Abstract
INTRODUCTION:
Identifying Ki67, a monoclonal antibody that recognizes proliferating cells, is important for defining the level of proliferative activity among patients with breast cancer. The purpose of our study was to evaluate the correlation between Ki67’s expression and histopathological grade, tumor size, disease-free survival (DFS), and overall survival (OS) among breast cancer patients.
METHODS:
Our retrospective cohort study involved examining 114 patients with breast cancer at our institution from January 2018 to December 2019. Participants were retrospectively followed to determine the progression of their disease, and their 2-year progress was examined with survival analysis, especially regarding whether they had postoperative relapse (i.e., DFS) or had died since being diagnosed (i.e., OS). The data were processed with a chi-square test and Kaplan–Meier test, with significance set at p < 0.05.
RESULT:
The overexpression of Ki67 correlated significantly with histopathological grade (p = 0.001), tumor size (p = 0.001), DFS (p = 0.001), and OS (p = 0.003).
CONCLUSION:
Ki67’s overexpression is significantly correlated with the tumor size, DFS, and OS of patients with breast cancer.
Introduction
Breast cancer, the most common cancer in women, accounts for one-third of all malignancies affecting women and has a high metastatic capacity that causes a high rate of mortality [1,2]. Treatment decisions are crucial steps for patients with breast cancer. In that process, molecular biomarkers such as the estrogen receptor, progesterone receptor, and HER2 play significant roles in assuring that patients with breast cancer receive appropriate treatment, namely by aiding prognosis and indicating which therapy or therapies would be most appropriate [3,4]. In turn, improved diagnosis using novel biomarkers is essential to improving the general diagnosis and treatment of breast cancer [2,5].
Proliferation marker Ki67, one of the most controversial parameters in breast cancer, is a nuclear protein first identified by Gerdes et al. in 1983 in a Hodgkin lymphoma cell line [6–8]. Associated with cellular proliferation, Ki67 is expressed in cell cycle phases S, G1, G2, and M phase in the nucleus and can be detected by immunohistochemical techniques [9]. Its expression differs throughout the cell cycle, peaks during mitosis, plays a role in cell division, and, as per recent evidence, may function in ribosomal RNA synthesis [10]. The gene coding for Ki67, MKI67, is located on chromosome 10q25-ter, and in normal breast tissue, Ki67 was observed to be expressed at low levels in estrogen receptor-negative cells [9,10].
Although monoclonal antibody Ki67 immunostaining can help to assess the growth of malignant cells, no acceptable cutoff value for Ki67 has been identified. Nevertheless, in routine clinical work, Ki67 is widely determined in breast cancer tissue and used as an additional factor for decision-making about adjuvant treatment strategies [11]. The aim of our study was twofold: to measure Ki-67’s average expression among patients with breast cancer in Samarinda and to determine whether Ki-67 can be used as a prognostic and/or predictive factor in determining the optimal therapy for each patient.
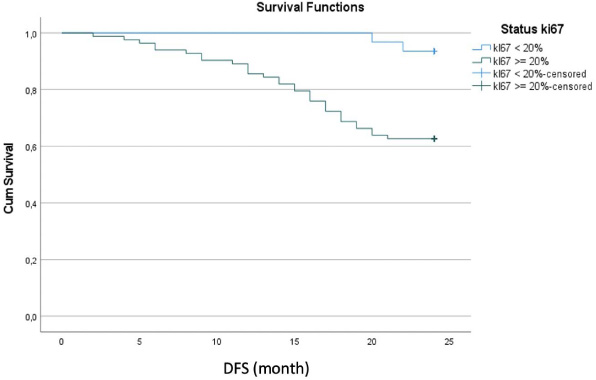
Kaplan–Meier curve for DFS rate.
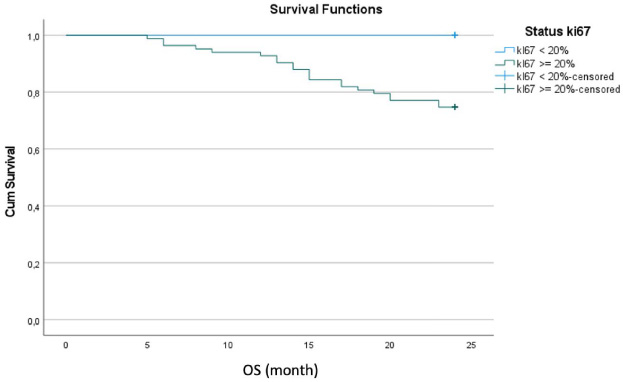
Kaplan–Meier curve for OS rate.
Clinical protocol
The study was performed in the Department of Surgery in a tertiary care hospital in Samarinda, East Borneo, Indonesia. Ethics committee approval was obtained for the study (No. 127/KEPK-AWS/VIII/2019) at Abdul Wahab Sjahranie Hospital in Samarinda, East Borneo, Indonesia. The population under study was women with breast cancer admitted to our institution from January 2018 to December 2019. Thus, all 114 patients with breast cancer admitted to our oncology unit within the past 2 years were enrolled in the study. The nature, methodology, and risk involved in the study were explained to patients, and their informed consent was obtained.
Data representing the patients were collected from the Medical Record Section at the hospital (2018–2019), first to determine whether patients met the study’s inclusion and exclusion criteria. The inclusion criteria were being a woman, having breast cancer but never having received therapy, having no other malignancies, never having undergone mastectomy or breast-conserving treatment, and having complete data concerning histopathological grade and immunohistochemistry. Conversely, the exclusion criteria were having received mastectomy or breast-conserving treatment, not having full data regarding histopathological grade and immunohistochemistry, immunohistochemistry staining results not showing a representative picture, and suffering from other types of cancer (e.g., ovarian, liver, and lung cancer).
Data analysis
The cutoff value for Ki67’s expression was assessed as weak (i.e., low) when staining in 100 visual fields revealed <20% expression and strong (i.e., high) with 20% staining on the nucleus and 10% staining on the cytoplasm. Histopathological grade was assessed with the pathological gradation based on the Nottingham Combined Histologic Grade, with the Elston–Ellis modification of the Scarff–Bloom–Richardson grading system. Prior to each patient’s surgery, tumor size was clinically determined according to its longest diameter by using 150-mm calipers.
Disease-free survival (DFS) and overall survival (OS) were evaluated from patients’ medical records, and data about disease recurrence and death following diagnosis with breast cancer and undergoing treatment were obtained from the patient and/or their family over the phone.
The collected data were analyzed in Statistical Package for the Social Sciences for Windows version 24 and tabulated. To determine the correlation between Ki67’s overexpression, histopathological grade, tumor size, DFS, and OS, the chi-square test and Kaplan–Meier test were performed, and results were considered to be significant at p < 0.05.
Results
Table 1 presents the characteristics of the 114 patients, who were from 32 to 72 years old. Their Ki67 values ranged from 4% to 85% (M = 41. 4%), with 27% expressing low values (i.e., <20%) and 73% expressing high values (≥20%).
Histopathological grade varied, with 15% having low grade, 33% having moderate grade (33%), and 52% having high grade. Tumor size ranged from 3 to 12 cm in diameter, with 17% being ≤5 cm, 64% being 5–8 cm, and 19% being >8 cm, for a mean of 6.8 cm. With a mean of 21.1 months, DFS rate was 71%, whereas OS rate, with a mean of 22.3 months, was 81% (Table 2).
Characteristics of patients
Characteristics of patients
Age, Ki67 value, tumor size, DFS, and OS
The overexpression of Ki67 significantly correlated with histopathological grade (p = 0.001), tumor size (p = 0.001), DFS (p = 0.01), and OS (p = 0.03), as shown in Table 3.
Correlation between Ki67’s expression with histopathological grade, tumor size, DFS, and OS
∗Chi-square test (p < 0.05).
As shown in Fig. 1, the Kaplan–Meier curve for DFS rate showed that patients with a high expression of Ki67 had experienced a progressive decrease in DFS from month to month, such that the mean DFS rate of 80% at Month 15 dropped to 65% by Month 24, when the study ended. By contrast, among patients with a low expression of Ki67, DFS at Month 15 was 100%, began to decline in Month 18, and in Month 24 was 90% (p < 0.001).
As shown in Fig. 2, the Kaplan–Meier curve for OS rate also showed that patients with a high expression of Ki67 had experienced a progressive decrease in OS beginning in Month 5 that by Month 24 was 70%. For patients with a low expression of Ki67, no decrease in mean OS occurred (p < 0.003).
The study involved evaluating the clinical significance of the Ki67 index as a prognostic marker in relation to outcomes among patients with breast cancer. The relationships between the overexpression of Ki67 and other prognostic indicators were investigated as well. The appropriate cutoff point for Ki67 in clinical practice continues to be debated and, in turn, studied among oncologists [12]. In our study, the cutoff point for Ki67 was more than 20% of positively stained cells, which approximated the one determined by Lombardi et al. [13].
Our study revealed a significant relationship between the high expression of Ki67 and higher histopathological grade, larger tumor size, higher DFS rate, and higher OS rate. The results of other studies, however, have not always shown a consistent relationship between histopathological grade and Ki67’s expression, potentially due to the heterogeneity of breast cancer. For example, in research involving 107 patients with breast cancer, Soliman et al. observed a significant relationship between Ki67’s expression with histopathological grade and mitotic count [14]. However, Madani et al., study, which involved 260 patients with breast cancer in Iran, indicated no significant relationship between Ki67’s expression and histopathological grade [15].
Our analysis confirmed that Ki67’s expression is a predictive factor for DFS and OS, as previously shown by Inwald et al. [8]. Despite numerous investigations on Ki67’s possible use as a prognostic marker for breast cancer, the optimal cutoff point and scoring protocol have not yet been standardized [14].
Spyratos et al. found that cell proliferation is a major determinant of the biological characteristics of breast cancer and identified a strong correlation between Ki67 status and cancer’s histopathological grade. Meanwhile, Inwald et al. found that Ki67 was associated with common histopathological parameters and correlated closely with tumor grade as parameters having a close association with proliferation [16].
In other work, Nishimura et al. [17] demonstrated that Ki67’s histopathological expression in primary tumors, irrespective of high or low Ki67 in recurrent tumors, correlated significantly with a lower survival rate. By contrast, Ibrahim et al. [18] reported that patients with a high expression of Ki67 in recurrent tumors showed significantly lower survival rates, irrespective of high or low Ki67 in primary tumors. Added to that, in a meta-analysis involving 12,155 patients, Ki67 positivity denoted a higher risk of recurrence and worse prognosis in patients with early-stage breast cancer [16]. Last, in another study, the relationship between proliferation markers and patients’ survival confirmed that the high expression of Ki67 is associated with a worse survival rate [18,19].
Conclusion
The results support the idea that Ki67’s expression may be a valuable biomarker and add prognostic information for patients with breast cancer to classical prognostic factors such as pathological grade, tumor size, and lymph node involvement. Future work should focus on standardizing Ki67’s assessment and specifying its role in making treatment decisions.
