is less than 3 cm in two studies [19,24] and ≤5 cm in one study [23]. Thus, the subjects in these studies might have T2 to T3 classification, and with 1 cm margin, it is reasonable to have 7 to 8 cm of incision length. In Wang et al., incision length of 13.58 ± 2.50 cm is caused by the addition of a second incision in the surface of the tumor to the first incision used to perform sentinel lymph node biopsy (SNLB) and intraoperative frozen pathology [23]. Studies conducted by Wang et al. and Xie et al. used single-port endoscopic-breast conserving surgery, resulting in shorter incision length [23,24]. Another reason is that there is a possibility of widening the incision when negative margin is not achieved in conventional-breast conserving surgery.
Longer operation time in E-BCS is attributed to the added time for set-up and limited access. Other concerns regarding the safety and long-term outcomes of E-BCS have been addressed by our study, showing that there are no significant differences between E-BCS and C-BCS in terms of postoperative complications and local recurrence rate.
To our knowledge, this is the first systematic review and meta-analysis that discusses the feasibility, safety, and long-term outcomes of E-BCS by comparing it with C-BCS for the management of breast cancer. A previous non-primary study that discussed E-BCS was published in 2019, but in the form of a literature review [34]. That study by Mok CW et al. [34] reviewed data from 15 studies on E-BCS and 14 studies on endoscopy-assisted nipple sparring mastectomy (E-NSM). However, from those included studies, only 4 studies that directly compare the outcomes from E-BCS with C-BCS, while the majority of the included studies reviewed consisted of single-arm interventions. The lack of comparisons may prevent us from obtaining clear evidence regarding the benefit of E-BCS for the management of breast cancer. In contrast, our study includes a total of 9 studies that all compare the outcomes between E-BCS and C-BCS. Moreover, our study includes a meta-analysis to recalculate the data obtained from the included studies into new data with more solid evidence.
Some of the outcomes of interest in this study have relatively high heterogeneity, which may be caused by different distributions of cancer stages and tumor locations among the included studies. The follow-up periods were also varied among the studies, which may add to the heterogeneity to the analysis results. Moreover, the results from our analysis were solely based on observational studies (prospective and retrospective cohorts) which may be influenced by selection and information biases. The only available clinical trial study about E-BCS for early breast cancer (INTEND-1) did not have data on the outcome of interest specified in our study [35]. Several limitations of these studies are single institution experience, small number of study subjects, retrospective in nature, and short follow-up period. A large, well-designed randomized clinical trial with longer duration of follow-up is still needed to confirm the results of our study. Nevertheless, we believe that the results from our study can provide further insight into the advancement of endoscopy-assisted techniques for the management of early breast cancer.
Conclusion
Our systematic review and meta-analysis of nine studies showed that E-BCS was superior to the conventional technique in improving incision length and cosmetic score for patients with early breast cancer. However, this superiority was not accompanied by an increased rate of postoperative complications or local recurrence, but there was an increased operation time and drainage volume with the use of the endoscopy-assisted technique. Overall, E-BCS is feasible and safe for the management of early breast cancer with shorter incision length and better cosmetic results than C-BCS.
Footnotes
Acknowledgements
None.
Conflict of interest
The authors report there are no competing interests to declare.
Data availability statement
The authors confirm that the data supporting the findings of this study are available within the article [and/or] its supplementary materials.
Funding
None.
Author contributions
AG and LRYH conceived the idea for the manuscript and collected the data needed for the analysis. Data analysis and interpretation were done by LRYH. AG and LRYH drafted and revised the manuscript to the final version. Supervision was done by AG.
