Abstract
Pseudoangiomatous stromal hyperplasia (PASH) is a benign breast pathology, which most commonly presents incidentally along with other breast pathologies. The etiology and pathogenesis of PASH are still unknown; however, there is some evidence suggesting PASH is hormone dependent. The clinical history, presentation, and imaging appearance of PASH are variable. Clinically, PASH has a wide spectrum of presentations, from being silent to gigantomastia. On imaging, PASH demonstrates various benign to suspicious features. Here we summarize PASH’s clinical presentation, histopathology, imaging features, and management.
Introduction
Pseudoangiomatous stromal hyperplasia (PASH) is an uncommon benign entity that was first described in 1986 [1] and has a wide spectrum of presentations, ranging from a clinically silent lesion accompanied by other breast pathologies to a huge palpable mass [2].
PASH is most commonly clinically silent and diagnosed incidentally during pathological examination of another benign or malignant lesion [3]. Incidental microscopic PASH can be found in up to 23% of consecutive breast specimens [4]. In contrast, PASH as a main pathological finding is a rare entity.
The etiology and pathogenesis of PASH are still unknown. As PASH is mostly associated with premenopausal women and, in rare cases, males with gynecomastia or post-menopausal women on hormone therapy, it is believed that PASH is hormone dependent [5]. PASH has not been recognized as a high-risk lesion, premalignant, or a risk factor for breast cancer; however, because it is usually detected incidentally, it may associate with malignant lesions [5].
Clinically, PASH can be asymptomatic and incidentally detected accompanying other benign or malignant breast pathologies or present as a fast-growing palpable mass or gigantomastia [6–8]. On imaging, PASH presentation is also variable, with a wide spectrum of benign-appearing lesions to a suspicious appearing mass [9,10].
This study aims to review the available data on PASH with a focus on the radiological features and imaging approach of PASH. The mammography, ultrasound, and MRI features of PASH are discussed.
Histopathology
Gross presentation
PASH is commonly seen as a smooth, solid, oval, rubbery circumscribed but not encapsulated mass with homogeneous tan or gray-white fibrous structure and without extensive hemorrhage and necrosis [11].
Microscopy
PASH has an intermixed proliferation of stromal cells and collagen and normal to hyperplastic epithelial cells. Characteristically, PASH has slit-like channels lined with myofibroblasts (spindle cells) resembling vascular channels. The name pseudoangiomatous was derived from the fact that it mimics vasoformative proliferation, which is characteristic of low-grade angiosarcoma [5,11]. The spindle cells are positive for vimentin, CD34, BCL2, CD99, and α-smooth muscle actin but negative for CD31 and factor VIII, an endothelium-specific marker. Unlike angiosarcoma, PASH does not have erythrocytes, nuclear atypia, mitoses, or pleomorphism [11].
In addition, PASH cells, which are hormonally sensitive, frequently express progesterone receptors and estrogen receptors relatively less frequently [12–14].
PASH is categorized into simple and fascicular/proliferative types. Simple PASH has a discontinuous layer of flat or spindle-shaped myofibroblasts lining interconnecting, slit-like spaces, while the fascicular/proliferative type has multiple clusters of proliferating bland spindle cells surrounding the empty spaces [11,12].
There are a wide range of other stromal pathologies in the breast ranging from benign hyperplasia to malignant lesions, such as benign fibrous tumor, fibrous mastopathy, myofibroblastoma, metaplastic breast carcinomas, sarcomas, and phyllodes tumors. These lesions may present with clinical symptoms, such as a mass. To rule out malignancy, breast biopsy is generally required [13–17]. Epithelial structures, eosinophilic fibrous tissue expanses, and linear spindle cell nuclei arrangements result in a tiger-stripe-like appearance [18].
Mohamad Sakibuzzaman et al. reported a unique tiger-striped microscopy profile for PASH with scattered non-neoplastic breast epithelial elements, with stromal expansion between and within these [18].
Myofibroblastic stromal hyperplasia (MSH) has also been described [14], which overlaps with the fascicular pattern of PASH, but it does not demonstrate pseudovascular structures, and it predominantly involves perilobular stroma. Recognition of this pattern will avoid discordant radiologic pathologic findings and unnecessary surgery/repeat biopsies [14] (Fig. 1).
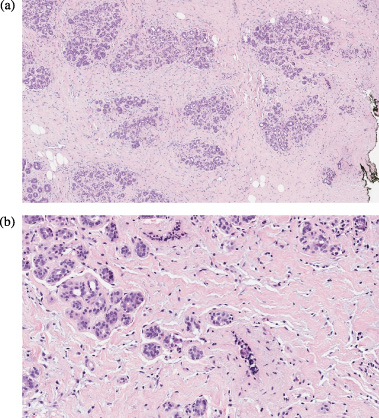
Pseudoangiomatous stromal hyperplasia (Hematoxylin & eosin stain, 4X and 20X) is a benign myofibroblastic proliferation with anastomosing slit-like spaces lined by myofibroblasts simulating blood vessels in dense collagenous stroma. No mitotic figures, necrosis or cytologic atypia is identified.
PASH in the breast is most commonly encountered as a clinically silent, incidentally detected lesion during the histologic evaluation of other benign or malignant lesions [19]. Other presentations of PASH can present as a palpable mass called nodular PASH or tumorous PASH [19,20]. Multifocal nodular or diffuse lesions with asymmetric enlargement of the breasts have also been reported on rare occasions [19,20]. PASH can also present with diffuse breast involvement and gigantomastia, even rarely involving both breasts and axillary regions simultaneously [16,21].
In nodular PASH, a well-circumscribed, firm, mobile mass without overlying skin change mimicking a benign fibroepithelial lesion is most commonly reported [15,17].
As already mentioned, PASH is most found in premenopausal women [5,22,23], and it can affect patients from ages 12 to 86 years [11]. Some other rare presentations of PASH also were reported, such as men with gynecomastia [15], pregnant women with bilateral PASH-related gigantomastia [8], acute onset gigantomastia [24], breast enlargement in the pediatric group [24–26], immunocompromised patients, including patients with HIV, neurofibromatosis type 1, and patients undergoing cyclosporine treatment [15].
PASH stromal cells express high-density progesterone receptors, while normal mammary stroma shows no progesterone receptor staining. Since progesterone is metabolized by cytochrome P450, the medications, which are metabolized by cytochrome P450, such as clonazepam, valproate, and risperidone, increase the level of progesterone that could stimulate PASH growth [10,27].
PASH has very rarely been reported in men [28]. The apparent link between PASH and hormonal stimulation highlights the unique nature of PASH in males. PASH can be seen in men exposed to estrogen and progesterone or who have known gynecomastia [28].
PASH has also been described in pediatric patients as young as 3 years old [29]. PASH has been recognized as a distinct entity in pediatric breast lesions and should be differentiated from other differentials such as low-grade angiosarcoma or fibroadenoma [29,30].
Imaging
According to the previous studies, PASH shows a non-specific, wide range of imaging characteristics [28,31]. The wide-ranging appearance of PASH on imaging supports the opinion that PASH is usually an incidental finding [28,31,32]. If any suspicious feature is appreciated in the imaging, tissue sampling is required because PASH can coexist with a malignant process [33].
Ultrasound
The sonographic appearance of PASH is also non-specific. The most common feature is a well-circumscribed hypoechoic or isoechoic oval mass enhanced through transmission, favoring a benign process and making further sampling unnecessary [26,33]. Simultaneous existence of fibrocystic changes can result heterogeneous appearance. Less common morphologies are echogenic area with hypoechoic central areas, mass with irregular or poorly defined borders, and mass with solid appearance and prominent internal cystic changes [21] (Fig. 2).
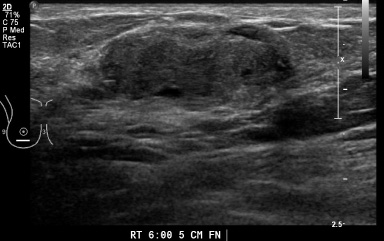
PASH in ultrasound. A palpable oval mass with heterogeneous echogenicity and some cystic changes.
It has been reported that almost 53% of patients with PASH present with an abnormality on screening mammography [2]. PASH may be mammographically occult, even when it represents the predominant histopathologic diagnosis [33]. The most common presentation of PASH in mammography is mass of variable size, usually round or oval, with circumscribed or partially circumscribed margins [32]. The second most common mammographic presentation of PASH is a focal asymmetry [2,31,33]. Other less common mammographic features of PASH are calcification or architectural distortion [32].
For the less common but suspicious mammographic features including architectural distortion, suspicious calcification, and spiculated mass, a biopsy is required to rule out cancer [33] (Fig. 3).
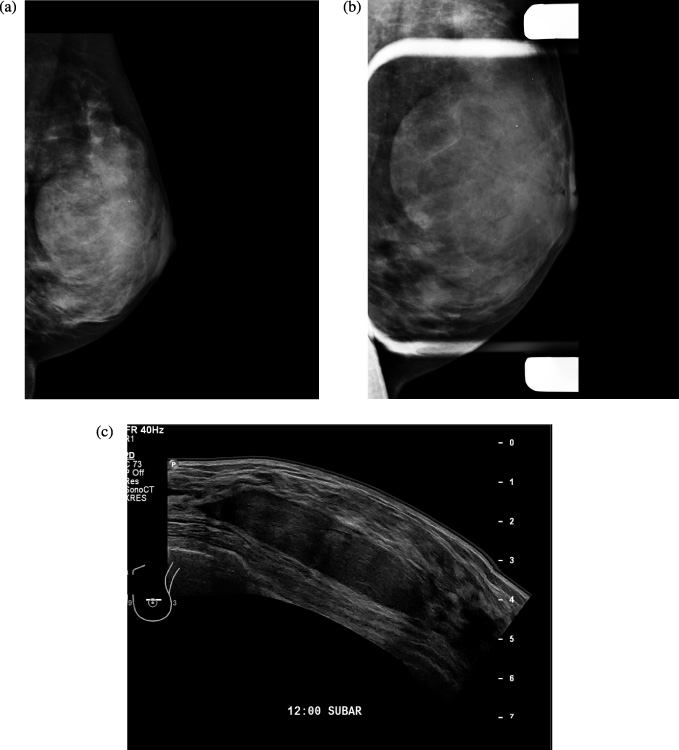
A large partially obscured oval isodense mass in mammogram, corresponding to a hypoechoic circumscribed mass in ultrasound.
There are few studies on the MRI appearance of PASH [21,27,31]. In the study by Emily Nia, the MRI characteristics of the two groups of patients with ≤50% PASH in pathology and the group with ≥51% PASH did not differ significantly [31]. All types of mass, non-mass enchantment and kinetics were seen for PASH [31] (Fig. 4), but a progressive enhancement, Type 1 kinetic, is the commonest type [2]. On T2-weighted and short tau inversion recovery (STIR) images, high-signal slit-like spaces may be seen, which correspond to empty clefts within acellular hyalinized stroma on histopathology [2]. The appearance of clumped non-mass enhancement with persistent kinetics is a common feature of PASH on MRI [31]. In the study by Alikhassi, a rare case presented with bilateral enlarging breasts, which demonstrated low signal on T1 and a high signal on T2, with masses with irregular and microlobulated margins. All three types of enhancing curves were plotted. Core biopsy showed PASH [21] .
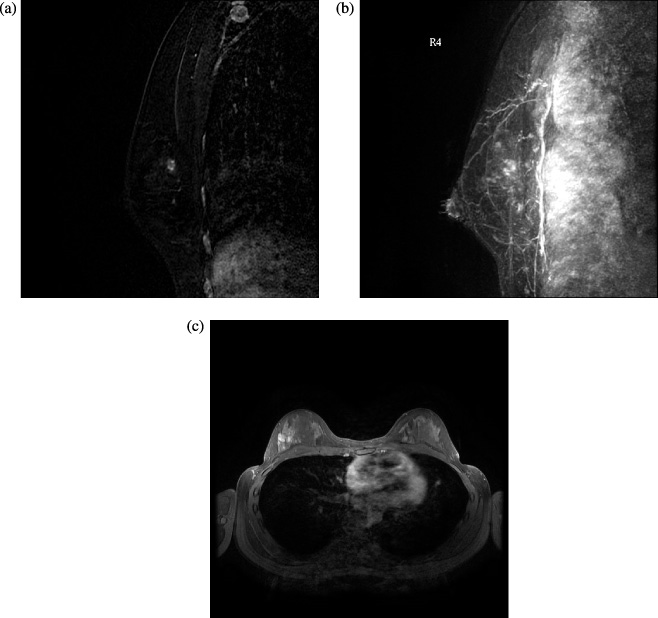
PASH in MRI. A developing deeply located focal non mass enhancement in the screening MRI of a high risk woman. No ultrasound or mammogram correlated lesion was detected. MRI guided biopsy showed dominant pathology of PASH.
There is no standard management for PASH [3,18]. Typical approaches are watch and wait, periodic surveillance, hormone therapy, surgical excision, and in rare cases, mastectomy [33–37].
The type of treatment is chosen based on the characteristics of the individual case. No additional interventions are required in cases of incidentally detected PASH, with concordant radiology and pathology biopsies [18,38].
Surgical excision may be considered for tumorous or nodular PASH, rapidly increased lumps, or in women at an increased risk for developing breast cancer [2,26,38]. There have been reported cases of diffuse PASH with gynecomastia with persistent pain, which required wide excision or mastectomy [21,37,39]. Incompletely excised PASH has a recurrence rate of up to 22%, probably due to the persistence of a residual mass after surgery [18,26].
Few studies have reported anti-hormonal therapy’s possible role in managing breast PASH. But there is no sufficient data on the efficacy of this type of medical management [36,37,40].
In the future, additional works are required to provide a standard guideline for appropriate management for women with PASH.
Prognosis
Based on the available literature, PASH has an excellent prognosis, and no PASH-related deaths have been reported [21,27,40].
Degnim et al. in Annals of Surgical Oncology in 2010 reported that the subjects with PASH in their cohort study were significantly less likely to develop breast malignancy than those without. On the other hand, they found a striking difference in the laterality of subsequent breast cancers between women with and without PASH. In 84.6% of the women with PASH and breast cancers diagnosed more than 5 years after biopsy, the cancers were in the ipsilateral breast, compared with 50.3% in the ipsilateral breast for women whose benign biopsies did not show PASH (P = 0.001). These authors suggested that the presence of PASH may interfere somehow with a permissive stromal environment facilitating breast carcinogenesis locally [41]. This is worth investigating as a research question in prospective trials in future, considering the limited studies on the topic.
Further studies are required to evaluate the long-term clinical outcome and natural course of PASH.
