Abstract
BACKGROUND:
Phyllodes tumors (PTs) are rare fibroepithelial breast tumors with diverse biological behavior.
OBJECTIVE:
This study aimed to analyze the characteristics, management, and outcome of phyllodes tumors.
METHODS:
Patients diagnosed with PTs in our institution from January 2013 to December 2017 were identified retrospectively by the ICD-10 code. Data were collected from medical records. The diagnosis of this tumor was based on histopathological results. Variables analyzed included age, tumor site, surgical option, axillary lymph nodes, chemotherapy and radiotherapy, metastases, disease-free survival (DFS), and overall survival (OS) rate.
RESULTS:
Sixty-nine cases of PTs were diagnosed during the period, of which 31 were benign, 38 were malignant, and 1 was a case of bilateral metachronous tumors. We did not find any borderline pathologic cases. The patient’s ages ranged from 20 to 71 years, with the highest number of cases (28) from the 40-49-year-old age group. The majority of patients (42) had PT on the right side. Simple mastectomy was the most performed treatment, with 53 such cases, including both benign and malignant. The axillary staging was carried out in 16 cases, of which none had nodal metastasis. Ten cases received chemotherapy, and 16 cases of malignant phyllodes received postoperative radiotherapy. We found 4 cases with distant metastases and 5 cases that developed local recurrence. The DFS rate reached 92.75%, and the overall 5-year survival rate was 84.21%.
CONCLUSIONS:
The incidence of PTs is highest in women in the 3rd and fourth decades of age. Simple mastectomy and wide excision are the treatments of choice, with a low recurrence rate in both benign and malignant PTs cases.
Introduction
Phyllodes tumors are rare fibroepithelial neoplasms. The incidence is only about 0.3–0.5% of all breast tumors, while the frequency of malignant lesions varies from about 5% to 30% [1–3]. The phyllodes tumor was first described by Johannes Muller as cystosarcoma phyllodes in 1838 [3–5]. Since it has a dramatic clinical manifestation and an aggressive histological finding, it attracts extra attention even though most of them have a benign nature. Hence, the term sarcomas are considered less accurate. Phyllodes tumors are also called giant fibroadenomas, cellular intracanalicular fibroadenomas, and some other names. These tumors were believed to be benign until 1943, when Cooper and Ackerman reported the potential biology of it towards malignancy. Currently, the nomenclature used is according to the World Health Organization (WHO, 1982), namely a phyllodes tumor [6,7].
These tumors are present at all ages, but most are found in the 3rd and fourth decades of life, peaking between the ages of 45 and 49 years old. Epidemiological data show that the incidence of phyllodes tumors is generally higher in White, Latina, and East Asian women [6].
The molecular correlation of the histologic level and malignant behavior and the genetic changes driving phyllodes tumor development remains unclear. The preferred pathogenesis theory is epithelial-stromal interaction. Leaf-like morphological assemblages with subepithelial stromata are indicative of a close relationship between epithelial and stromal elements [7,8].
In 1981, WHO adopted the term phyllodes tumors and classified them into types: benign, borderline, and malignant, based on stromal characteristics and updated in 2012 [3,9–11]. These characteristics include the degree of atypical cellular stroma, mitotic activity per 10 large visual fields, the presence or absence of overgrowth stroma, and infiltrative tumor margins or well-defined tumor margins. Overgrowth of stroma has been associated with metastatic activity, which is absent in benign and borderline types [12–14].
Methods
This study is a retrospective study that aims to analyze the characteristics of phyllodes tumors at Wahidin Sudirohusodo Hospital in Makassar, Indonesia, which has not previously been reported. We conducted a descriptive study with retrospective analysis by describing data on patients who have experienced phyllodes tumors with ICD-10 code D24 and D48, based on medical records for the period January 2013–December 2017. The confirmed diagnosis of this rare tumor was based on histopathological results. All cases will be analyzed based on demographic parameters. Demographic characteristics were considered, including age, tumor location, surgery, enlargement of the axillary lymph nodes, chemotherapy and radiotherapy measures, metastases, and local recurrences. For histopathological aspects, the sample is divided into three groups: benign, borderline, and malignant. Patients were then followed up through the study period to obtain disease-free survival rates and overall survival rates.
Result
There were 69 cases of phyllodes tumor during the period January 2013–December 2017. All patients were female, and no cases were obtained by borderline histopathology. The characteristics are summarized in Table 1.
Patient characteristics (n = 69)
Patient characteristics (n = 69)
Table 1 indicates that the largest age group was 40–49 years old had 28 cases (40.58%), the second most was the 30–39-year-old group with 18 cases (26.08%), followed successively by the 50–59-year old group with 14 cases (20.29%), the age group 20–29 years with 7 cases (10.14%), and the groups of 60–69 years and 70–79, each with 1 case (1.45%). We found that the youngest age in the study period was 20 years and the oldest 71 years from the data collected.
Based on histopathology, 31 cases of benign phyllodes tumor (44.93%), with 1 case of benign bilateral synchronous and one benign case with invasive ductal carcinoma (IDC) component. There were 38 malignant cases (55.07%), with one bilateral metachronous tumor (1.45%). We obtained a case of a 33-year-old patient with a malignant tumor at 35 weeks of gestation and performed a simple mastectomy.
The most common location of the tumor was in the right breast, with 42 cases (60.87%), while on the left breast, there were 25 cases (36.23%). There was also 1 case (1.45%) of a patient aged 24 years with a benign bilateral synchronous tumor and a case (1.45%) of a patient aged 45 years with bilateral metachronous tumor results on the right breast and malignant results on the left breast.
For surgery, simple mastectomy is still the primary choice for cases of benign and malignant phyllodes tumors. The number of simple mastectomies in benign cases was 21, and in malignant cases, 32, 53 cases (76.81%). Wide excision is still the second choice. The number reached 11 cases (15.94%), 8 cases in benign results, and 3 cases in residing malignant cases. In the metachronous tumor, wide excision was performed on the right breast with benign results and a simple mastectomy on the left breast with a malignant result. Radical mastectomy should be considered for patients with tumors that have infiltrated the pectoralis muscle. The above data were performed radical mastectomy in benign patients containing components in invasive ductal carcinoma (IDC), and there were 4 cases of malignant radical mastectomy, for a total of 5 cases (7.14%).
Enlarged axillary lymph nodes were found in 16 cases (23.18%) in both benign and malignant cases, but the histopathological results did not find any metastatic cells. All preparations showed sinus histiocytosis. There were 9 cases of enlarged axillary lymph nodes and malignant lymph nodes in 7 cases in benign cases.
Chemotherapy was performed in 10 cases (14.49%) and radiotherapy in 16 cases (23.18%). There was a benign case undergoing chemotherapy because the histopathological results contained IDC components, the chemotherapy regimen used were paclitaxel and doxorubicin for six cycles. As many as 9 cases of malignancy underwent chemotherapy, 3 cases of which metastases to the lungs, 2 cases used the ifosfamide, doxorubicin, mesna, vincristine, and dacarbazine regimens, and 1 case used the cyclophosphamide regimen, epirubicin, 5-Fluorouracil, and dacarbazine. In 6 cases without metastases, we found that 1 case used ifosfamide-epirubicin-mesna regimens, 3 cases used ifosfamide-doxorubicin-mesna, 1 case used paclitaxel-doxorubicin, and 1 case used docetaxel-doxorubicin-cisplatin.
In our study, radiotherapy was performed outside the Wahidin Sudirohusodo General Hospital due to the unavailability of equipment. Even though radiotherapy patients are outside hospitals, periodic controls are still undergoing in the outpatient tumor surgery polyclinic at Wahidin Sudirohusodo General Hospital. The data collected included a total of 38 malignant cases, with 9 cases of chemotherapy, 16 cases of radiotherapy, and 13 other cases that refused follow-up.
Although almost non-existent, the data showed a metastatic rate of 4 cases (5.78%). In 1 case with three metastatic locations, namely in the lungs, pubic bone, and eyes, chemotherapy was not performed because the patient refused chemotherapy. Meanwhile, in the other 3 cases, metastasis had spread to the lungs, and chemotherapy was performed.
During the 5-year study period, we obtained data for patients with local recurrences. There were 5 cases (7.24%) of local recurrence, with 2 cases of benign histopathology, with a distance of 2 years and 5 years after the first diagnosis, and the histopathological results after recurrence were the same as before. Meanwhile, in malignant cases, there were 3 cases of local recurrence at the same time relatively faster, that is, in a matter of months, 1 year, and 2 years and no radiotherapy after surgery. So that there were 64 disease-free cases in the 5 years of the study with a disease-free survival rate reaching 92.75% (Fig. 1 and Fig. 2).
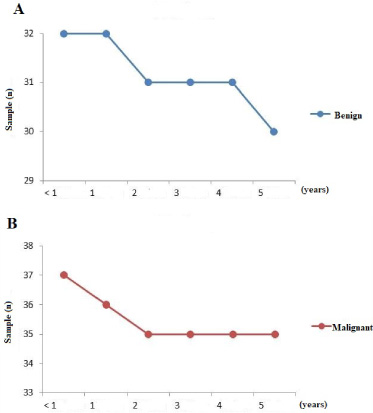
Disease-free survival curves: (A) benign and (B) malignant.
We performed follow-up on malignant cases in the study period to see the overall survival rate. We found a survival rate of 84.21%, with 6 of those with malignant cases dying (15.79%), including 4 cases dying under a year after diagnosis and 2 cases after a year. In one case, a patient aged 50 years died within six months of diagnosis. Another patient with metastases of the lungs, pubic bone, and eye, had a simple mastectomy but did not perform chemotherapy because the patient refused. Furthermore, less than a year after diagnosis, three patients died due to febrile neutropenia at ages 20 years, 22 years, and 56 years. These three patients had undergone surgery, chemotherapy and had no metastases. Another 2 cases died over a year after diagnosis, a patient aged 35 years with a recurrent tumor, who had lung metastases and had undergone surgery and chemotherapy. The other patient was 49 years old, had no metastases, and was given six cycles of chemotherapy.
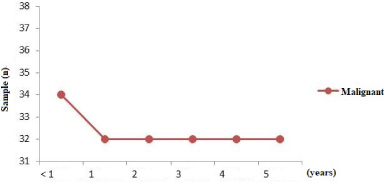
Overall survival curve for phyllodes tumor at 5 years.
Phyllodes tumors are rare breast tumors. In our study, there were only 69 cases in the 5-year study period. Ye-Fa Toh et al. reported that from January 2004 to December 2011, there were a total of 64 phyllodes tumor patients based on histopathological results at the Faculty of Medicine, University of Malaya, all of which were female [15]. Fairuz et al. examined cases from 2011 to 2014 at Hasan Sadikin General Hospital in Bandung, Indonesia, and found 64 cases of phyllodes tumor with 40 patients (66.7%) benign, 33 patients (53.2%) having an infected right breast, and 1 case of bilateral [16]. Meanwhile, Hassouna et al. reported that there were 106 cases of phyllodes tumor in Tunisia during fifteen years. Sixty-two of the cases (58.49%) showed a benign tumor, 28 cases were malignant (26.4%), and 16 cases (15.09%) showed borderline [17]. Meanwhile, in Singapore, the incidence of phyllodes tumors was reported to be only 6.92% of all breast malignancies [13].
Malignant tumors can occur in 10–30% of all types of phyllodes tumors. There is a great deal of literature regarding invasive ductal carcinoma of the breast and its potential malignity, but phyllodes malignant tumors are not widely studied due to the scarcity of cases [18].
Gerges et al., in their study, reported a benign patient (3%), 13 borderlines (37%), and 21 malignant patients (60%). Although in other studies, the average number of benign cases was up to 50%, in the study of Greges et al., only one patient was benign and, in our study, there were more cases of malignancy. It could be explained due to the regional surgeons treating all benign cases and not sending them to the cancer center where Greges et al. were [19]. Likewise, our study found that the percentage of malignant cases was more than the benign cases. Besides, Testori et al. reported that the percentage of malignant incidence varies from 8% to 45% [20] because of the low incidence of phyllodes tumors.
Carcinoma that develops concurrently with phyllodes tumors is rare, with only 26.4% reported in the literature. In previous reports of coexistence, the mean ages of patients were 39.6 years (range: 14–71). Benign, borderline, and malignant phyllodes tumor mean ages were 33.5, 41, and 41.1 years, respectively. Malignant and borderline phyllodes tumors occurred in 77.3% of patients older than 35 years versus 45.2% for benign phyllodes tumors [17]. In our data, we found a case of a patient aged 56 years old with a benign appearance and the presence of an IDC component, which was in line with a case reported by Parfitt et al. [21] of a 26-year-old woman with a lump on the left breast with the result that the histopathology of phyllodes tumor was benign and contained an epithelial component of ductal carcinoma in situ, with multiple foci of invasive breast carcinoma [21].
From our data, we found that 1 case of a 33-year-old woman with 35 weeks of gestation underwent a simple mastectomy with a malignant histopathology result. Ahmet Bal et al. [22] reported that a 32-year-old woman with a prior history of surgery three years previous, with a left breast tumor, performed a wide excision with a 1 cm border. Then the patient felt a lump again 18 months after the first operation, and the patient was at the time 20 weeks pregnant, and the lump proliferated during pregnancy [22]. Lactation has been suggested as a precipitating factor in bilateral tumor metamorphosis, whereas the role of female hormones remains unclear [23].
Despite the recommended criteria for clues to classification, the interpretation of individual histological parameters and their integration into decisions about benign, borderline, and malignant categories is highly subjective. Many authors have used the degree of stromal hypercellularity and atypia, rate of mitosis, stromal overgrowth, and microscopic margin properties as histological features [3,9,17]. The presence of necrosis is also considered, whereas Pietruszka and Barnes incorporated stromal overgrowth into the main definition of phyllodes tumor. Others have divided the tumor into benign and malignant and further categorize the malignant group into low grade and high grade [24]. Most cases of phyllodes tumor occur in the 4th decade, are rare in adolescents, and can occur at any age [3,14]. Carlos et al., of Colombia’s surgical department, reported 77 cases of phyllodes tumor in 2002–2012, and the mean age was 47 years [25]. The 4th decade of age is also supported by the study of Gerges et al. in Kuwait, who reported that out of 35 cases, the mean age was 42 years [19]. Cheng-Chiao Huang et al. in Taiwan reported the patient age at primary diagnosis of phyllodes tumor ranged from 12 years to 76 years (mean age 39 years). The mean age of patients with benign phyllodes tumor was 36 years, and the mean age for patients with malignancy was 42 years [26]. Fairuz et al. reported in their study that the age range obtained was 19–66 years [16]. Meanwhile, Tan et al. in Singapore had a younger average age of 25 to 30 years [13].
Puay-Hoon Tan et al. reported no significant comparison between the right and left sides, where the right is 50.1%, the left 49.3%, and bilateral synchronous 0.6% [24]. The results of this study were also obtained by Fairuz et al., where the right side was slightly more frequent than the left, namely 33 patients on the right, 28 patients on the left side, and 1 patient was bilateral [16]. Cheng-Chiao Huang et al. reported that of 170 patients, only two patients had synchronous bilateral tumors [26]. Supriya et al. reported that out of 33 patients studied, and there was only one patient with bilateral [27]. Rajesh et al. [23] found a nulliparous woman aged 24 years with bilateral metachronous tumors. This is very rare because most of the reported cases of bilateral phyllodes tumors occurred in young women associated with pregnancy and lactation [23].
Cheng-Chiao et al. reported that 80 cases underwent wide excision surgery, 12 cases of simple mastectomy, and 18 cases of radical mastectomy [26]. Supriya et al. reported 7 cases underwent wide excision, 20 cases underwent a simple mastectomy, and 6 cases underwent radical mastectomy [27]. Surgery is always the first choice in cases of phyllodes tumor, regardless of the histological subtype. Most studies recommend an excision margin of more than 1 to 2 cm based on evidence that local recurrences are more common in patients with an operative margin of less than 1 to 2 cm. However, marginal excision is often not possible in large phyllodes tumors because of the narrow area of breast tissue around the tumor mass [16]. Determination of tumor cell-free at the surgical incision boundary is difficult when the tumor mass is very large. Simple mastectomy is performed on the condition that a lump has occupied almost the entire breast so that only a small amount of healthy breast tissue remains [16,28].
Wang et al. reported that although surgery is the main choice for the treatment of phyllodes tumors, the surgical technique of choice is controversial. The excision of tumors with a negative edge of 1 to 2 cm is recommended. A simple mastectomy is preferred if a negative threshold could not be reached. Local recurrence is almost unavoidable despite mastectomy. However, the best type of surgery for isolated recurrent phyllodes tumors is rarely reported in the literature. It is recommended transverse rectus abdominus myocutaneous (TRAM) flap [29].
There are only a few studies in the literature reporting lymph node metastases. Treves reported 1 case of axillary lymph node metastasis out of 33 cases of phyllodes tumor. Out of 94 cases studied by Norris and Taylor, 16 cases presented with enlarged lymph nodes, and only 1 case had metastases, histologically. Reins found 11 cases of enlarged lymph nodes out of 55 cases, but only 1 case showed metastasis. Minkowitz also reported 1 case with axillary lymph node metastases [14,30]. While Gerges et al. reported mastectomy in 20 cases and staging of enlarged axillary lymph nodes was performed intraoperatively in 9 cases, and axillary dissection was performed in 4 cases, and 5 cases were only biopsied, all of which showed no metastasis on histopathologic results [19]. Axillary lymph node enlargement and involved borderline or malignant histopathology were significantly associated with recurrence rates, while borderline positive and postoperative radiation therapy was not associated with recurrence rates [31].
Axillary lymph node involvement is extremely rare in phyllodes tumors, so routine dissection of the axillary lymph nodes is often unnecessary [20]. The proportion of patients with lymph node metastases is <1% (lymph node enlargement about 10%), and routine axillary dissection is not recommended. Axillary dissection is required when histologically positive for malignant cells. As with soft tissue sarcomas, it rarely causes lymph node metastases [6]. National Comprehensive Cancer Network guidelines recommend mastectomy with axillary lymph node dissection if there is an enlargement of more than 4 cm and the biopsy shows an aggressive type of tumor (capsule infiltration, high mitotic rate, and high atypical cellular level). If there is an indication of lymph node involvement clinically or on imaging studies, a needle biopsy with ultrasound guidance should be performed. If the result is negative, an incisional biopsy may be considered [32,33].
Turalba et al. demonstrated that doxorubicin- and ifosfamide-based chemotherapy had some success in women with metastatic phyllodes tumors [34]. Systemic chemotherapy and hormonal therapy are still under debate in the case of a phyllodes tumor. A 10-year observational study with patients treated with doxorubicin plus dacarbazine or just observed after surgery failed to improve outcomes compared with observation [35]. Based on the limited data available on systemic therapy of soft tissue sarcomas, adjuvant chemotherapy may be offered to patients with large (>5 cm), high-risk or recurrent malignant tumors. Hormonal therapy is ineffective in phyllodes tumors [18].
The benefits of adjuvant chemotherapy are controversial. There are no prospective or randomized studies of adjuvant chemotherapy in phyllodes tumor strains. An observational study in Mexico published by Morales-Vásquez reported 28 cases with phyllodes malignant tumors treated within ten years (1993–2003) with doxorubicin plus decarbazine chemotherapy. There were no local recurrences or differences in survival in the two study groups [35]. However, this is a small, retrospective, uncontrolled study that did not use ifosfamide, which is a drug superior to decarbazine, which is currently the standard for other sarcoma therapies. Lately, international management guidelines do not recommend the use of routine chemotherapy and may be considered for patients at high risk of recurrence (high-grade, large tumors, or young age). Systemic chemotherapy is considered to have poor benefit as adjuvant [25,33].
Ossa et al. reported that out of 77 cases of phyllodes tumor, 26 benign cases were managed surgically, 6 of 24 borderline phyllodes tumors underwent radiotherapy, and 17 of 27 malignant phyllodes tumors underwent radiotherapy [25]. Gerges et al. reported 4 cases of malignant radiotherapy and no radiotherapy in 13 borderline cases and 17 malignant cases [19].
The benefits or roles of adjuvant therapy are not well explained. This is due to the lack of randomized trials and the rarity of tumor types. There is currently no consensus on radiotherapy. In a prospective multi-institutional study, when adjuvant radiotherapy in 46 cases with borderline or malignant phyllodes tumor was treated with wide local excision followed by radiotherapy, there was no local recurrence with a mean follow-up of 56 months [18].
Radiotherapy is recommended to patients for local control, although its impact on disease-free survival or overall is controversial. The role of adjuvant chemotherapy also requires further research. There is no consensus on the role of chemotherapy in phyllodes malignant tumors [29]. Adjuvant radiotherapy and chemotherapy can be given to patients with high-grade tumors, marginally positive surgery, or postoperative recurrences [20].
Supriya et al. reported that the rate of distant metastasis in phyllodes tumors varied widely, ranging from 6.6% to 70%. Most occurred in the lungs (84.5%) and bones (39%) [27]. In general, borderline tumors can metastasize, but this is uncommon. In the case report by Moffat et al., only 4% of patients with borderline tumors metastasized. Malignant tumors are more common with metastases. In several studies, the metastatic rate for malignant tumors has been reported to be between 9% and 32% [18]. The most common route is hematogenous spread, which occurs predominantly in the lungs, pleura, and bones, as in sarcomas [20].
Overall, 10% of patients with phyllodes tumor develop distant metastases, and they occur in about 25% of patients with malignant tumors. The most common sites for distant metastases were the lungs (66%), bone (28%), and brain (9%), and, in rare cases, the liver and heart. Metastatic risk does not appear to be influenced by the initial rate of surgery and is commonly determined by tumor biology. Metastatic phyllodes tumor has a poor prognosis and no long-term survival [6].
In our research, we found 5 cases (7.24%) of local recurrence so that the disease-free survival rate reached 92.75%. Niazi et al. reported that the recurrence rate was found in 10 cases (13%), with a median time of up to 12 months. In a Japanese study, 17 cases recurred within a mean duration of 4 years [31]. Studies by Kapiris et al. and West et al. show that among patients who have metastases, 60% to 85% have developed local recurrences [27]. Several studies have reported recurrence rates of malignant phyllodes to be around 20% to 65%. Some of the factors that show an increase in local recurrence changes are tumor size, positive operating margin, stromal overgrowth, high mitotic count, and necrosis. Oktar et al. found that overgrowth increases the likelihood of local recurrence by sevenfold, while if the operating limit is less than 1 cm, the risk increases fivefold, and if the tumor size is more than 10 cm, the recurrence prevalence is four-times greater in larger tumor size than in small [18]. However, local recurrence is not always associated with the development of distant metastases [6,12].
Other studies have also demonstrated a local recurrence rate of 15% to 20% and have shown the association with positive excision margins rather than tumor grade or size [32]. The challenge lies in predicting which tumors will recur locally or metastasize. Since not all malignant phyllodes tumors are recurrent or metastatic, they may recur in benign tumors [18]. For recurrent benign tumors, the choice of surgery depends on the surgeon’s preference. Some surgeons perform mastectomy, and other surgeons perform local excision or wide excision to achieve negative margins [26].
Two patients with malignant phyllodes tumor who died of metastasis underwent simple mastectomy and radiotherapy [18]. Another American study reported benign and borderline phyllodes tumor patients with disease-free survival rates of 91% at 5 years and 79% at 10 years. Meanwhile, the survival rates for malignant cases were 82% after 5 years and 42% after 10 years [32].
Patients with benign phyllodes tumors had a 100% survival rate, while the survival rate for malignant cases was 91.5%. In comparison, a recent study from Taiwan reported a 5-year disease-free survival of 59%, while the overall 5-year survival rate was 81% [31]. Death is usually associated with metastases or recurrent malignant tumors [31,36]. Most patients who develop metastases die within three years of diagnosis [36].
The prognosis for benign phyllodes tumor is excellent, and the chance of recurrence is also meager. The prognosis for patients with borderline or malignant tumors varies. Borderline tumors have the potential to become carcinomas, and borderline and malignant phyllodes tumors have the potential to metastasize [36]. The prognoses for phyllodes malignant tumors are very poor, and the role of the various treatment modalities is unclear due to the rarity of the disease [20]. There are no clinical prognostic factors that could predict local recurrence or metastasis. The patient’s age is not meaningful enough, but tumors that appear in adolescence appear to be less aggressive regardless of histological type [6].
Conclusion
The incidence of phyllodes tumor (PT) is highest in women of the 3rd and fourth decades of age. Simple mastectomy and wide excision are the treatments of choice, with a low recurrence rate in benign and malignant PT cases.
Footnotes
Acknowledgements
The authors do not have acknowledgments.
Conflicts of interest
None.
Funding
None.
