Abstract
BACKGROUND:
Breast cancer (BC) is the most prevalent cancer in women, with increasing incidence and death rates in recent years. Disruptions of different signaling pathways partially cause breast cancer. Hence, different genes through particular pathways are involved in BC tumorigenesis.
METHODS:
In this study, we evaluated the expression level of GLIS2 and CCND1 genes in 50 patients. Also, in-silico analyses were used to enrich related signaling pathways involving the mentioned genes.
RESULTS:
The results showed an increased expression level of Cyclin D1 and decreased expression level of GLIS2 in BC patients. Moreover, a relationship between aberrant expression levels of GLIS2 and CCND1 and BC development was determined.
CONCLUSION:
These observations could help uncover new therapeutic targets for treating patients with BC in the progressive stage.
Keywords
Introduction
Breast cancer (BC) is the most common type of cancer, accounting for about one-third of malignancies diagnoses among women and the fifth cause of cancer-related deaths worldwide [1,2]. BC’s incidence and death rates have increased over the last three decades, and studies estimated 2.7 million new cases and 0.87 million deaths in 2030 [1]. The 5-year survival in developed countries is 89.6% and 75.4% for localized and regional breast cancer, respectively [3]. Based on World Health Organization (WHO) classification, at least 18 different histological BC types were distinguished [4]. BC is the most common cancer in iranian women with agestandardized of 33.21 and 14.2 per 100,000 for incidence and mortality, respectively [5].
Aside from histological subtypes, BC can be divided into four molecular subtypes based on mRNA gene expression levels, including luminal A, luminal B, human epidermal growth factor receptor 2 (HER2)-enriched, and basal-like [6]. Several risk factors play pivotal roles in the occurrence of BC, such as gender (female), hormonal replacement therapy, older age, family history (of breast or ovarian cancer), overweight/obesity, and smoking [1]. Despite progression in diagnosis and treatment strategies, metastatic BC types remain challenging due to numerous factors, including molecular heterogeneity, intrinsic resistance to therapy, genetic mutations, and intracellular signaling pathways alternation.
During tumorigenesis, various signaling pathways are involved in which any alteration could support tumor progression and malignancy [7]. One of the important pathways involved in the development of the mammary gland is Wnt/β-catenin signaling, and its oncogenic role is determined in more than 60% of BC cases [8,9]. Among the Wnt/β-catenin pathway repressor genes, the GLIS2 has been well-known as belonging to the GLIS family and sub-family of Krüppel-like zinc finger proteins. GLIS2 is essential in regulating cellular processes and pathologies of oncogenesis [10–12]. Based on the evidence, GLIS2 is associated with many cancers, including colorectal cancer [13]. It is argued that GLIS2 activation can affect the expression of trophoblast cell surface antigen 2 (TROP2), which may stimulate AP-1, NF-κB, and cyclin D1 expression (CCND1) [14]. GLIS2 inhibits the expression of CCND1 via β-catenin/T-cell factor (TCF). CyclinD1 is one of the cell cycle’s primary regulators, particularly in the G1 to S transition stage. Thus, in BC, CCND1 gene can show oncogenic potential triggered by aberrant accumulation of β-catenin, resulting in uncontrolled cell proliferation and tumorigenesis [15]. Another important pathway is the Hedgehog (Hh) signaling. Hh signaling can induce progression, metastasis, chemo-resistance and prepare the tumor cells for the epithelial-mesenchymal transition (EMT), particularly in aggressive subgroups of BC. Hh signaling contributes to BC development by interacting with other main signaling pathways and pro-oncogenic factors [16].
Many studies underline the importance of tightly controlled Hh pathway and Wnt/β-catenin signaling activation in mammary development and tumorigenesis [17,18]. However, the regulatory genes and potential role of Hh signaling and the Wnt/β-catenin pathway in BC are poorly understood.
Based on studies, it can be supposed alteration of GLIS2 and CCND1 expression levels and their possible interaction may play a role in BC initiation and progression. So, it is fundamental to clarify the role of GLIS2 and CCND1 in BC and the exact interaction with BC-related pathways to understand the precise mechanism of cancer progression. In this study, we evaluated the expression levels of GLIS2 and CCND1 and their possible association with clinical features in BC patients. Furthermore, in combination with in silico analysis, this study examined the possible roles of the genes above during BC development.
Materials and methods
In silico studies: Selection of CCND1 and GLIS2 and molecular signaling pathway enrichment analysis
Using DisGeNET database [19], genes related to neoplasm (CUI: C0027651), malignant neoplasm (CUI: C0006826), carcinogenesis (CUI: C0596263), and malignant neoplasm of breast (CUI: C0006142) were obtained. Subsequently, all related genes were collected and inputted into the VENNY diagram 2.1 (http://bioinformatics.psb.ugent.be) to gain overlapped genes. Also, EnrichR database [20] was used to understand possible pathways in wich the common genes could be involved.
Patients
Healthy and cancerous tissues were collected from 50 patients who were referred to the Al-Zahra Hospital and Sadouqi Hospital, Isfahan, Iran during the period August, 2021 to March, 2022. After obtaining signed informed consent, samples were collected during surgery. Normal and tumor tissues were separated, and expert pathologists determined the grade of tumor tissues. The samples were also evaluated for the presence of ER, PR, and HER2 receptors using immunohistochemical (IHC) analysis. The procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (Isfahan University of Medical Sciences, Isfahan, Iran) and with the Helsinki Declaration of 1975, as revised in 2000.
Real time primer sequences
Real time primer sequences
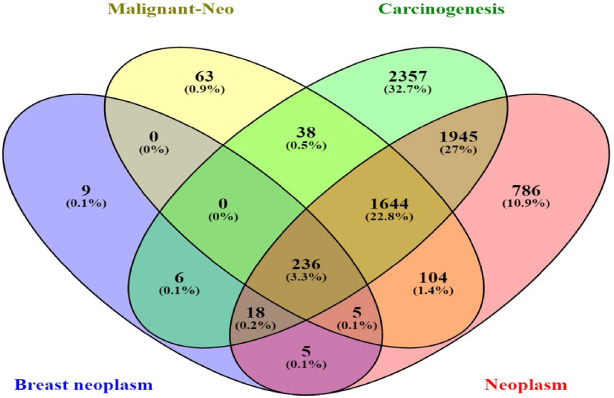
Authentication common genes among neoplasm, malignant neoplasm, carcinogenesis, and breast neoplasm. As it is shown, 236 genes are common among the four groups, and are selected for the next in-silico analysis.
Total RNA extraction was performed for all samples using the RiboEx kit (GeneAll, Seoul, South Korea), according to the manufacturer’s protocol. The quantity and integrity of the extracted RNA were measured by a NanoDrop spectrometer (Thermo Scientific, USA). cDNA synthesis was carried out using the RevertAid First Strand cDNA Synthesis kit (Fermentas, Waltham, Massachusetts, USA). cDNA synthesis was fulfilled by gradient thermocycler PCR (BIO-RAD, Germany). The reaction was carried out at 25 °C for 5 min, 42 °C for 60 min and 70 °C for 5 min.
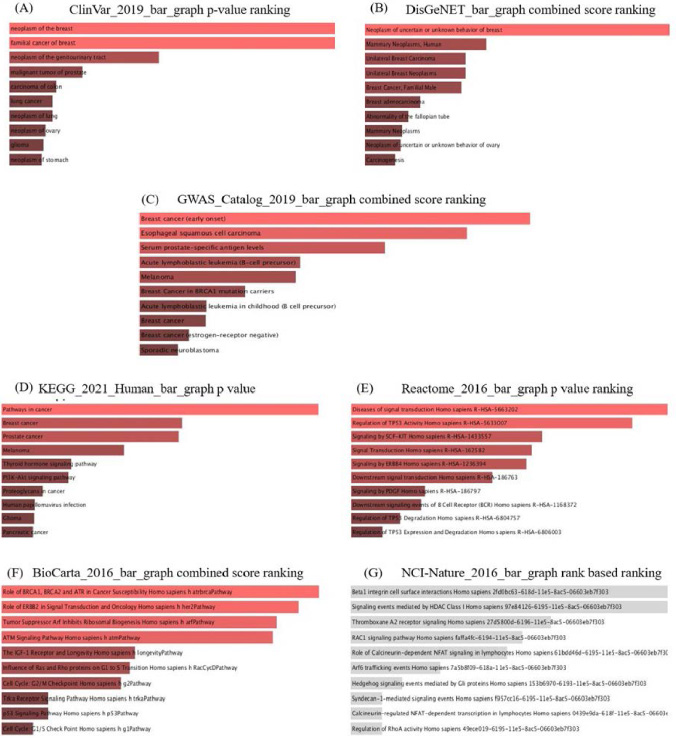
(A–G). Enrichment and data mining of overlapped genes among Neoplasm, Malignant neoplasm, Carcinogenesis, and Breast Neoplasm indicated that these genes are involved in the pathogenesis of breast cancer based on evidence and literature.
Specific primers were designed using AlleleID V7.7 software (PREMIER Biosoft, Palo Alto CA, USA) and ordered to the Macrogen company (Seoul, South Korea) (Table 1). qPCR was performed using SYBR Green Master Mix (Amplicon, Odense, Denmark) in triplicate. The reaction cycling included 94 °C for 5 min as the initial denaturation step, 35 cycles of 94 °C for 1 min, 60 °C for 45 sec, and 72 °C for 45 sec, followed by one cycle of 72 °C for 7 min as the final extension. The relative expression levels of each mRNA were calculated using the 2−ΔΔCt method relative to GAPDH (as a housekeeping gene).
Statistical analysis
Statistical analysis was performed By SPSS V.22 software (IBM, New York, USA), and a paired parametric T-test was used to analyze expression levels between groups. Pearson correlation analysis was applied to examine the linear correlation between gene expression and biological features, including grade, stage, ER, PR, and HER2 positiveness, taking steroid drugs, and lymph node involvement. Results were presented as mean ± SEM (standard error of the mean). P ≤ 0.05 was set as statistically significant. Finally, all data were entered to an excel sheet and Graphs were depicted by Graph Pad Prism statistical software, V.6.07.
Results
Gene selection and pathway analysis
The 236 overlapped genes from the gene list associated with neoplasm, malignant neoplasm, carcinogenesis, and breast neoplasm were obtained (Fig. 1). Among these genes, we searched for those previously validated in other cancers. Finally, CCND1 and GLIS2 were selected to be examined in the current study.
Enrichment analysis indicated the desired genes are involved in vital signaling pathways in neoplasm and malignancy, including P53 signaling, Wnt and Rap1 signaling, NOTCH, mTOR, PI3k-Akt, estrogen, and Hedgehog pathways (Fig. 2A–G). The direct or indirect correlation between CCND1 and GLIS2 and enriched pathways via other genes is illustrated in Fig. 3.
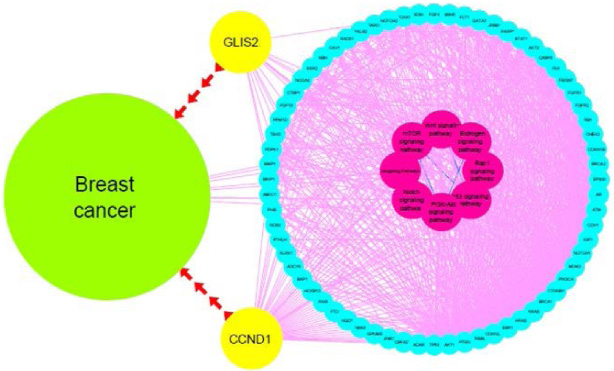
Cytoscape picture of the protein-protein interaction network of GLIS2 and CCND1 and their related signaling pathways.
According to pathological observations, 20%, 50%, and 30% of the tumor tissues have been classified as grades I, II, and III, respectively. The mean age of subjects was 48.13 ± 11.88. Also, lymph node involvement was observed in 70% of the studied samples. The clinical and general features of patients are summarized in Table 2.
Characteristics of patients
Characteristics of patients
The relative expression level of the GLIS2 gene was significantly decreased (P ≤ 0.002) by an average of six folds in tumor tissues relative to controls (Fig. 4A).
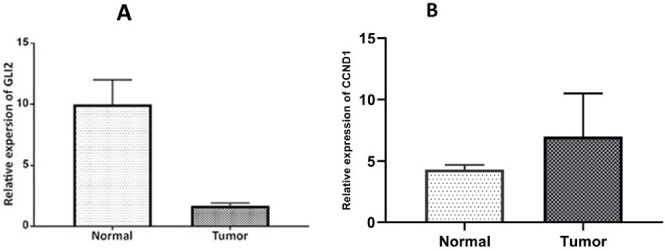
The relative expression of GLIS2 gene (A) and CCND1 (B) in tumor tissues compared to healthy. GAPDH was utilized for an endogenous reference to standardize mRNA expression levels. The error bars represent the standard error of the mean (SEM).
We found that in tumor samples with negative ER and PR receptors, the GLIS2 expression level was significantly reduced (Foldchange: −2.95, P ≤ 0.008). However, decreased GLIS2 gene expression in tumors with positive ER and PR receptors was insignificant (Foldchange: −0.59, P ≤ 0.311). In addition, our results represented a considerable reduction in GLIS2 expression in tumor samples with negative HER2 receptors (Foldchange: −2.47, P ≤ 0.002). In contrast, down-regulation in the expression of GLIS2 was not significant in HER2 positive samples (Foldchange: −0.87, P ≤ 0.276). GLIS2 expression reduction showed no association with neither positive nor negative LN involvement. 10 out of 12 samples of the patients with a history of using steroid drugs have shown reduced GLIS2 expression levels; however, the reduction was not significantly correlated with the GLIS2 down-regulation (Foldchange: −0.34, P ≤ 0.16).
Our results demonstrated that the expression level of CCND1 is significantly increased in cancerous tissues compared with controls (Foldchange: 2.31, P ≤ 0.002) (Fig. 4B). However, the altered expression level of CCND1 was not significantly correlated with any of the clinical features (ER, PR, HER2, TNBC, and LN) (P ≥ 0.05). In addition, Pearson correlation analysis showed no correlation between GLIS2 and CCND1 expression levels (Fig. 5).
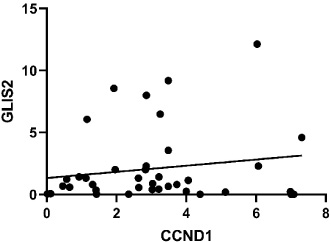
Correlation analyzing graph. Pearson correlation analysis showed no correlation between GLIS2 and CCND1 expression levels (P ≤ 0∕282).
Breast cancer is a heterogeneous disease with morphological, molecular, clinical, and genetic diversity. The primary cause of cancer is variation in the biological activity of tumor suppressors and oncogene genes. These genes are involved in specific pathways and interactions with other signaling pathways. Many signaling pathways play a potential role in tumor transformation, cellular metabolism, metastasis, apoptosis, proliferation, and cancer stem cell. Aberrant activation of one or more signaling pathways is a typical featuer in a malignant cell and plays a critical role in cancer fate [21]. Cancer seems complicated by the interaction between these pathways, so it is necessary to uncover the exact functions of genes and their related pathways in malignant cells.
Two well-known pathways, Wnt/β-catenin and Hh signaling play an essential role in BC. Hedgehog signaling activation was reported in several types of cancer, such as small-cell lung cancer [22], esophageal cancer [23], diffuse-type gastric cancer [24], pancreatic cancer [25], liver cancer [26], non-Hodgkin’s lymphoma, multiple myeloma [27], and BC [17]. There is an association between the overexpression of hedgehog signaling and its regulators with BC’s proliferation, migration, and aggressiveness [28,29]. Wnt/β-catenin signaling is an important pathway, and its alternation was reported in BC cells. Since expression of different Wnt members correlates with abnormal cell proliferation in human breast tissue, suggesting the potent role of aberrant activation of the Wnt signaling pathway in breast tumor initiation and metastasis [21]. Wnt/β-catenin can efficiently stimulate cell proliferation and is associated with a higher tumor grade and poor patient survival through promoting BC stem cell phenotype and drug resistance [21]. Moreover, there are pieces of evidence for a link between the hedgehog/Gli and Wnt/β-catenin signaling pathways [30,31]. In the current study, GLIS2 and CCND1 as regulatory genes of Wnt and hedgehog signaling pathways were selected by in-silico analysis. As well as identifying the paths involved, we tried to identify the relationships between them. The gene expression level of GLIS2 and CCND1 in BC patients were evaluated, and our results showed a significant reduction of GLIS2 in BC patients. Also, we observed down-regulation of GLIS2 in triple-negative (ER, PR, and HER2) samples and grade II and staged 3 of BC. It has been shown that the GLIS2 gene is the most common genomic location of ER signature genes in patients with BC [32]. Also, we observed that the differentially expressed GLIS2 was statistically significant in patients with negative ER and PR receptors. However, it was not significant in positive HER2 patients. since Positive HER2 patients are often ER/PR negative, it could cause conflict in the results. Several studies reveal that the triple-positive subtype accounts for about 10% of all breast cancer cases [33,34]. However, our data may come from a need for more samples because collecting sample biopsies from BC patients requires patients’ and physicians’ consent, making the collection of a large sample size too difficult.
GLIS2 represses gene transcription by binding to regulatory regions of target genes located at GLIS binding sites (GLISBS). Also, there is high homology between GLIS and the GLI as a component of the hedgehog signaling pathway. Thus GLIS and GLI proteins can recognize similar DNA binding sequences; they interfere with each other’s regulatory capabilities [35]. In this regard, the lack of GLIS2 leads to increased activation of the Hh signaling pathway and tumor progression. Based on evidence GLIS2 suppress the β-catenin signaling by enhancing the nuclear localization of β-catenin [36]. Consequently, reduction of GLIS2 may increase transcriptional activation by β-catenin and, therefore, progress BC. GLIS2 is also one of the modulator elements of the Calcium Signal Transducer 2 (TACSTD2 or TROP2) signaling pathway. Overexpression of TACSTD2 was seen in many cancers and is associated with cell proliferation, EMT, and metastasis. TROP2 involves increased expression and activation of several kinase pathways, including JAK2/STAT3, PI3K/AKT, PKA, NF-κB, and ERK MAP kinase. Studies showed that the reduction in GLIS2 gene expression leads to a lack of repressing effect of GLIS2 on TACSTD2 and promotes cancer progression indirectly [10,37]. Also, GLIS2 acts as a negative regulator of the Hh pathway by binding to its suppressor, SUFU. Moreover, considering the negative regulation of the Wnt/β-catenin signaling pathway by the SUFU factor, there is a possibility that GLIS2 interferes with both Hh and Wnt signaling pathways [38,39].
In this study, our findings showed the increased gene expression level of Cyclin D1 in BC patients. Several studies confirmed that the CCND1 is overexpressed in different human tumors and is a relatively early occurrence during tumorigenesis [40,41]. According to reports, CCND1 is a downstream target of the Hh signaling pathway, and cell proliferation is regulated by Hh/Gli1-mediated regulation of CCND1 [42].
Studies noted that cyclin D1 was one of the most characterized targets for Wnt/β-catenin in breast cancer, and a correlation between β-catenin activity and cyclin D1 expression was found in both BC cell lines and tissue samples of patients [43–45]. Studies demonstrated that Wnt/β-catenin signaling promotes cell growth by activating the CCND1 and Akt signaling pathways [46,47]. Also, Kim et al. showed that GLIS2 represses cyclin D1 promoter activity, and GLIS2 down-expression enhances the β-catenin-induced increase in cyclin D1 [48]. On the other hand, activation of GLIS2 is associated with the overexpression of trophoblast antigen 2, subsequently driving the G1/S- specific cyclin- D1 (CCND1) pathway and leading to tumorigenesis [37,49]. According to previous studies determining the potential role of the Hh and Wnt/β-catenin pathways in cancer, the results of this study favor the suppressive role of GLIS2 in the mentioned pathways.
On the other hand, our in-silico analysis indicated a correlation between the relative expression of the studied genes and Wnt and Rap1 Hedgehog signaling pathways. Based on the in silico analysis results, the TP53 tumor suppressor is the primary node in molecular signaling pathways and genes involved in other pathways. These interactions produce pathobiological conditions in tumorigenesis.
In summary, Our findings suggested that the decrease in GLIS2 expression may be associated with the progression of BC. Moreover, a potential correlation between the significantly higher expression level of the CCND1 gene and BC development has been observed. These observations could help uncover new therapeutic targets for treating patients with BC in the progressive stage. Nevertheless, further studies with larger sample sizes are required to validate these findings.
Footnotes
Acknowledgements
We thank our colleagues for their association and helpful discussions in this study.
Conflict of interest
The authors declare that they have no conflict of interest.
