Abstract
This study aimed to investigate the impact of human umbilical cord–derived mesenchymal stromal cells (hUC-MSCs) on food allergy (FA) mice induced by ovalbumin. The percentage of regulatory T cells (Tregs) was assessed by administering hUC-MSCs intravenously to FA mouse models with oral challenges, allergic responses and levels of related allergic cytokines. The phenotypes of hUC-MSCs were analysed using flow cytometric analysis. Immunohistochemistry was used for histology observation. Real-time polymerase chain reaction (PCR) was used for gene expression. Jejunum tissue was analysed by transcriptome sequencing. Our results demonstrated that in the current FA model, hUC-MSC therapy significantly alleviated allergic responses and diarrhoea. Levels of immunoglobulin E (IgE), as well as cytokines, such as interleukin (IL)-6 and tumour necrosis factor-α associated with T helper 2 cells, were reduced. Conversely, transforming growth factor (TGF)-β levels increased with hUC-MSC therapy. In addition, enhanced TGF-β expression along with IL-10 messenger ribonucleic acid levels and an increased percentage of CD4+Foxp3+ Tregs were observed. In long-term FA mice models, hUC-MSC therapy exhibited sustained effects in mitigating rectal temperature decrease and mortality rates while reducing the levels of IgE, IL-6 and proportion of IgE+ cells; it also elevated TGF-β levels. Furthermore, hUC-MSC therapy attenuated pathological injury in both current and long-term FA mouse models. Transcriptome sequencing showed that upregulated differentially expressed genes were mainly concentrated in neural activation–ligand interaction, the cyclic guanosine monophosphate–protein kinase G signalling pathway and the TGF-β signalling pathway. The hUC-MSC therapy holds promise for alleviating both immediate and persistent FA conditions; targeting TGF-β and IL-10 secreted by hUC-MSCs may be a potential approach for treating FA.

Introduction
Food allergy (FA), which affects 10% of the population in Europe and 8% of children worldwide, has emerged as a significant epidemiological trend in allergic diseases over the past two decades globally. This increase may be attributed to various factors, such as genetic predisposition, dietary habits, environmental influences and immune responses 1 . FA typically manifests during childhood and reaches its peak during adolescence. Children experience a greater burden of allergic disease than adults due to their immature immune system characterised by an enhanced T helper 2 (Th2) cell polarisation. Consequently, any food can potentially act as an allergen, with milk proteins being the most common triggers for children2,3. Furthermore, nearly 30% of patients exhibit multiple types of FA that can progress into other allergic conditions, such as asthma, eczema, rhinitis or even inflammatory bowel disease. Among these allergies is egg albumin–induced FA in children, where ovalbumin (OVA) accounts for approximately 54%–69% of egg albumin content 4 .
FA represents a complex and continuous immune response towards food allergens involving several key steps: dendritic cells capture antigens; IgE-mediated effector cells are activated and bind to antigenic epitopes; this interaction leads to the release of various cytokines, including histamine, interleukin (IL)-4, IL-6 and IL-33; regulatory T cell (Treg) function is impaired, resulting in heightened allergic reactions along with physiological dysfunction and subsequent clinical symptoms 5 . Skin sensitisation exacerbates food-related FA by amplifying systemic Th2 responses6,7. Unfortunately, there is currently no effective treatment available for FA except for emergency adrenaline administration. Long-term usage of glucocorticoids or antihistamines is not recommended due to potential adverse effects 8 . Studies have shown that avoiding food antigens can increase the risk of immediate allergic reactions in patients with food-triggered atopic dermatitis 9 . In addition, long-term administration of small doses of oral antigens is necessary to manage food allergies successfully through oral immunotherapy (OIT). However, only 50% of patients complete the treatment, and just 10% achieve sustained unresponsiveness within 2 years10,11.
In recent years, mesenchymal stromal cells (MSCs), particularly human umbilical cord–derived MSCs (hUC-MSCs) with minimal immunogenicity, have shown promising potential in treating allergic and autoimmune diseases by secreting paracrine cytokines, such as transforming growth factor (TGF)-β and IL-10, which enhance immune tolerance12,13. Unlike bone marrow–derived MSCs, hUC-MSCs are more easily obtainable and can be prepared in large quantities for clinical use 14 . The human umbilical cord is a readily available, noncontroversial source of tissue, often discarded as biological waste after birth. Its collection is noninvasive and does not involve ethical concerns, unlike embryonic stem cells15–17. Furthermore, the yield of MSCs from umbilical cord tissue is substantial, and these cells can be easily isolated and expanded in culture to obtain quantities sufficient for clinical applications. Previous studies have demonstrated that both hUC-MSC and bone marrow–derived MSC treatments alleviate anaphylaxis in mice with OVA-induced FA by reducing serum levels of IgE, IL-4 and tumour necrosis factor (TNF)-α in spleen and intestinal tissue 18 . However, there is a lack of research on the sustained effects of hUC-MSC therapy. Therefore, this study aims to investigate the therapeutic efficacy as well as the continuative effects of hUC-MSC therapy in OVA-induced FA models. In addition, we explore specific cytokines released by hUC-MSCs that contribute to alleviating intestinal inflammation in FA mice. These findings support the potential use of MSCs as an effective treatment approach for FA.
Materials and methods
Animals
Four-week-old female BALB/c mice (weighing 18 ± 2 g) were obtained from SPF (Beijing) Biotechnology Co., Ltd. They were housed in microisolator units under a 12-h light/dark cycle at a temperature of 24°C–26°C and a humidity of 30%–50% at the relevant institution, following Animal Research: Reporting of In Vivo Experiments guidelines and the National Institutes of Health Animal Research Guidelines (NIH Publications No. 8023, revised 1978). This study was approved by the relevant institution.
The FA model experiment involved dividing 36 mice into four groups (n = 9 per group). Twenty-seven mice in three OVA-challenged groups received intraperitoneal (i.p.) injections of 200 µg of OVA (No. A5503, Sigma) in 1 mg of alum adjuvant (No. 239186, Sigma), dissolved in phosphate-buffered saline (PBS) to make it a volume of 200 µl on days 0, 5 and 10 19 , 20 . On day 15, after tape stripping their skin once to strengthen the FA, mice were challenged with intragastric gavage administration of 50 mg of OVA in 200 µl of PBS once in the OVA1 group and were challenged every other day six times (days 15, 17, 19, 21, 23 and 25) in the OVA6 group21–24. Food antigens were not applied to the skin after tape stripping. Mice that were challenged six times received treatment with hUC-MSCs (1 × 106 cells) intravenously on days 19 and 23 in the OVA/MSC group. Mice in the control group were i.p. injected and challenged with the same amount of PBS. Another 27 long-term mice were divided into control, ‘LN’ (long-term immunised group) and ‘LMSC’ (long-term immunised group treated with hUC-MSCs) groups (n = 9 per group). Mice in the LN and LMSC groups were immunised i.p. and orally challenged 10 times on days 15, 17, 19, 21, 23, 25, 46, 48, 50 and 52. Mice in the LMSC group received hUC-MSCs treatment (1 × 106 cells) on days 19 and 23. Mice in the control group were immunised and challenged with the same amount of PBS (Fig. 1).

Process of allergic sensitisation to food allergy mice with OVA, hUC-MSCs. OVA, ovalbumin; hUC-MSC, human umbilical cord–derived mesenchymal stromal cell.
The decision to assess the allergic response at 1 h post-treatment was based on previous findings that mMCP-1 levels peak at this time, while IgE remains unaffected 25 . In addition, our observations showed that, after the final gavage challenge, the body temperature of allergic mice gradually returned to normal within 1 h. However, after six challenges, temperature recovery became more difficult. Therefore, the allergic response was assessed up to 1 h after the final OVA challenge using a 6-point Likert scale, as follows26,27: 0 = no symptoms; 1 = <4 episodes of scratching and rubbing around nose and head; 2 = 4–10 episodes of scratching and rubbing around nose and head; 3 = >10 episodes of scratching and rubbing around nose and head; 4 = hunching and piloerection; 5 = immobility (unresponsive to nonharmful, tactile disturbance). Diarrhoea was evaluated using a 4-point Likert scale: 0 = normal stools; 1 = a few wet and unformed stools; 2 = some wet and unformed stools with moderate perianal staining of the coat; and 3 = severe and watery stools with severe perianal staining of the coat. The rectal temperature of the mice in each group was measured prior to the final challenge, subsequent changes in rectal temperature were recorded, and diarrhoea was evaluated for a duration of up to 60 min following the last challenge.
hUC-MSCs isolation, culture, differentiation and tracing
The hUC-MSCs were isolated, cultured, differentiated and traced in this study. Fresh, healthy human umbilical cords were obtained from our hospital with consent from all participants. Sterile umbilical cord samples without arteries and veins were repeatedly washed with normal saline and cut into 3- to 5-mm3 pieces. These pieces were then inoculated in T75 culture flasks and cultured at 37°C with 5% CO2 using MSC basal medium (No. NC0103&NC0103.S, YOCON) supplemented with 100 U/ml penicillin and streptomycin and 10% foetal bovine serum (FBS). The medium was replaced every 7 days initially, followed by replacement every 3–4 days after 2 weeks when the adherent cells reached 80%–90% confluence. Cultures were trypsinised using 0.25% Trypsin-EDTA (1×) (No. 25200-056, Gibco) and transferred into a new flask for further expansion. The passages within the range of 4–7 were used in this experiment. Literature supports the use of passage 5 hUC-MSCs, showing that they retain strong differentiation capabilities. In addition, proliferation rates and population doubling time remain stable between passages 3–11 28 , 29 . While low-passage (≤4) MSCs are generally preferred for maintaining optimal functionality and differentiation potential, CM-Dil labelling required further cell expansion for this experiment. We will prioritise low-passage MSCs in future studies to ensure the stability of cellular properties and functions.
In our direct experience, primary MSC cultures obtained from two newborn umbilical cords exhibited an expansion factor of approximately 7–8 times during the first passage. After further cultivation, second-passage cells were cryopreserved at a concentration of 3–5 × 10⁶ cells per cryovial, resulting in at least 50 cryovials, corresponding to a total of 1.5–2.5 × 10⁸ cells. With additional expansion (3–4 passages), the yield typically increased by an additional threefold, further demonstrating the potential for large-scale production of MSCs suitable for clinical applications.
The ability of hUC-MSC to undergo tri-lineage differentiation was measured (Shanghai Applied Cell Biotechnology Co. Ltd., China). The hUC-MSCs were cultured using osteogenesis differentiation basal medium (No. AC-1001027) for 21 days and assessed by Oil Red O (No. AC-1001021). Meanwhile, hUC-MSCs were cultured using adipose differentiation basal medium (No. AC-1001029) for 14 days and assessed by Alizarin Red (No. AC-1001022), using chondrogenic differentiation basal medium (No. AC-1001028) for 21 days and assessed by Alcian Blue (No. AC-1001023).
Flow cytometry
The phenotypes of hUC-MSCs were analysed using flow cytometric analysis. We selected passages 3 and 4 for fluorescence-activated cell sorting (FACS) identification because these passages provide a more stable and homogeneous population, ensuring accurate cellular characterisation. Primary or ≤2 passages may exhibit a mix of stem cell subtypes or other cell types, which could compromise the reliability of the identification. The hUC-MSCs were prepared at a concentration of 5 × 105 per cells, washed with cold PBS with 1% FBS solution twice and isolated by a centrifuge at 300 × g for 5 min. Single cells were then labelled with each antibody (5 µl) at 4°C for 30 min. Subsequently, the cells were washed again with PBS containing 1% FBS and resuspended in 200 µl of PBS before being analysed using a FACSCalibur flow cytometer (Bechman, DxFLEX, USA). All data were analysed using FlowJo (version 10, Tree Star, Inc., Ashland, OR, USA). The antibodies used for surface phenotype analysis were as follows: CD45-FITC (No. 304005, BioLegend), CD34-FITC (No. 347494, BioLegend), CD73-FITC (No. 344015, BioLegend), CD105-PE (No. 100-1057-42, eBioscience), CD90-FITC (No. 328107, BioLegend), HLA-DR FITC (No. 327005, BioLegend).
The instructions of the organ tissue lymphocyte isolation solution kit (No. P8870, Solarbio) were followed. After lymphocyte extraction, cells were divided into each flow tube according to the concentration of 0.5–1 × 106 and labelled as a sample tube, and a control single standard antibody tube and an unlabelled blank tube were set up. The following procedure was then followed: centrifuge at 2000 r/min for 5 min, discard the supernatant; press the mouth of the pipe on the paper towel 2–3 times, thoroughly absorb the residual liquid of the pipe mouth, vortex oscillation; wash twice with pre-cooled PBS + FBS; add 97.5 µl pre-cooled PBS + 1% FBS into the sample tube, followed by the corresponding flow antibody (in this study, CD45 antibody (103130, BioLegend), CD4 antibody (No. 96127s, Cell Signaling) 2.5 µl, IgE antibody (No. 50-1172, Innochem) 5 µl and single labelled antibody tubes were added, respectively); mix and incubate in dark ice for 45 min; add 1 ml PBS + 1% FBS, wash twice, flick and swirl well; add Foxp3 antibody 5 µl into the Foxp3 specimen tube and single antibody tube (in this study, according to the instructions of Foxp3 membrane-breaking and fixing kit (No. 00-5523-00, eBioscience), the ratio of membrane-breaking solution to aseptic water was 1:10. The corresponding membrane-breaking and fixing solution was added to each tube with 1-ml membrane-breaking and 1-ml cell-fixing solution); centrifuge for 5 min, discard the supernatant, flick and swirl to mix well; add 2 ml of film-breaking working liquid to each tube, centrifuge for 5 min, discard the supernatant, flick and swirl to mix well, repeat twice; add 100 µl membrane-breaking working solution to re-suspend cells without washing; add Foxp3 antibody and incubate at room temperature for 40 min; add 2 ml of film-breaking working liquid, centrifuge for 5 min, discard the supernatant, flick and swirl to mix well, repeat twice. Finally, 200 µl PBS cells were detected at 2°C–8°C by flow cytometry.
Histology and immunohistochemistry
Following euthanasia, the same tissue section from all mice was promptly fixed in 4% paraformaldehyde and subsequently processed for both paraffin and frozen sections. Haematoxylin and eosin staining was performed in the jejunum 30 . The hUC-MSCs were isolated and labelled with CM-Dil (Thermo Fisher Scientific, Waltham, MA, USA) according to the manufacturer’s instructions. The mice were injected with the CM-Dil-labelled hUC-MSCs (1 × 106 cells/mouse) via the tail vein. The mice in the control group were injected with 1 ml of physiological saline via the tail vein. After 48 h of treatment, the mice were euthanised; their liver, lungs, spleen, stomach and intestines were removed, and 4-μm frozen sections were cut. The CM-Dil-labelled hUC-MSCs were localised using a fluorescence microscope.
Enzyme-linked immunosorbent assay
To determine the best transplantation effect, different doses of hUC-MSCs were chosen for intravenous transplantation to treat food-allergic mice. The doses of different groups were as follows: low-dose group − 0.1 × 106 cells were transplanted into the tail vein on days 19 and 23; medium-dose group − 0.5 × 106 cells were transplanted into the tail vein each time; high-dose group − 1 × 106 cells were transplanted into the tail vein each time. The changes in serological inflammation indicators IgE and TGF-β in mice were observed in the different transplantation groups.
The levels of IgE, IL–6, TNF-α, TGF-β and histamine in the serum were quantified using commercially available enzyme-linked immunosorbent assay (ELISA) kits following the manufacturer’s instructions: IgE (No. 555248 and No. 550534, BD), IL–6 (No. EMC004, NeoBioScience), TNF-α (No. EMC 102a, NeoBioScience), TGF-β (No. 433007, BioLegend). Briefly, 96-well plates were pre-coated or coated overnight at 4°C with the capture antibody. Subsequently, 100 µl/well of serum sample was added and incubated at 37°C for 90 min. After five washes with wash buffer, 100 µl/well of biotinylated detection antibody was added and incubated at 37°C for 1 h. Following another round of five washes, streptavidin–horseradish peroxidase (100 µl/well) was added and incubated at 37°C for 30 min. Wells were further washed five times before adding substrate solution (100 µl per well), followed by an incubation period of 15 min at 37°C. Finally, a stop solution was added, and optical density values were measured at a wavelength of 450 nm.
Expression of cytokine messenger ribonucleic acid levels
After the sixth gavage, a total of 100 mg of jejunum tissue was flushed with ribonucleic acid (RNA)-free PBS. Total RNA extraction was performed using the RNA prep pure Tissue Kit (No. DP451, TIANGEN) following the manufacturer’s instructions. Reverse transcription was carried out using the Exscript RT reagent kit (No. RR036A, TAKARA) and random primers followed by real-time PCR amplification. Real-time PCR analysis included TNF-α, IL-10, TGF-β and β-actin genes using TB Green Premix ExTM TaqII (Tli RNase H Plus) (TAKARA Bio). The following primer pairs were used: TNF-α, forward 5′-CCTGTAGCCCACGTCGTAG-3′ and reverse 5′-GGGAGTAGACAAGGTACAACCC-3′; IL-10, forward 5′-TGCTAACCGACTCCTTAATGCAGGAC-3′ and reverse 5′-CCTTGATTTCTGGGCCATGCTTCTC-3′; TGF-β, forward 5′-GTGTGGAGCAACATGTGGAACTCTA-3′ and reverse 5′-TTGGTTCAGCCACTGCCGTA-3′; β-actin2, forward 5′-AGCCATGTACGTAGCCATCC-3′ and reverse 5′-CTCTCA GCTGTGGTGGTGAA-3′. An Mx3000p PCR system (Stratagene, La Jolla, CA, USA) was used. The PCR conditions consisted of an initial denaturation step at 95°C for 30 s, followed by 40 cycles of amplification with a denaturation step at 95°C for 5 s and an annealing/extension step at 60°C for 30 s. Target messenger RNA (mRNA) levels were normalised to β-actin mRNA as an internal control in each sample. Results were expressed as the relative ratio compared with the average of the control group.
Bioluminescence imaging cell label
The Fluc activity of the transplanted hUC-MSCs movement in this study was assessed using a Fusion FX7 Edge Imaging System (Vilber Bio-Imaging Corporation, Paris, France). Briefly, following anaesthesia, mice were subjected to imaging for 1–10 min using the imaging system, and the average radiance of the peak bioluminescence imaging signal was quantified from a fixed-area region of interest over the abdomen 31 .
Bioluminescence imaging cell label
Total RNA was isolated using TRIzol reagent, and the RNA quality was determined using a NanoDrop 2000 (Thermo Fisher Scientific, USA). Samples were analysed for RNA integrity and deoxyribonucleic acid contamination by agarose gel electrophoresis. Then, the constructed library was sequenced using a NovaSeq 6000 (Illumina, USA). Differentially expressed genes (DEGs) in the control, stem cells from human exfoliated deciduous teeth (SHED)-derived exosome-treated and TWS119-SHED-derived exosome (functionalised exosome)-treated groups were assessed using edgeR software. The DEGs between the control, SHED-derived exosome-treated and functionalised exosome-treated groups were selected by different multiples (log2 fold change) and significance levels (P < 0.05). Subsequently, gene set enrichment analysis was performed.
Statistical analysis
Data were analysed by GraphPad Prism v8 (GraphPad Software, San Diego, CA, USA, www.graphpad.com) and SPSS version 21.0 (IBM SPSS, Chicago, USA). Data are expressed as the mean ± standard deviation. Analysis of variance (ANOVA) was used for inter-group comparison between multiple groups, and the t-test was used for comparison between two groups. When a certain indicator underwent measurement at multiple time points, statistical analysis was performed using repeated measurement data ANOVA (such as temperature changes in mice). A P-value of <0.05 was considered statistically significant.
Results
Effect of hUC-MSC therapy on the reaction of FA model
The success of modelling is the basis for the credibility of research results. The hUC-MSCs were isolated, the FA model was evaluated and the response of the FA model to the hUC-MSCs was preliminarily observed. The hUC-MSCs were evaluated for MSC characteristics and identification criteria, as established by the International Society for Cellular Therapy 32 . Flow cytometric analysis revealed high expression of MSC markers, including CD90, CD73 and CD105, while haematopoietic markers, such as CD34, CD45 and HLA-DR, were absent (Fig. S1A-B). Differentiation studies also demonstrated the ability of hUC-MSCs to differentiate into adipocytes, osteoblasts and chondroblasts (Fig. S1C-E).
To determine the best transplantation effect, different doses of hUC-MSCs were chosen for intravenous transplantation to treat food-allergic mice. It was found that the differences in the serum inflammatory mediators IgE, IgG and TGF-β of FA mice after vein transplantation treatment were statistically significant between the low-dose group and the high-dose group, and the differences in IgG and TGF-β between the medium-dose group and the high-dose group were statistically significant (P < 0.05), indicating that the high-dose group had the best effect (Fig. S2).
OVA-induced mice exhibited significant anaphylactic responses characterised by scratching around the nose and head, difficulty in respiration, immobility and mortality. Allergy scores and diarrhoea scores increased significantly with repeated oral challenges in the FA model (P < 0.001); however, hUC-MSC therapy effectively alleviated anaphylactic responses (P < 0.01) (Fig. 2a, b). In Fig. 2b, the OVA/MSC group exhibited a relatively large error range, which reflects the variability in individual responses within this group. This variation may be related to individual differences in the immunomodulatory effects of hUC-MSCs. Furthermore, solid faeces accumulation was observed in the colons of the FA mice (Fig. 2c). Rectal temperature significantly increased at 15 min after the first oral challenge (OVA1) but dropped sharply at 15 min after six oral gavages (OVA6); however, hUC-MSC treatment mitigated these temperature changes in the FA mice (P < 0.05) (Fig. 2d).

hUC-MSC therapy released allergy responses in FA model. (a–b) Allergy and diarrhoea scores in control, OVA1, OVA6 and OVA/MSC groups. (c) Stools and colons of mice in different groups. (d) Rectal temperature–time curve. Each value indicates the mean ± SD. Data are representative of three independent experiments. n = 9/group. *P < 0.05; **P < 0.01; ***P < 0.001; #P < 0.05 15 min versus 0 min, &&P < 0.01 30 min versus 15 min. hUC-MSC, human umbilical cord–derived mesenchymal stromal cell; OVA, ovalbumin.
Changes in histology, inflammatory markers and cell populations with the treatment of hUC-MSCs
To clarify the effect and mechanism of action of hUC-MSCs, the changes in histology, inflammatory markers and cell population of mice after hUC-MSCs therapy were observed. Haematoxylin and eosin staining was performed to assess the therapeutic effect of hUC-MSCs in different groups. Compared with the control group, FA mice showed more pronounced oedema following repetitive oral challenges, but this pathological injury was reduced by the administration of hUC-MSCs (Fig. 3).

Histological examination of intestinal tissue with the treatment of hUC-MSCs. (a) Observation in the intestinal tissue by haematoxylin and eosin staining (magnification, ×200). (b) Toluidine blue (TB) staining was employed to visualise pathological injury and mast cells in the jejunum (magnification, ×100). hUC-MSC, human umbilical cord–derived mesenchymal stromal cell.
To assess the inflammatory response in FA mice, serum samples were collected 1 h after the final challenge on day 25 and stored at −80°C before being analysed using ELISA. Significant alterations were observed in the levels of IgE, IL-6, histamine and TGF-β expression between OVA1 and the control groups; however, no significant changes were noted in TNF-α expression. Following six oral challenges, elevated levels of IgE, IL-6, histamine and TNF-α in the OVA6 group showed a significant reduction upon hUC-MSC therapy, while the suppressed level of TGF-β was significantly increased (P < 0.05) (Fig. 4a–e).

Inflammatory cytokines levels in serum measured by ELISA. Results of (a) IgE, (b) IL-6, (c) TNF-α, (d) TGF-β, (e) histamine of mice in all groups. Each value indicates mean ± standard error of the mean. n = 9/group. *P < 0.05; **P < 0.01; ***P < 0.001. IL, interleukin; TNF, tumour necrosis factor; TGF, transforming growth factor.
In spleen tissue, IL-4 upregulation seen in the OVA6 group was downregulated with hUC-MSC treatment, whereas downregulated IL-10 and TGF-β expressions were significantly upregulated. Meanwhile, in jejunal tissue, TNF-α upregulation seen in the OVA6 group was downregulated with hUC-MSC therapy, whereas downregulated IL-10 and TGF-β expressions were significantly upregulated (P < 0.001) (Fig. 5a–f).

mRNA expressions of cytokines in the spleen and jejunal tissue. Results of qPCR for (a) IL-4, (b) IL-10 and (c) TGF-β mRNA expressions in the spleen tissue in different groups. qPCR for (d) TNF-α, (e) IL-10 and (f) TGF-β mRNA expressions in the jejunal tissue in different groups. *P < 0.05; **P < 0.01; ***P < 0.001(n = 4–6). TNF, tumour necrosis factor; IL, interleukin; TGF, transforming growth factor.
The impact of hUC-MSCs on the expression of surface markers on CD45+ T cells in the spleen was investigated. Flow cytometry analysis showed that hUC-MSC treatment significantly increased the percentage of CD4+Foxp3+ Tregs among CD45+ T cells, indicating enhanced Treg proliferation (P < 0.001) (Fig. 6a). Furthermore, hUC-MSC treatment reduced the population proportion of IgE+ cells in the spleen compared with FA mice (P < 0.001) (Fig. 6b). The original images for Fig. 6a–b have been provided as Supplementary Material for reference.
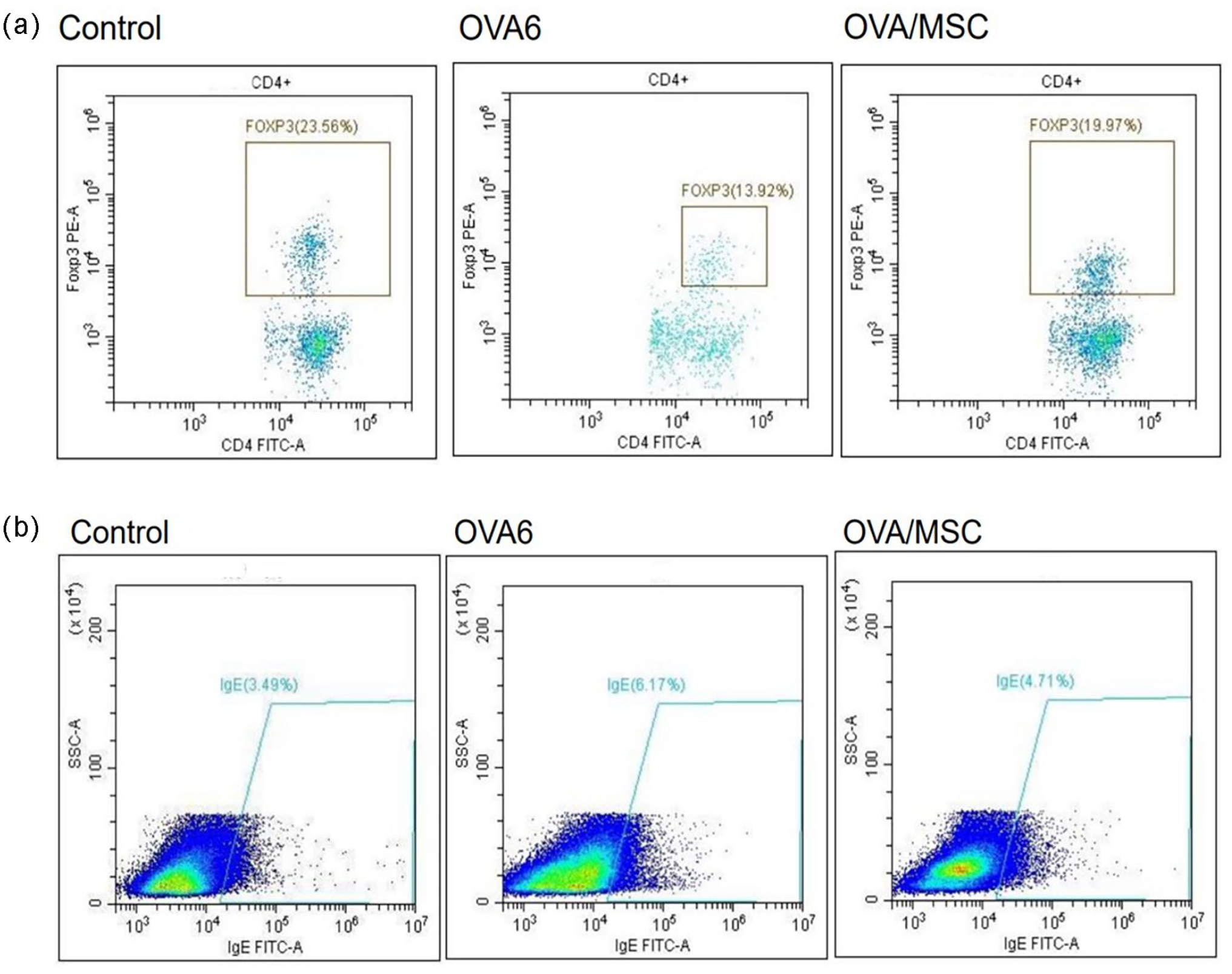
CD4+ Foxp3+ Tregs in CD45+ T cells proportion in the spleen (a). Proportion of IgE+ cells in the spleen (n = 3) (b). CD, the cluster of differentiation; Fox, forkhead box; hUC-MSC, human umbilical cord–derived mesenchymal stromal cell (n = 3).
Trafficking of hUC-MSCs in FA mice
To observe the distribution of hUC-MSCs in FA mice, the cells were labelled with CM-Dil before injection. At 24, 48 and 72 h after transplantation, frozen section results showed that the transplanted cells were mainly located in the liver, lungs and spleen. In vivo imaging results showed that hUC-MSCs mainly accumulated in the lung and liver areas and eventually mainly in the lungs, with signals in the gastrointestinal area only visible at 24 h (Fig. 7a, b).

CM-Dil hUC-MSCs labelled after 24 h, 48 h, 72 h of hUC-MSC treatment in food-allergic enteritis model. (a) Labelled cells were found in the liver, lung, spleen in frozen sections. (b) Robust signals of hUC-MSCs by bioluminescence imaging, scale bar = 100 µm.
Continuative of hUC-MSC treatment on long-term FA mice
To evaluate the sustained effect of hUC-MSCs in long-term FA mice, a second round of gavages was performed with a total of 10 oral challenges (on days 15, 17, 19, 21, 23, 25, 46, 48, 50 and 52). The results demonstrated that rectal temperature dropped more sharply at both the 30 and 60-min time points in the LN group without hUC-MSC therapy. In addition, the mortality rate significantly increased after the 10th oral challenge in long-term mice (P < 0.001). In the experiment, each group included nine mice; in the LMSC group, two mice died, and in the LN group, four mice died (Fig. 8a). Conversely, hUC-MSC treatment did not alleviate the allergic diarrhoea score or occurrence in long-term FA mice (Fig. S3). Following the last challenge on day 52, a serum sample was collected 1 h later for ELISA measurement. The results revealed significant changes in the levels of IgE, histamine, IL-6 and TGF-β between the LN and LMSC groups (P < 0.001) (Fig. 8b). Furthermore, in comparison with the LN group, the proportion of IgE+ cells in spleens from long-term FA mice was attenuated by hUC-MSC treatment (Fig. 8c). The original images for Fig. 8c are been provided as Supplementary Material for reference.

Continuative effect of hUC-MSC therapy in long-term mice. (a) The mortality and changes in the rectal temperature in long-term FA mice, n = 9. (b) Inflammatory cytokines levels of IgE, histamine, IL-6 and TGF-β in serum. (c) Proportion of IgE+ cells in the spleen. *P < 0.05; **P < 0.01; ***P < 0.001. hUC-MSC, human umbilical cord–derived mesenchymal stromal cell (n = 3).
Haematoxylin and eosin staining was also conducted on jejunal tissue. Compared with the LN group, the LMSC group receiving hUC-MSC therapy exhibited reduced injury with fewer infiltrating inflammatory cells and less apoptosis (Fig. 9).

Histological examination of jejunal tissue with the treatment of hUC-MSCs in the long-term FA mice. Observation in the intestinal tissue by haematoxylin and eosin staining. Enhanced aciophase chromatin, single nuclear inflammatory cells; edema are indicated by black, blue and red arrows, respectively (magnification, ×100).
Transcriptome sequencing analysis of mouse jejunum tissue
To explore in depth the specific mechanism of MSC treatment and the changes in intestinal tissue after treatment, next-generation sequencing on jejunal specimens was performed. The jejunum tissues of mice in the control, OVA6 and MSC groups were selected for transcriptome sequencing and bioinformatics analysis. It was found that there were multiple gene expression differences at the transcription level among the three groups. Significantly expressed DEGs were defined as false discovery rate <0.05 and fold change ≥2. Transcriptome sequencing results showed that there were a total of 1350 significantly expressed DEGs among the three groups. Among the downregulated genes in the control group compared with the OVA6 group of mice, 359 genes were upregulated after hUC-MSCs treatment. Among the upregulated genes in OVA6 mice compared with the controls, there were nine downregulated genes after hUC-MSCs treatment, including Retnlg and CH25H (Fig. 10a).

Results of significant DEGs in RNA sequencing with hUC-MSC therapy (a) Numbers of significant DEGs in RNA sequencing with hUC-MSC therapy, (b) GO analysis of significantly upregulated DEGs, (c) KEEG dotplot of significantly upregulated DEGs.
Gene Ontology analysis was performed on 359 significantly differentially expressed upregulated genes, covering three aspects: biological process (BP), molecular function (MF) and cellular component (CC). Cell components have the most DEGs. In terms of BPs, the upregulated genes are mainly related to ion transport, chemical synaptic transport, potassium ion transport and transmembrane transport. In terms of MFs, the upregulated genes are mainly related to protein connection, calcium ion binding and agonist protein binding. In terms of CCs, the upregulated genes are mainly related to the cell membrane, cytoplasm, cytoplasmic membrane, synapse and cell connection (Fig. 10b).
Kyoto Encyclopedia of Genes and Genomes analysis was performed on 359 significantly differentially expressed upregulated genes, and the results showed that the related pathways were mainly concentrated in neural activation–ligand interaction, mineral absorption, adsorption ligation, insulin secretion and cyclic guanosine monophosphate–protein kinase G (cGMP-PKG) signalling pathways, cholinergic synapses, anti-ageing, the mTOR signalling pathway, Wnt signalling pathways and the TGF-β signalling pathway (Fig. 10c).
Discussion
The α2-adrenergic agonist epinephrine is currently the first-line drug for severe FA and is crucial in preventing death from anaphylaxis, reversing hypotension, shock and other severe allergic symptoms (e.g. urticaria, bronchospasm, oedema, gastric irritation, gastrointestinal oedema, gastrointestinal irritation, gastrointestinal tract disease). The diversity of food antigens in modern society leads to the frequent occurrence of fatal food allergies. Glucocorticoids, antihistamines and other drugs can reduce the inflammatory response, but long-term use has obvious side effects; therefore, long-term use is not recommended 33 . In recent years, OIT 33 has achieved immune tolerance by exposing patients to small doses, gradually increasing the dose until the maintenance dose and then regularly ingesting specific food allergens to prevent allergic reactions caused by exposure to allergens10,11. However, for patients with severe FA, the use of allergens (even at very low doses) during OIT may trigger unpredictable severe immune reactions, which may even be life-threatening, and the course of treatment may last for several months. Years of treatment often lead to reduced compliance. Recently, the incidence of FA has increased every year, especially the number of non-IgE-mediated FA cases in infants and young children. The clinical manifestations are varied, and the organs involved are different according to individual differences. There are still no sufficient evidence-based grounds for avoiding allergenic foods during pregnancy, delaying allergenic complementary foods or applying probiotics or prebiotics to prevent FA, and there is a lack of safe and effective treatment methods. MSCs have shown therapeutic potential in allergic diseases, such as allergic asthma, rhinitis and atopic dermatitis. It plays paracrine and autocrine roles in different microenvironments, promotes immune tolerance, improves the immune state of the body in the treatment of FA and reduces the occurrence of allergic reactions, especially fatal allergic reactions. Our findings also suggest a possible role in improving intestinal barrier function, so stem cells and their related derivatives, such as exosomes, conditioned media or combined stem cell strategies, hold promise for alleviating acute and long-term FA symptoms, promoting tissue repair and reducing the inflammatory cascade.
Our study demonstrated that hUC-MSC therapy expedited the restoration of normal rectal temperature, reduced the incidence of diarrhoea and allergy scores and alleviated histological injuries in intestinal tissue in FA mice. These findings are consistent with previous studies34,35, indicating that hUC-MSCs play a beneficial role in both systemic and gastrointestinal immunity. Furthermore, our results showed significant reductions in temperature changes and mortality rates in long-term FA mice treated with hUC-MSCs, suggesting continuative effects of hUC-MSCs on long-term FA.
The Th2 cells, which are crucial components of allergic reactions, play a significant role in the humoral immune response and the development of allergies and autoimmune diseases. Any form of abnormal stimulation can disrupt the balance between Th1/Th2 cells and trigger degranulation of mast cells, leading to the release of inflammatory mediators, such as histamine, cytokines, chemokines and prostaglandins36,37. Previous studies have suggested that mast cells enhance Th2 polarisation by upregulating B-cell lymphoma 6 expression in Th2 cells. MSC therapy exerts regulatory effects on mast cell activation through a prostaglandin E2-dependent mechanism involving Toll-like receptor 7 stimulation under inflammatory conditions while also decreasing the percentage of circulating Th2 cells38–40. In our study, hUC-MSC treatment reduced levels of IgE, IL-6 and TNF-α associated with natural T cell–mediated Th2 cell polarisation. Furthermore, hUC-MSCs exhibited long-term suppressive effects on allergic inflammation in FA mice by reducing levels of IgE, histamine and IL-6. The transcriptome analysis revealed several upregulated and downregulated DEGs associated with key pathways, including the TGF-β, Wnt and cGMP-PKG signalling pathways, which are involved in immune regulation, cell proliferation and tissue repair. Specifically, the upregulation of the TGF-β pathway may contribute to the enhanced immune tolerance observed in hUC-MSC-treated mice, while the activation of the Wnt pathway could be linked to the promotion of tissue repair and regeneration. The cGMP-PKG signalling pathway is known to play a role in smooth muscle relaxation and could be relevant to reducing gastrointestinal inflammation. In addition, genes related to neural activation–ligand interactions and cell membrane integrity were significantly regulated, which may enhance the maintenance of intestinal barrier function and reduce inflammatory responses in the gut. These findings provide deeper insights into the molecular mechanisms underlying the therapeutic effects of hUC-MSCs in alleviating FA symptoms and suggest that multiple signalling pathways are involved in modulating both immune responses and tissue homeostasis.
Both TGF-β and IL-10 play a key role in the induction and maintenance of oral tolerance depending on the conversion of naive T cells into Tregs and leading of Tregs homing to the gut. It has been shown that the Tregs in the intestinal tissue had downregulatory properties for Th1 cells and played an important role in the oral immune41,42. In our study, hUC-MSC treatment significantly increased the levels of TGF-β, IL-10 and the proportion of CD4+Foxp3+ Tregs, suggesting enhanced Treg proliferation. This might be mediated through pathways such as the TGF-β signalling pathway, promoting anti-apoptotic functions, membrane integrity and cellular junctions. These mechanisms likely contribute to increased immune tolerance and reduced systemic inflammation in long-term FA mice.
MSCs regulate adaptive and innate immunity by interacting with immune cells such as T cells, B cells and macrophages. They can recognise chemical signals and screen them for tissue damage signals triggered by conditions such as tumours, injury or infection. It is attracted and chemoattracted and participates in the repair, regeneration and regulation process of the injured site 43 . MSCs are large and will quickly accumulate in the lungs after entering the blood circulation in the initial stage of transplantation and then gradually migrate to other parts. Ultimately, less than 1% of MSCs can reach and implant into the target site; however, their efficacy is very significant, which indicates that MSCs may reach target organs or target sites by secreting exosomes and interacting with recipient cells 44 . To explore how hUC-MSCs exert their therapeutic effect in the experiment, we evaluated the distribution of hUC-MSCs in mice. Our study found that transplanted cells were detected in frozen sections of the lungs, liver and spleen from 24 to 72 h after transplantation. In vivo imaging showed that they mainly accumulated in the lungs from 24 to 72 h after transplantation and enhanced signals in the digestive tract area appeared only at 24 h. Food-allergic colitis often involves multiple systems, and the spleen is also a very important immune organ in the development of FA 45 . Therefore, in the microenvironment of the allergic state in our experiment, hUC-MSCs may increase the levels of TGF-β and IL-10 through their paracrine effect, promote the differentiation of Treg cells, alleviate the inflammatory response of food-allergic enteritis and alleviate the cascade reaction.
In addition, our transcriptome sequencing results suggest that hUC-MSCs exert their therapeutic effects through multiple signalling pathways, including anti-ageing, Wnt and TGF-β. Cell components significantly regulate genes related to cell membranes and cell connections and downregulate the expression of macrophage- and neutrophil-related transcription genes (e.g., CH25H, Retnlg). Among them, CH25H produced by macrophages can change the cholesterol level in the plasma membrane using autocrine and paracrine and enhance lipid-laden macrophages. Cellular inflammatory response changes the signal expression of Toll-like receptor 4, promotes the expression of pro-inflammatory genes mediated by nuclear factor-κB and aggravates cell apoptosis 46 . Other studies have shown that hUC-MSCs have a more obvious therapeutic effect on asthma with refractory neutrophilia. Therefore, the therapeutic and maintenance effects of hUC-MSCs in food-allergic mouse enteritis may be through a variety of factors, including TGF-β. It is realised through relevant signalling pathways, including anti-apoptosis, promoting cell membrane integrity and cell-linking function, which still needs to be further confirmed by relevant research.
While our study demonstrates the beneficial effects of hUC-MSCs in reducing allergic symptoms in an OVA-induced FA model, the precise mechanisms through which these cells exert their immunomodulatory effects remain to be fully elucidated. It is hypothesised that hUC-MSCs may influence immune regulation by secreting anti-inflammatory cytokines, such as TGF-β and IL-10, which promote the expansion of Tregs and help modulate the Th2 immune response typically associated with allergic reactions. Furthermore, hUC-MSCs may affect mast cell activation and degranulation through paracrine signalling, potentially reducing histamine levels and thereby mitigating allergic symptoms. In this experiment, the OVA dose administered via intraperitoneal injection was sufficient to induce sensitisation, and tape stripping further enhanced the allergic response. Therefore, no food allergens were applied after the skin stripping. However, the exact signalling pathways and cellular interactions, such as the role of MSC-secreted exosomes or direct cell-to-cell contact with immune cells, require further investigation.
In addition to our focus on hUC-MSCs, recent studies have highlighted the advantages of placental-derived MSCs, including their greater yield, enhanced differentiation potential and broad immunomodulatory capabilities, which make them suitable for a wider ran ge of disease applications. Some studies have shown that placental-derived MSCs yield a higher number of cells with greater proliferation and self-renewal capacities compared to hUC-MSCs47,48. Furthermore, these cells exhibit reduced immunogenicity and stronger anti-inflammatory effects, making them ideal candidates for both autologous and allogeneic transplantation with a lower risk of immune rejection49,50. Placental tissue is easily accessible as medical waste without requiring invasive procedures such as adipose tissue collection. Unlike embryonic stem cells, its use avoids ethical controversies. Both placenta-derived MSCs and hUC-MSCs can be obtained from this source. Due to its complexity, the placenta is conceptually divided into the foetal side (comprising the amnion, chorion and umbilical cord) and the maternal side (comprising the decidua) 51 . Predefining the specific region of the placenta for cell extraction is essential for processing.
In terms of microbial safety, the placenta has evolved robust mechanisms of microbial defence to restrict vertical transmission during pregnancy. The physical barriers of the villous surface prevent attachment and invasion by non-viral pathogens such as Listeria monocytogenes and Toxoplasma gondii, although some viruses can overcome these barriers and enter primary trophoblasts 49 . The placenta also employs distinct antimicrobial defence mechanisms 52 , further reducing potential infection risks. While processing placental MSCs may require specialised techniques due to tissue complexity, the placental tissue’s inherent antimicrobial defences and therapeutic potential are well-documented. Its physical and molecular barriers minimise infection risks and support the safe application of placenta-derived MSCs in transplantation therapies49,52. Numerous studies in recent years have highlighted the therapeutic potential of placenta-derived MSCs in treating various diseases53–55. Considering their strong anti-inflammatory and proliferative capacities, placenta-derived MSCs hold great promise as a viable option for transplantation therapies. Given these advantages, we are considering the potential use of placenta-derived MSCs for transplantation therapy in future studies.
Future studies focusing on the mechanistic pathways of MSC-mediated immune modulation in FA could provide a deeper understanding of their therapeutic potential. The current cell dosing may be higher than what has been clinically used, and the transition from basic research to clinical practice is a gradual process. It is possible that using lower passage cells, staging treatments, utilising exosomes or other derivatives, or even oral delivery methods may allow for effective outcomes while determining a more appropriate clinical dose. These factors will be considered in future studies to explore clinically suitable dosing strategies. In addition, the use of humanised mouse models is being considered for future studies, as they offer a closer approximation to human disease mechanisms. By incorporating human B and T cells, these models allow for the in vivo production of allergen-specific human IgE, which could enhance the relevance of our findings to clinical applications.
In summary, food-allergic enteritis is often accompanied by systemic immune response. The spleen, as an important immune defence organ involved in the body, also participates in the process of oral immune tolerance. Research on the treatment of hUC-MSCs transplanted from food to allergic enteritis is still in its infancy. Our study confirmed the efficacy and sustainability of the treatment, explored the possible involvement in the secretion of key factors TGF-β and IL-10 and explored the relevant mechanism of action of hUC-MSCs in terms of genes. It may play a very important role in maintaining cell membrane integrity and anti-apoptosis. Therefore, stem cells and their related derivatives, such as exosomes, conditioned media or combined stem cell strategies, are expected to alleviate acute and long-term food-allergic enteritis symptoms, promote tissue repair and reduce the inflammatory cascade. The relevant participating factors, mechanism of action, transplantation mode, dose and duration of action remain to be further studied.
Conclusion
Overall, our findings demonstrated that hUC-MSC therapy exerts therapeutic benefits through mediated modulation of TGF-β and IL-10 levels systemically, promoting Tregs proliferation while inhibiting mast cells. This promising approach holds the potential for alleviating FA with TGF-β and IL-10 influenced by hUC-MSCs serving as potential targets. Future studies should aim to elucidate the underlying mechanisms and explore more accessible transplantation methods, potentially utilising substances like exosomes or culture medium.
Supplemental Material
sj-jpg-1-cll-10.1177_09636897251326899 – Supplemental material for Therapeutic and continuative effects of human umbilical cord–derived mesenchymal stromal cells in food-allergic mice
Supplemental material, sj-jpg-1-cll-10.1177_09636897251326899 for Therapeutic and continuative effects of human umbilical cord–derived mesenchymal stromal cells in food-allergic mice by Yuan Zhao, Yabing Ding, Zhaoyan Wang, Qian Wang, Dou Ye and Zuo Luan in Cell Transplantation
Supplemental Material
sj-jpg-2-cll-10.1177_09636897251326899 – Supplemental material for Therapeutic and continuative effects of human umbilical cord–derived mesenchymal stromal cells in food-allergic mice
Supplemental material, sj-jpg-2-cll-10.1177_09636897251326899 for Therapeutic and continuative effects of human umbilical cord–derived mesenchymal stromal cells in food-allergic mice by Yuan Zhao, Yabing Ding, Zhaoyan Wang, Qian Wang, Dou Ye and Zuo Luan in Cell Transplantation
Supplemental Material
sj-jpg-3-cll-10.1177_09636897251326899 – Supplemental material for Therapeutic and continuative effects of human umbilical cord–derived mesenchymal stromal cells in food-allergic mice
Supplemental material, sj-jpg-3-cll-10.1177_09636897251326899 for Therapeutic and continuative effects of human umbilical cord–derived mesenchymal stromal cells in food-allergic mice by Yuan Zhao, Yabing Ding, Zhaoyan Wang, Qian Wang, Dou Ye and Zuo Luan in Cell Transplantation
Footnotes
Author contributions
YZ and ZL designed the study. YZ and DY performed experiments. YZ and YD analysed the data. ZW and QW confirmed the authenticity of all the raw data. All authors read and approved the final manuscript.
Ethics approval
This study was approved by the Animal Experiment Committee at Beijing Center for Physical & Chemical Analysis (No. 210520-SWDWF-005).
Statement of human and animal rights
This article does not contain any studies with human or animal subjects.
Statement of informed consent
There are no human subjects in this article and informed consent is not applicable.
Data and materials availability statement
The data exhibited in this study are available on request from the corresponding author.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
