Abstract
BACKGROUND:
Multifocal (MFBC)/multicentric (MCBC) breast cancer is being more recognized due to the improved imaging modalities and the greater orientation with this form of breast cancer, however, optimal surgical treatment, still poses a challenge. The standard surgical treatment is mastectomy, however, breast-conserving surgeries (BCS) may be appropriate in certain situations.
METHODS:
A total of 464 cases of MF/MCBC out of 4798 cases of breast cancer were retrospectively analyzed from the database of the Oncology Center, Mansoura University (OCMU), between January 2008 and December 2019.
RESULTS:
Radiologic involvement of multiple quadrants was reported in 27.9% by ultrasonography, 19% by mammography, and 59.1% by magnetic resonance imaging. BCS was performed in 32 cases (6.9%) while 432 cases underwent a mastectomy. Postoperative pathology revealed infiltration of other quadrants grossly in 23.5%, and under the microscope in 63.6% of the examined cases. Mean disease-free and overall survival were 95.5 and 164.6 months, respectively. When compared with MFBC, MCBC showed higher pathologic tumor size (p < 0.001), higher stages (p < 0.001), higher recurrence rates (p = 0.006), and lower DFS (P = 0.009) but with similar OS (P = 0.8).
CONCLUSION:
Mastectomy is still the primary treatment option for MCBC with higher recurrence rates compared with MFBC. However, BCS for properly selected MFBC is considered oncologically safe, following the same rules of breast conservation for unifocal disease.
Introduction
Breast cancer can be classified, according to the number of lesions into unifocal or multifocal/multicentric breast cancer (MF/MCBC) [1,2]. These lesions could be identified preoperatively, intraoperatively, or on final pathological examination of the surgical specimens [3]. The reported incidence of MF/MCBC varies widely among publications over the years (9–75%) [4–12].
The differentiation between unifocal and MF/MCBC is debatable. It is based either on the location within one or more quadrants or the distance between multiple lesions. If two or more synchronous ipsilateral lesions are separated by benign tissue and located within the same quadrant, they are considered multifocal but when located within different quadrants they are considered multicentric [13–15]. However, if we take into account the distance between multiple lesions, MFBC describes lesions within ≤5 cm apart and MCBC describes lesions within >5 cm apart [14,16–18]. These various definitions, used in the literature, lead to more confusion and difficulty in study comparisons [19].
Recently, a more accurate description was settled where MFBC defines the presence of several invasive tumors in the same quadrant or different quadrants if the distance between the foci is less than 5 cm, while MCBC is the presence of at least two invasive tumors in two different quadrants or the same quadrant but at least 5 cm apart [20].
Originally, BCS was relatively contraindicated in cases with MF/MCBC [21]. However, more recent studies showed that BCS is a safe option for cases of MF/MCBC, with either no effect or a statistically insignificant increase in local recurrence (LR), following the same rules of breast conservation in unifocal disease [22–28]. Recent guidelines still contraindicate BCS in most cases with MF/MCBC unless wide local excision with clear safety margins and acceptable cosmetic results could be achieved [29,30].
Patients and methods
Patient’s cohort and study design
Using the electronic database of the Oncology Center Mansoura University (OCMU) and after receiving the approval from Institutional Review Board of the Faculty of Medicine, Mansoura University (RP.18.08.21), we retrospectively identified 464 cases with MF/MCBC fulfilling the inclusion criteria out of a total number of 4798 cases diagnosed with breast cancer in the period between January 2008 and December 2019. Clinical, radiological, and pathological data, reports of surgical procedures, follow-up visits, and survival data were reviewed till December 2022.
Inclusion and exclusion criteria
Breast cancer lesions that were considered unifocal clinically or radiologically were included only if they proved pathologically in the surgical specimen to be MF/MC. On the contrary, cases that were reported to be MF/MC clinically or radiologically but proved to be unifocal on postoperative pathological assessment were excluded from this cohort.
A subset of patients (n = 32) who had MF/MC lesions radiologically who were initially treated with neoadjuvant chemotherapy (NACT) and were found to be unifocal on post-resection pathologic assessment, were included, even though none of these cases underwent pre-therapy biopsy from all these radiologically suspicious lesions.
Multiple lesions closely related to the primary focus (within ≤5 mm) were considered satellites (irregular extensions of the primary focus appearing as two separate lesions in 2D sections and could be connected in another plane of section), (CAP guidelines for invasive breast cancer, resection versions; 3.2.0.0 and 4.4.0.0) [31,32]. These lesions were not considered MF and were also excluded from the study. Patients with unifocal disease associated with lymphovascular tumor emboli in other breast quadrants were also excluded. Post-NACT, locally advanced unifocal breast cancer cases were assessed pathologically, and those with fragmentation effects were also excluded. However, those with additional lesions outside the primary tumor vicinity either grossly or microscopically were included. Lastly, patients with synchronous bilateral MF/MCBC were reviewed as two separate cases.
All the primary lesions were invasive, but additional foci/lesions were either invasive, non-invasive (in situ), or both.
Pathologic sampling and assessment
Due to the retrospective nature and the long duration of the study, no single definition was uniformly applied to all cases; some were defined according to quadrant distribution, and others according to the distance between lesions regardless of their distribution. A histopathologically-free breast tissue in-between MF/MC lesions was mandatory for diagnosis.
All specimens retrieved after both mastectomy and BCS were examined grossly for number, size in three dimensions, distances in between, and sites of invasive carcinoma. In mastectomy specimens, the tumor site was reported by quadrant.
Other pathological findings such as lymphovascular and perineural invasion, intraductal component, and post-NACT response were reported. Hormonal receptor status and Ki-67% were tested for the primary focus only.
Statistical analysis
Data were fed to the computer and analyzed using SPSS version 26.0 (IBM Corp., Chicago, IL, USA) on MacOS. Qualitative data were described using numbers and percentages and compared using the Chi-Square test. Quantitative data were described using medians for non-parametric data and means ±SD for parametric data, after testing normality using the Kolmogrov-Smirnov test. Student t-test or Mann-Whitney U test was used to compare independent groups. Kaplan-Meier test was used to calculate the OS and the DFS by using log-rank 𝜒2. Cox regression was used to calculate the predictors affecting the OS and DFS with the calculation of the hazard ratio. The significance of the obtained results (p-value) was judged at or below 0.05.
Outcomes
The primary outcome is the comparison of clinicopathological features and prognosis between MFBC and MCBC; While the secondary outcomes were (1) report the imaging findings of multi-quadrant involvement; and report the postoperative pathological report about the infiltration of other quadrants. (2) Display the recurrence rate and pattern of MF/MCBC and factors affecting DFS and OS.
Results
In this study, 461 patients were enrolled, and 3 female patients presented with bilateral synchronous MCBC; each side was presented as a separate case (n = 464). According to radiological and pathological data, MCBC was diagnosed in 328 cases (70.7%) and MFBC in 136 cases (29.3%) including 32 cases that underwent CBS.
Clinico-epidemiological characteristics (Table 1)
Showing the clinico-epidemiological characteristics
Showing the clinico-epidemiological characteristics
SD; Standard deviation, BMI; body mass index, No; number.
Most of the cases (459) were females, while only 2 males were included. The mean age was 50.3 ± 10.8 years. Family history of breast cancer was documented in 342 cases, a few of them showed positive family history (16.4%). The mean body mass index (BMI) was 34.9 ± 7.3 Kg/m2.
None of the patients was diagnosed through the national screening program, which started in late 2019. Clinically palpable lumps were reported in 374/414 (90.3%). A unifocal lesion was clinically detected in 261 patients (69.8%), two lesions in 28 (7.5%), and multiple in 5 (1.3%). The clinical size was measured roughly in 245 cases with a median of 3 cm. Axillary lymph nodes (ALN) were clinically assessed in 264 cases; suspicious ALNs were reported in 120 patients (45.5%). Nipple discharge was reported in very few cases (n = 15∕454, 3.3%); bloody discharge was reported in 9 cases. Nipple erosion was reported in 13 (3.2%) cases. Other skin manifestations such as skin dimpling/tethering, peau d’orange, and pathologic nipple retraction were positive in 181 (45%) cases.
Showing the characteristics of preoperative imaging
Showing the characteristics of preoperative imaging
MRI; magnetic resonance imaging, NAT; neo-adjuvant therapy, BIRADS; breast imaging and reporting data system.
The most common imaging modalities used were ultrasonography (US) (n = 432, 93.1%), followed by mammography (n = 312, 67.2%), and lastly MRI (n = 24, 5.2%). Calcifications were detected in US and mammography (n = 91∕179, 50.8% vs. n = 93∕225, 41.3%, respectively). Types and distribution of calcifications were reported.
Radiologically detected breast lesions were reported by the US (n = 409, 94.7%), mammography (n = 229, 73.4%), and MRI (n = 22, 91.6%). The number of these lesions was also reported.
Multiple quadrants involvement was reported by the US (n = 112∕401, 27.9%), mammography (n = 43∕226, 19%), and MRI (n = 13∕22, 59.1%). The median mass size reported by the US was 2.9 cm and by MRI was 3 cm.
Core needle biopsy (CNB) was the most used (n = 381, 83.7%), followed by excisional biopsy (n = 45, 9.9%), fine needle aspiration cytology (FNAC) (n = 18, 3.9%), and finally incisional biopsy (n = 11, 2.4%).
Different pathological types were diagnosed and the commonest was IDC (n = 325, 71.4%), followed by combined IDC + DCIS (n = 42, 9.2%), and ILC (n = 35, 7.7%).
Neoadjuvant therapy and surgical approaches
Almost half of the cases in this study (n = 231, 49.8%) received NACT. The tumor response was assessed both clinically and radiologically by RECIST 1.1. The radiological response recorded in 153 cases was as follows: tumors regressed in 81 (52.9%), remain stationary in 56 (36.6%), progressed in 12 (7.8%), and complete response was reported in 4 (2.6%) cases.
BCS was tried for a total of 49 cases (24 of which received NACT); however, it was successful for only 32/49 cases (65.3%). Axillary management was performed on 455 sides including initial axillary LN dissection (ALND) in 421 (92.5%), SLNB only in 14 (3.1%), and 20 (4.4%) SLNB proceeded to ALND. Immediate breast reconstruction was offered to 36 cases (7.8%) using either autologous flaps or silicone implants.
Postoperative pathology (Table 3)
Showing the characteristics of postoperative pathology
Showing the characteristics of postoperative pathology
*One case underwent ALND but with missed data regarding pN. DCIS; ductal carcinoma in-situ, IDC; invasive ductal carcinoma, ILC; invasive lobular carcinoma, NOS; non-otherwise specified, CPR; complete pathological response, ER; estrogen receptor, PR; progesterone receptor, HER 2 neu; human epidermal growth factor receptor 2, MF; MultiFocal, MC; MultiCentric, P; pathological, Y; history of neoadjuvant therapy, T; Tumor size, N; Nodal status.
IDC represented the most common pathological type (n = 381, 82.1%) while ILC was reported in 35 (7.5%), followed by mixed IDC + ILC in 21 cases (4.5%). The intraductal component was documented in a total of 211 cases of different invasive pathological types, of which 187 were reported in association with IDC pathology.
Regarding the number of masses, a single mass was detected in 214 (46.1%), two masses (n = 154, 33.2%), three masses (n = 39, 8.4%), and four or more (n = 48, 10.3%). Three patients (0.6%) could not be assessed due to the absence of definite tumor masses with only residual individual tumor cells or ill-defined areas of residual tumor tissue found. Complete pathological response (CPR) was documented in 6 cases (1.3%). The median pathological size of the largest tumor foci was 3.5 cm.
A gross and microscopic examination of the other quadrants away from the primary tumor foci was performed. Gross examination revealed the presence of additional malignant suspicious areas or foci (n = 99∕421, 23.5%), while microscopic examination showed malignant infiltration in two-thirds of patients (n = 273∕429, 63.6%). Invasive tumors were reported in half of the patients (n = 222, 51.7%).
Radiologic involvement of multiple quadrants was reported in 168 patients, while postoperative pathology revealed infiltration of other quadrants in 273 patients, representing a 61.5% accuracy rate of imaging in detecting MF/MCBC.
Postoperative adjuvant therapy was used frequently as per the hospital’s protocol. Chemotherapy (n = 313∕398, 78.6%), hormonal therapy (n = 327∕361, 90.6%), and radiotherapy (n = 302∕352, 85.8%) were prescribed.
Characteristics of recurrence
Recurrence was documented in nearly a third of patients (n = 152∕429, 35.4%). The most common form was a single distant metastatic site (DM) (n = 108, 71.1%), then local recurrence (LR) (n = 24, 15.8%). Bone was the commonest site of DM (n = 47, 39.2%). Multiple DM recurrences were also reported (n = 30, 25%).
Recurrence was diagnosed in only five patients from the BCS group vs. 147 cases in the mastectomy group. On analysis of the latter group, 83 patients showed positive invasive tumor involvement of other quadrants, 12 had DCIS only and another 9 had both in-situ and invasive disease.
Overall (OS) and disease-free survival (DFS) (Table 4)
Univariate and multivariate analysis of DFS
Univariate and multivariate analysis of DFS
DFS; disease free survival, HR; hazard ratio, NAT; neo-adjuvant therapy, CBS; conserving breast surgery, DCIS; ductal carcinoma in-situ, LVE; lympho-vasacular emboli, PNI; peri-neural invasion, ER; estrogen receptor, PR; progesterone receptor, HER 2 neu; human epidermal growth factor receptor 2, POP; post-operative pathology, MF; MultiFocal, MC; MultiCentric, T; tumor size, N; nodal status, CT; chemotherapy, RT; radiotherapy.
For all the patients in this cohort, the mean DFS and OS, from the time of surgery, were 95.5 (95% CI 84.5–106.5) and 164.6 (95% CI 161.3–167.9) months, respectively.
On univariate analysis, DFS increased significantly in the patients (1) who did not receive NACT (p < 0.001), (2) who did not have skin manifestations other than nipple erosion (p = 0.001) (Fig. 1), who did mastectomy (p = 0.025), no LVE (p = 0.016), with positive ER, PR status (p = 0.005, p = 0.006, respectively), low KI67 (p = 0.039), less mass number (p = 0.037), MF in comparison to MC (p = 0.009), lower staging, T and N status (p < 0.001 all), with adjuvant radiotherapy (p = 0.006).
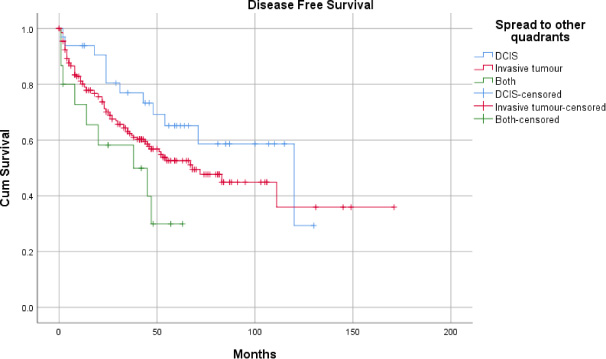
Showing Kaplan-Meier curve for DFS as regard microscopic involvement of other quadrants.
COX regression was run with NAT, breast surgery, ER, PR POP mass number, adjuvant RT, and either (pT & pN) or (P stage) or (skin manifestations & pN) and (MF/MC). LVE and Ki were excluded although significant in the univariate analysis as they are recorded in a small number of patients, limiting the validity of the multivariate analysis.
However, on multivariate analysis, the following shortened DFS: receiving NACT 1.7 folds, advanced stage (8 folds for stage III in comparison to stage 1), T (T4 3.4 folds in comparison to T1), and N (pN3 4.8 folds to N0). While receiving adjuvant radiotherapy lengthened DFS 2 folds.
Comparison between MF vs. MC breast cancer
Comparison between MF vs. MC breast cancer
POP; Post-operative pathology, MF; MultiFocal, MC; MultiCentric, BMI; body mass index, NAT; neo-adjuvant therapy, CBS; conserving breast surgery, ER; estrogen receptor, PR; progesterone receptor, HER 2 neu; human epidermal growth factor receptor 2, P; pathological, T; tumor size, N; nodal status, DFS; disease free survival, OS; overall survival, Min; minimum, MAX; maximum.
MCBC patients, compared with MFBC, showed a slightly higher mean age of incidence (p = 0.09), nearly similar BMI (p = 0.952), similar ER and PR status (p = 0.749, p = 0.99, respectively), slightly lower HER-2-neu +3 expression (p = 0.602), and lower Ki-67% (p = 0.130). However, patients with MCBC showed a significantly larger tumor size (median = 4 cm vs. 2.75 cm) (p = <0.001), higher pathological T, N, and stage (p = <0.001, each), and higher recurrence rate (39.7% vs. 25.9%, p = 0.006). MCBC showed significantly lower DFS compared with MFBC (mean 88.9 vs. 102.5 months, respectively, p = 0.009) (Fig. 2), and non-significantly higher OS (mean 164.4 vs. 154.9 months, respectively, p = 0.8).
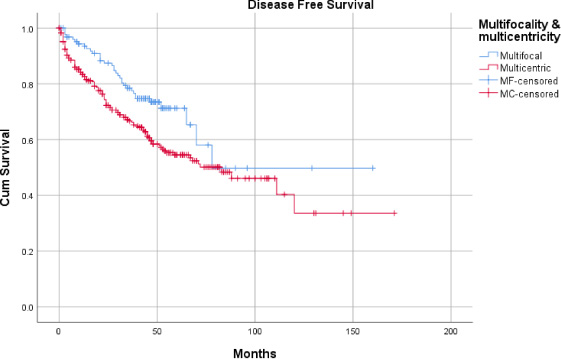
Showing Kaplan-Meier curve for DFS comparison between MF and MC breast cancer.
Cheatle [33] was the first to identify the presence of simultaneous ipsilateral breast cancer lesions away from the primary tumor, then it was further distinguished by Qualheim and Gall [4] into MF/MC according to its distribution within breast quadrants.
Many theories tried to describe the exact origin of MF/MCBC; either the intramammary spread of a single primary focus or a simultaneous outgrowth of independent cancer foci without or within an area of extensive DCIS [21]. The support of intra-mammary metastasis theory comes from the clonal homogeneity within the majority of MF/MC lesions [34], which describes a concordance of hormonal receptor status between multiple breast lesions [35]. Despite that, there is a chance of inter-lesion heterogeneity [36], especially if there is a difference in grades or histological subtypes between these lesions [34].
The incidence of MF/MCBC in our series was 9.67% which is in concordance with that of previous studies (9–75%) [4–12]. This wide variation in incidence in the literature is entirely dependent on the method used for specimen sampling [37].
Additionally, routine sampling of suspicious lesions and random biopsies from other grossly free quadrants, as per the hospital’s protocol, lead to the detection of more MCBC/MFBC cases (70.6%, 29.4%, respectively). Microscopic malignancy was detected in 63.6% while gross malignancy was detected in only 23.5% of patients in our cohort.
Different imaging modalities are being used in the diagnosis and assessment of multicentricity. Ultrasonography showed higher specificity and sensitivity compared to mammography in the younger age groups <40 years [38–40]. However, MRI showed a higher detection rate of MF/MCBC when compared with other imaging modalities [41,42] (up to 16% of women showing additional lesions) [42]. This resulted in higher rates of conversion to mastectomy (6.5–25%) [42–46].
Our results showed a higher mass detection rate with both US and MRI compared with mammography (94.7% and 91.6% vs. 73.5%, respectively), despite the low number of MRI scans in our series. There was also a higher detection rate for multiple lesions by MRI compared with US and mammography, despite not being biopsied to exclude the false positive lesions.
Before starting NACT, a biopsy from all radiologically-detected lesions should be done as recommended by both ESMO [29], and NCCN guidelines [30]. Unfortunately, that was not the common practice in our center due to cost-benefit and technical issues.
Ataseven et al. [47] collected 6134 patients from three major studies (GeparTrio, GeparQuattro, and GeparQuinto). They found that only MCBC cases showed significantly worse DFS and OS, however, in case of successful BCS or obtaining PCR following NACT, MF/MCBC were not inferior in terms of LR, DFS, and OS. Our results showed a statistically significant association between MCBC, and recurrence rates compared with MFBC cases with significantly lower DFS and higher albeit non-statistically significant OS.
Originally, BCS was relatively contraindicated in MF/MCBC due to a presumed higher risk of recurrence, both locally (up to 40%) and distantly [48–50]. Recent studies showed that BCS is a safe option for cases of MF/MCBC, with either no effect or a statistically insignificant increase in LR, following the same rules of breast conservation for unifocal disease [22–28]. Nos et al. [22] reported that the 5-year LR of patients with MFBC was 11% and the OS was 94% (BCS group) and 90% (mastectomy group). Additionally, Kaplan et al. [51] did not find a difference between these groups concerning the 5-year DFS (p = 0.20). Similarly, Lim et al. [23] found no difference in 5-year OS (p = 0.208), 5-year DFS (p = 0.451), and LRR (p = 0.378). Wolters et al. [26] reported that in the MFBC, both BCS and mastectomy would be appropriate guideline adherent options for T1/2 tumors and they could not demonstrate any significant difference in survival between these two surgical options.
In our series, 32 patients underwent successful BCS, all were MFBC. Five patients only had recurrence during the follow-up period; two had LR, and three had distant metastasis (two in bone, one in lung). This proves that BCS could be safely performed with acceptable oncological results in properly selected cases, mainly the MF variety.
It is worth noting that patients requiring mastectomy following NACT showed a higher risk for locoregional recurrence and should therefore be monitored closely [47]. In our series, recurrence in the mastectomy group, according to NAT status, was documented as (n = 85 post NACT, n = 62 without NACT). Microscopic involvement of other quadrants was also detected (n = 141 post NACT, n = 132 without NACT) in general, and regarding association with recurrence (n = 62 post-NACT, and n = 42 without NACT). The commonest pattern was the invasive type, emphasizing the importance of different factors like pre-NACT staging, response to NACT, extent, and pattern of the disease in remaining breast tissue, and the availability of adjuvant radiotherapy. This was supported by multivariate analysis showing significant associations between DFS and NACT and adjuvant radiotherapy.
Nonetheless, recent guidelines still contraindicate BCS in most cases of MF/MCBC unless wide local excision with clear safety margins and an acceptable cosmetic result could be achieved [29,30].
Limitations of the study
Our study had some weaknesses including its retrospective descriptive design, failure to prove pathological multifocality/multicentricity by preoperative biopsy before NACT in many instances, and the heterogeneity of applied definition and data availability among cases. Thus, the presented results and their interpretations should be perceived with caution taking into consideration stronger evidence in the previously published studies in the literature.
Conclusion
Mastectomy is still the primary treatment option for MCBC. Higher recurrence rates are reported with MCBC compared with MFBC variety. However, BCS for properly selected cases of the MFBC variety is oncologically safe following the same rules of breast conservation for unifocal disease.
Footnotes
Ethical approval
All procedures performed in studies involving human participants were following the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Conflict of interest
All authors declare no conflict of interest.
