Abstract
BACKGROUND:
The normal imaging appearances of the common agents used in injection mammoplasty and the challenges of mammography screening will be reviewed.
METHODS:
The local database from a tertiary hospital was accessed for imaging cases of injection mammoplasty.
RESULTS:
Free silicone is seen as multiple high-density opacities on mammograms. Silicone deposits can often be seen within axillary nodes due to lymphatic migration. Sonographically, a snowstorm appearance is seen when the silicone is diffusely distributed. On MRI, free silicone is hypointense on T1-weighted and hyperintense on T2-weighted images, with no contrast enhancement. Mammograms have a limited role in screening due to the high density of silicone. MRI is often required in these patients.
Polyacrylamide gel and hyaluronic acid are seen as multiple collections on mammography. Polyacrylamide gel collections are of the same density as cysts, while hyaluronic acid collections are of higher density but less dense than silicone. On ultrasound, both can appear anechoic or show variable internal echoes. MRI demonstrates fluid signal with hypointense T1-weighted and hyperintense T2-weighted signal. Mammographic screening is possible if the injected material is located predominantly in the retro-glandular space without obscuring the breast parenchyma.
On mammograms, autologous fat locules appear as lucent masses. Rim calcification can be seen if fat necrosis had developed. On ultrasound, focal fat collections can demonstrate varying levels of internal echogenicity, depending on the stage of fat necrosis. Mammographic screening is usually possible for patients after autologous fat injection as fat is hypodense compared to breast parenchyma. However, the dystrophic calcification associated with fat necrosis may mimic abnormal breast calcification. In such cases, MRI can be utilized as a problem-solving tool.
CONCLUSION:
It is important for the radiologist to recognize the type of injected material on the various imaging modalities and recommend the best modality for screening.
Introduction
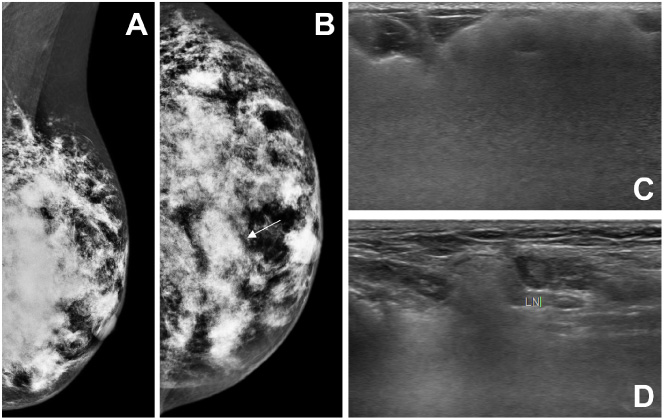
Mammography and ultrasound images of a 72-year-old woman with free silicone injected in both breasts. Mediolateral oblique (A) and craniocaudal (B) mammographic views of the left breast show multiple dense opacities, with an example in (B) demarcated by a white arrow. On ultrasound, free silicone is seen as echogenic area within the breast with loss of detail posteriorly, known as “snowstorm” appearance (C). Ultrasound of the left axilla demonstrates silicone deposits in a left axillary node due to lymphatic migration (D).
Injection mammoplasty has been utilized as an alternative to surgical breast implantation or as a procedure to achieve even breast contours. Compared to surgical breast augmentation, some of its advantages include this being an outpatient clinic procedure, instant cosmetic results, and a quick recovery period [1]. However, injection mammoplasty can be affected by short term complications and may lead to difficulties in future mammographic screening [2]. Its long-term complications and risks are largely unknown. In this article, we will review the normal imaging appearances of the common agents used in injection mammoplasty – free silicone, polyacrylamide gel, hyaluronic acid, and autologous fat. Their associated complications and the challenges of subsequent mammography screening will also be discussed. Liquid paraffin injection will not be discussed in the article as the use of this agent for breast augmentation had been discontinued in the mid-1900s due to short- and long-term complications [1,3].
Silicone, a polymer derived from the element silicon, was used as an injection mammoplasty material for breast augmentation decades ago in countries worldwide. In the United States, it was used in the 1950’s and 1960’s but has since been abandoned due to safety concerns and replaced by surgically implanted breast prostheses [4]. However, it continues to be performed in some countries in South America and Asia [1,2].
The mammographic hallmark of free silicone in the breast is multiple dense opacities of varying sizes which may be associated with calcifications [1] (Fig. 1A,B). Silicone may also be seen as a single conglomerate dense mass within the breast [5]. Sonographic appearance of free silicone is variable, ranging from anechoic cysts to hypoechoic nodules or masses. The nodules typically show an echogenic anterior margin with posterior acoustic shadowing. When more diffusely present, free silicone is seen as a highly echogenic area with loss of detail posteriorly (Fig. 1C), also known as a “snowstorm” appearance [5].
The magnetic resonance imaging (MRI) sequences typically used for evaluating the integrity of silicone implants can be applied to patients with silicone injection mammoplasty. Silicone appears as round masses or collections without borders which are hypointense on T1-weighted (Fig. 2A) and hyperintense on T2 weighted images. Free silicone is best seen as hyperintense areas on the silicone-selective sequence that utilizes inversion recovery technique with water- and fat-suppression (Fig. 2B). These same areas demonstrate corresponding signal loss on a T2-weighted sequence with silicone suppression (Fig. 2C). Hyperintense signal silicone can be differentiated from intermediate signal native parenchymal tissues on T2-weighted sequences. On the dynamic post-contrast T1-weighted sequences, silicone does not enhance, as opposed to native breast parenchyma which demonstrates progressive enhancement (Fig. 2D).
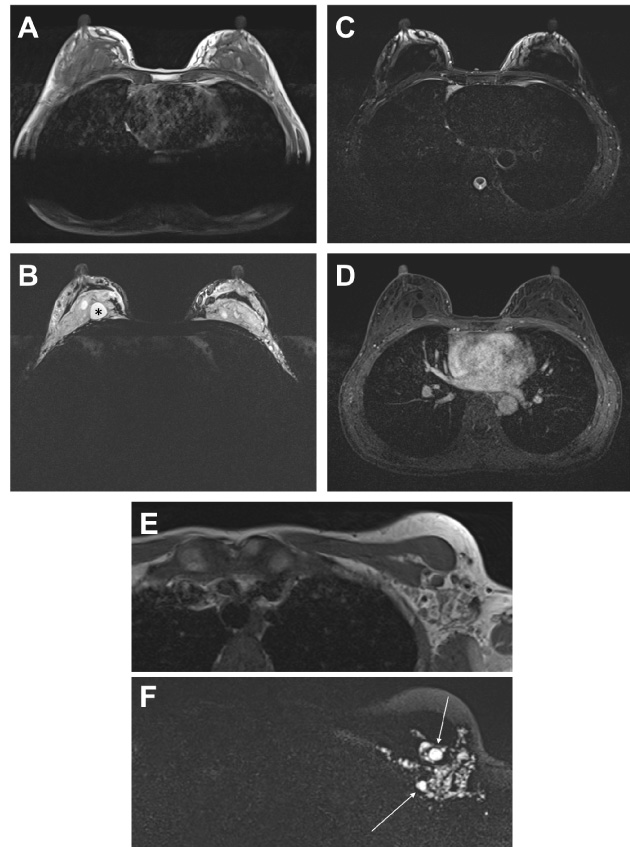
Magnetic resonance imaging of a 49-year-old woman with free silicone injected in both breasts. Axial T1-weighted sequence (A) shows multiple hypointense silicone collections in both breasts. Axial silicone-selective sequence with water- and fat-suppression (B) demonstrates hyperintense collections representing silicone (an example is marked with an asterisk). Axial silicone-suppressed sequence (C) demonstrates signal suppression from the silicone deposits. Post-contrast axial T1 fat-suppressed sequence (D) shows no enhancement of the silicone deposits, with mild background parenchymal enhancement noted. Axial T1 (E) and silicone-selective (F) images at the same level shows silicone extension to the left axillary region with focal deposits within axillary lymph nodes (white arrows in F).
Silicone deposits can often be seen within axillary nodes due to lymphatic migration of silicone [2] (Figs 1D, 2E,F). Silicone can also migrate to the chest wall, pleural cavity, abdomen, and upper limbs. The various MRI sequences allow for the delineation of the free silicone within the breasts and the evaluation of native breast parenchyma for malignancy. Free silicone gradually causes foreign body granulomatous reactions which can become very prominent. This results in marked granuloma formation and fibrosis, giving rise to an irregular enhancing mass with architectural distortion which can mimic a malignant lesion [6]. Biopsy of these lesions is usually necessary to distinguish them from malignancy. In some patients, granulomatous reaction and extension of silicone into the dermis of the breast may result in skin inflammation, necrosis and fistula formation [7]. Although a meta-analysis showed no association between silicone breast implants and the risk of connective tissue disease [8], there is still some controversy regarding the association of connective tissue disease and the presence of silicone within the breasts.
The foreign body granulomatous reaction from free silicone results in hard nodular masses within the breasts. This limits the role of physical examination in the detection of breast cancer [2]. Conventional mammographic screening and breast ultrasound are also extremely limited due to obscuration of the breast parenchyma. For women who had received free silicone injection to the breasts, screening MRI is recommended by the American College of Radiology [9].
PAAG consists of 2.5–5% of polyacrylamide, formed from the polymerization of acrylamide monomers, suspended in 95–97.5% water. It is also marketed under different names such as Aqualift TM and Aquamid TM [10]. PAAG has been used as a filler for facial contouring and injection mammoplasty in China, countries of the former Soviet Union, Eastern Europe, and parts of Asia. PAAG injection mammoplasty is performed under local anesthesia through a small incision, most commonly at the inframammary crease, where a variable amount of PAAG is injected into the retroglandular space.
As PAAG is similar to water in density, imaging shows fluid collections within the breasts, usually located in the retroglandular space [11]. The size and location of the fluid collections will depend on the way the material was injected. On mammogram, water-density collections similar to the density of cysts are typically located in the retroglandular space, with or without defined margins. They may appear to blend into the adjacent parenchyma as they do not have a true capsule [2] (Fig. 3A). On sonography, PAAG appears as a fluid collection in the retroglandular pre-pectoral location which may be anechoic or contain variable internal echoes (Fig. 3B). MRI signal of a PAAG fluid collection demonstrates typical fluid signal with hypointense T1-weighted and hyperintense T2-weighted signal intensities (Fig. 3C). There may be discrete T2 hypointense foci within which correspond to echogenic foci seen on sonography; these may represent debris, air bubbles or impurities. An uncomplicated PAAG collection may show delayed thin rim enhancement.
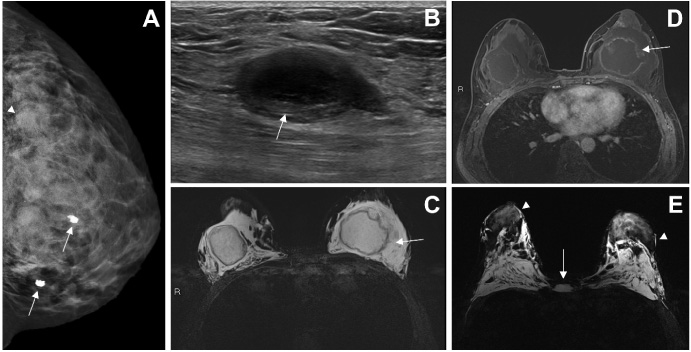
Mammogram, ultrasound, and magnetic resonance imaging (MRI) images of two patients with polyacrylamide gel (PAAG) injected in both breasts. Images (A)–(D) are from a 43-year-old woman. Image (E) is from a 61-year-old woman. (A) Craniocaudal mammographic view of the left breast showing diffuse water density in the retroglandular space without defined margins (white arrowhead). Incidental note of two foci of coarse popcorn calcification representing fibroadenomas (white arrows). (B) Ultrasound shows an anechoic collection of PAAG containing internal echoes (white arrow). (C) Axial turbo inversion recovery magnitude MRI image showing hyperintense signal intensity of extravasated PAAG (white arrow) in both breasts. (D) Post-contrast axial T1 fat-suppressed MRI sequence shows extravasated PAAG and rim enhancing PAAG collections (white arrow) within central region of both breasts. (E) Axial turbo inversion recovery magnitude MRI image shows extensive collections of fluid signal present in bilateral pectoral muscles, retropectoral and retroglandular regions, representing extravasated PAAG. PAAG collections are also seen in the parenchymal and subcutaneous (white arrowheads) tissues of both breasts, as well as in the midline subcutaneous tissue anterior to the sternum (white arrow).
Complications following PAAG injection mammoplasty usually occur a few years after the procedure and include gel migration or extravasation (Fig. 3C,D), leading to unsatisfactory cosmesis, breast pain or lumps. The most commonly encountered complication is the formation of breast lumps [11,12]. As PAAG is injected blindly, the outcome can be unpredictable. Intra-glandular injection of PAAG may result in multiple PAAG collections within the breast parenchyma. PAAG may also be injected erroneously into the pectoralis muscle (Fig. 3E). If injected into the subcutaneous plane, PAAG can spread to form subcutaneous nodules in the inframammary fold, axilla, sternal and infra-clavicular region [13]. Calcifications from foreign body reaction can occur years after PAAG injection mammoplasty [14], which can confound mammographic screening [13].
Secondary infection is a complication that has been reported to occur in association with lactation [11,15], resulting in breast pain and swelling. The affected PAAG collection significantly increases in volume and demonstrates diffusely increased internal echoes on ultrasound.
Women with prior PAAG injection mammoplasty may be eligible for mammographic breast screening if the PAAG is confined to the retroglandular space with minimal obscuration of the breast parenchyma. In these patients, “pinched” views, similar to those performed for patients with surgically placed breast prosthesis, make mammographic screening possible.
Hyaluronic acid (HA) is a polysaccharide that is present in all mammalian tissues. Macrolane TM is a gel-like substance derived from HA. It was approved for breast enhancement from 2008–2012 [16]. As it is naturally degraded and resorbed over 12–18 months, it was marketed as a bio-compatible non-permanent injectable filler for volume restoration and contouring of the breasts. Up to 100 ml of Macrolane TM is injected in either a single pass or multiple passes into the retroglandular space. The breast is then massaged to attain the desirable contour. However, controversies regarding its side effects led to its withdrawal from the breast augmentation market world-wide in 2012 [17]. Some studies have shown that Macrolane TM was still visible on imaging at 24 months [18]. Therefore, the clinical and radiological sequelae may persist for an unknown period of time.
On mammography, HA injection mammoplasty results in single or multiple collections of fluid density, the size and distribution of which depends on the injection technique. The collections are usually of a higher density than simple cysts, but less dense than silicone injections (Fig. 4A). When injected into the pectoralis muscle, a hyperdense bulging convexity of the muscle can be seen on the craniocaudal and mediolateral views.
Mammogram and ultrasound images of a 39-year-old woman with hyaluronic acid injected in both breasts. (A) Mediolateral oblique mammographic view of the right breast showing several opacities representing collections of hyaluronic acid. An example is demarcated by a white arrow. These are of higher density than cysts. (B) Ultrasound showing a hyaluronic acid collection within the breast containing low level internal echoes.
HA is seen as multiple fluid collections of different sizes within the breast and sometimes within the pectoralis muscle on ultrasound. They are usually anechoic but may contain internal echoes of varying echogenicities (Fig. 4B). Due to the way that HA is injected, the fluid collections may be found in disparate locations within the breast or the pectoralis muscle, which may be associated with inter-communicating channels. HA collections may show a more solid appearance over time [19]. Ultrasound can be used to guide aspiration in symptomatic patients who may have a lump, breast hardness or pain. However, due to the thick consistency of HA, aspiration often yields scanty material, leaving most of the collection behind. Residual HA collections may persist and cause anxiety to the patient [19].
The complications associated with HA are similar to those associated with PAAG. Migration of these collections lead to unsatisfactory contour, breast lumps or pain. Secondary infection can also occur, although these have not been reported to be related to lactation unlike PAAG [20]. Capsule formation with calcifications may form around HA collections [19].
Screening mammography may be possible for patients with prior HA injection mammoplasty if the injected material is located predominantly in the retroglandular space with no obscuration of the breast parenchyma. Calcifications which form in the capsule around these collections may mimic abnormal breast calcifications. These require comparison with the prior mammographic films, sonographic correlation with the location of HA collections and follow up. There are concerns of the association of HA with breast cancer and the long-term effects are unknown [21].
Native fat is the ideal material for breast augmentation as there is no risk of hypersensitivity or foreign body reaction [1]. Fat is removed from the abdominal or upper thigh region by liposuction, centrifuged and then injected in multiple aliquots into the breast. This technique has been used to augment the breasts, as well as to contour the breasts after surgery [22].
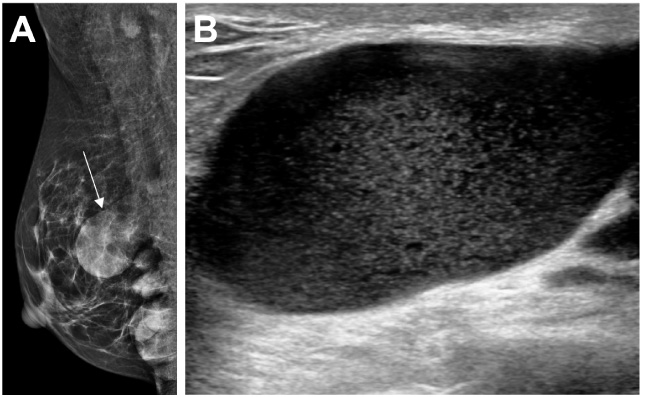
On mammogram, the fat locules appear as varying number of lucent masses depending on the injection technique [23]. On ultrasound, the injected fat initially appear as areas of increased echogenicity with no defined borders. Due to variable resorption, the injected fat sometimes do not become vascularized and incorporated into the breast tissues. The fat starts to aggregate in focal fat collections and eventually fat necrosis occurs. The lucent masses with fat necrosis start to develop rim calcifications that are well-demonstrated on mammography (Fig. 5A,B). Sonographically, focal fat collections appear as multiple cysts. These can be anechoic or demonstrate varying levels of internal echogenicity, depending on the stage of fat necrosis (Fig. 5C,D). If rim calcifications have developed, there can be an echogenic rim with posterior shadowing.
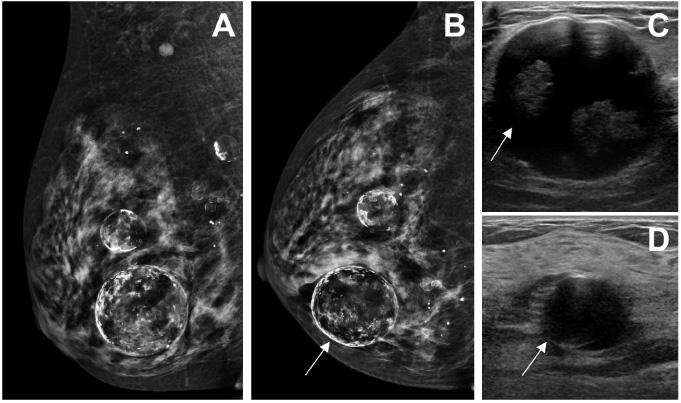
Mammogram and ultrasound images of a 43-year-old woman with injections of autologous fat in both breasts. Mediolateral oblique (A) and craniocaudal (B) views of the right breast showing round lucent masses with rim calcifications compatible with fat necrosis, with an example in (B) demarcated by a white arrow. (C) Ultrasound demonstrates a focal fat collection as round cystic mass with internal echoes (white arrow). (D) Ultrasound shows fat collection as anechoic cyst (white arrow).
Late fat necrosis is associated with increased fibrosis and is a mimicker of malignancy. This can present as a spiculated density on mammogram, or an ill-defined mass on ultrasound with posterior acoustic shadowing. The appearance on MRI depends on the stage of fat necrosis [2]. Early fat necrosis tends to show enhancement whereas late fat necrosis demonstrates fibrosis and spiculation. The presence of fat in the central aspect of the mass, which is hyperintense on T1-weighted and T2-weighted sequences with signal suppression on the fat-suppressed sequences, is characteristic of fat necrosis.
Mammographic screening may be possible for patients after autologous fat injection mammoplasty as fat is of low density compared to the breast parenchyma. However, the dystrophic calcification associated with fat necrosis may mimic abnormal breast calcification [24]. This is usually not a problem with adequate clinical history and serial follow-up studies. The identification of a lucent fat-containing area in the central aspect of the opacity on mammogram, or fat signal within the mass on MRI, is helpful in its diagnosis. Ultimately, biopsy may be required for histological analysis if malignancy cannot be excluded despite complete imaging work-up.
Patients with prior injection mammoplasty may present to the radiologist. It is important for the radiologist to recognize the type of injected material on conventional mammogram and ultrasound, as well as to recognize the associated complications of the different types of material. The radiologist should at the same time determine if the patient is suitable for mammographic screening in future and make recommendations accordingly. Patients who had undergone injection mammoplasty with PAAG, HA or autologous fat may be eligible for mammographic screening, which is usually not possible for patients who had undergone free silicone injections. In cases whereby mammographic screening is possible, comparison with prior mammograms is essential. If mammography is deemed difficult or challenging, screening with MRI may be considered.
Footnotes
Acknowledgements
None.
Author contributions
Conception: YS Lee, SY Teo
Interpretation or analysis of data: WXT Goh, YS Lee, SY Teo
Preparation of the manuscript: WXT Goh, YS Lee, SY Teo
Revision for important intellectual content: WXT Goh, YS Lee, SY Teo
Supervision: YS Lee, SY Teo
Conflict of interest
The authors declare that they have no conflict of interest.
