Abstract
INTRODUCTION:
Studies in recent years have shown that parental environmental experiences can affect their offspring’s risk of breast cancer (BC). We assessed the effect of different paternal factors on BC risk in offspring by reviewing the existing literature.
METHOD:
This systematic review followed the Joanna Briggs Institute’s (JBI) method for systematic reviews of qualitative evidence. The primary keywords were searched in reliable databases such as PubMed, Google Scholar, Elsevier, SID, and Wiley in English until 31 December 2021. Two authors independently examined the articles in terms of inclusion criteria and quality assessment of the articles.
RESULTS:
Of the 438 studies, 19 met the inclusion criteria of this systematic review and were included in the study. Paternal factors investigated in these studies included age at delivery, diet, occupational exposures, occupation type and education. The reported relationships between these factors and breast cancer varied among different studies.
CONCLUSION:
Studies considered in this article show that fathers’ age at the time of delivery of the child, dietary habits, overweight and occupational factors can affect the incidence of BC risk in the next generation.
Introduction
Breast cancer is the most common cancer worldwide, with more than 2 million new cases in 2020 and the fifth cause of cancer-related deaths globally in women [1,2]. Likewise, in Iran, breast cancer is one of the most frequent malignancies in women [3,4]. Thus, addressing the predisposing factors of breast cancer has become one of the most critical research areas. Breast cancer risk factors come in a wide range, from genetics to environmental factors [5].
About 5% to 10% of breast cancer cases are hereditary, meaning that they result directly from gene changes (mutations) passed on from a parent. The most common cause of hereditary breast cancer is an inherited mutation in the BRCA1 or BRCA2 genes which are seen in one out of every 400 individuals. In other cases, the individual’s genetic mutations and environmental factors can directly or indirectly affect the genomics of individuals and can cause cancer [6].
Interestingly recent studies have highlighted that parent’s past environmental experiences, such as risk factor exposures, diet, and socioeconomic factors, can affect the risk of breast cancer in the next generation by leaving a molecular memory through genetic and epigenetic changes. Epigenetic modifications, such as DNA methylation and histone modification are heritable alterations that are due to changes in DNA accessibility and chromatin structure apart from changes in DNA sequence [7–11]. According to our literature research, this influence has been studied mainly from a maternal perspective, emphasizing intrauterine factors however, discovered paternal factors form a small part of the evidence.
Therefore, this study aims to review the evidence and determine the effect of different paternal factors on breast cancer risk in offspring. In general, paternal factors investigated in the literature, and therefore in this systematic review, include age, diet, overweight, and occupational factors.
Method
This systematic review of qualitative studies followed the Joanna Briggs Institute’s (JBI) model, which is used for systematic reviews of qualitative studies [12] and the PRISMA statement [13]. Our major question in this study is Can paternal environmental experiences affect the BC risk in offspring?
Inclusion and exclusion criteria for the selection of studies
This study included all the human or animal studies that investigated the relationship between paternal factors, including all environmental factors and the incidence of BC in offspring, which were published until the end of December 2021. Studies investigating only genetic or maternal factors without considering paternal environmental factors were removed.
Search strategy
Keywords including “Paternal factors”, “Breast cancer”, and “offspring” without using (“”) were searched using OR and AND operators in databases such as Google Scholar, PubMed, ISI, SCOPUS, Elsevier, and Wiley. In order to obtain all available pieces of information, we did our search among all studies published until the end of December 2021.
Selection of texts
We found 753 articles in databases. Duplicate studies (N = 438) were removed. Based on the selection criteria, out of 438 screened abstracts, 82 abstracts were extracted and reviewed. Due to inconsistency with the objectives of the study (N = 52), unavailable full text (N = 1), Case report (N = 1), and Review (N = 7) were excluded by reading their full texts. In the end, 19 records remained (Fig. 1).
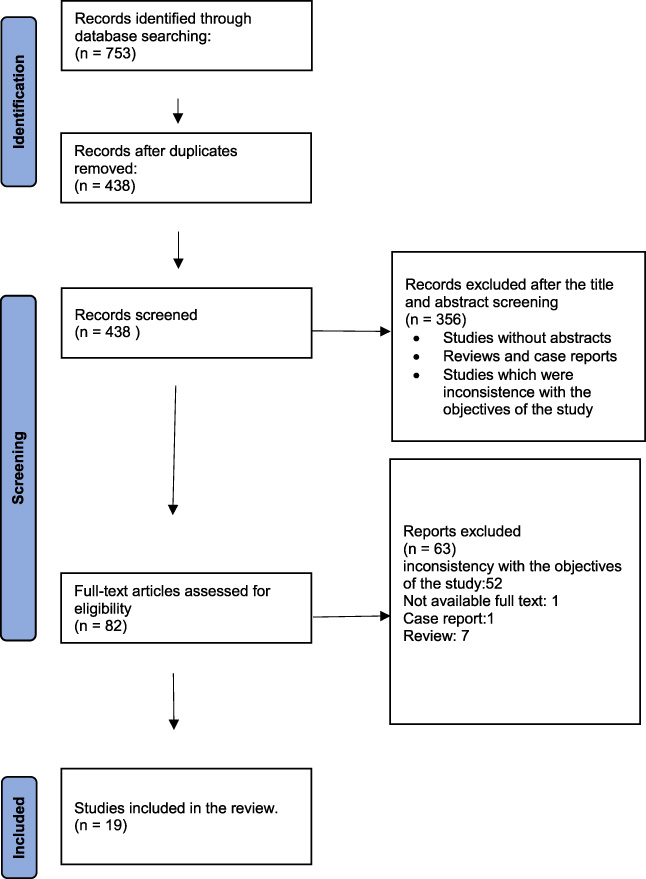
Flow of information through the various phases of the systematic review.
Two independent reviewers critically assessed eligible studies for methodological quality according to the JBI critical appraisal checklist for qualitative research [12]. Any disagreements between two reviewers were resolved with the help of a third reviewer [14]. All studies were included in the analysis, and they scored 8 or more out of 10. The results are given in the Table 1.
Limitations
Despite an extensive search in this systematic review and a detailed review of each article, a significant number of articles addressing important paternal factors were animal studies, and no similar human studies were found; thus, findings from these studies may not be generalizable to other settings. In addition, a limited number of studies were found in the literature which investigated the relationship between paternal factors and breast cancer risk; meanwhile, maternal factors were an interfering factor in many of these articles, especially in studies that investigated paternal age as a risk factor. These points highlight the need for more extensive human studies in this area to address the shortcomings of the evidence.
Results
During the initial search, which was conducted among all the studies until 31 December 2021, 438 studies were obtained from considered databases after the removal of duplicated articles; the study selection process resulted in the inclusion of 19 independent studies, all held from 1991 to 2021, including 14 human studies and five animal studies. Paternal factors investigated in these studies fall into different categories: age at delivery, diet, occupational exposures, occupation type, and education. 11 studies investigated the association of the father’s age at the time of delivery with BC risk in their children. Four considered diet, four socioeconomic status on BC development in offspring. The results obtained from these studies are as follows (Table 3):
Critical appraisal results for included studies using the JBI-Qualitative Critical Appraisal Checklist
Critical appraisal results for included studies using the JBI-Qualitative Critical Appraisal Checklist
Q1: Is there congruity between the stated philosophical perspective and the research methodology? Q2: Is there congruity between the research methodology and the research question or objectives? Q3: Is there congruity between the research methodology and the methods used to collect data? Q4: Is there congruity between the research methodology and the representation and analysis of data? Q5: Is there congruity between the research methodology and the interpretation of results? Q6: Is there a statement locating the researcher culturally or theoretically? Q7: Is the influence of the researcher on the research, and vice-versa, addressed? Q8: Are participants, and their voices, adequately represented? Q9: Is the research ethical according to current criteria or, for recent studies, and is there evidence of ethical approval by an appropriate body? Q10: Do the conclusions drawn in the research report flow from the analysis, or interpretation, of the data? Abbreviations: N, No; U, Unclear; Y, Yes.
The characteristics of included articles
R = Related, F = Found, NS = Not studied, NR = Not related.
This section explains the results of different studies separately based on the type of risk factor. In all these studies, p-value < 0.05 was considered statistically significant.
11 studies investigated the relationship between breast cancer risk and paternal age at the time of birth, and eight of them reported a positive relationship between advanced paternal age and risk of breast cancer in offspring.
According to the results of a cohort study done by Zhang et al. in 1995 on 2620 females considering paternal age, the breast cancer incidence rate was lowest for women whose fathers were less than age 30 at their birth, and highest for women whose fathers were 30 to 34 at their birth, however, this results were no longer significant after adjusting maternal age [17].
Five years later Innes et al. conducted a case-control study and they also reported that breast cancer risk rises progressively with increasing paternal age (p-value = 0.03) [19].
In 2004 Hodgson et al. conducted a case-control study on 196 cases with BC and 167 healthy women as controls. After adjustment for maternal age, older paternal age increased risk in the oldest and youngest women’s own age categories (relative to 23–27 years of age at the woman’s birth: OR 1.6, 95% CI 0.8–3.1 for age 15–22 years; OR 1.2, 95% CI 0.7–2.2 for age 28–34 years; and OR 1.5, 95% CI 0.7–3.2 for age 35–56 years) [20].
The results of another case-control study by Choi et al. in 2005 on 1011 cases (patients with histologically confirmed breast cancer) and 1011 controls showed that the risk of breast cancer increases significantly as the paternal age increases (p-value = 0.025), and this association becomes stronger after controlling for maternal age; women whose fathers were over 40 years old at their birth had a 1.6-fold increased risk of breast cancer compared to women whose fathers were under 30 years old [21].
A cohort study of 42 822 females by Weiss-Salz et al. in 2007 reported a positive association between advanced paternal age and the occurrence of early breast cancer (p-value = 0.005) [22].
Same results were concluded from another cohort study in the same year (2007) on 109773 females done by Xue et al.; even though paternal age at delivery seemed positively related to the BC risk in women (p-value = 0.03), this association disappeared after adjustment for the mother’s age [23].
In 2011 Abreeve et al. conducted a cohort study on 2177 females and results showed that daughters with older fathers were less likely to stay free of BC (p-value = 0.0012) and had longer leukocyte telomere length, which can be a cancer risk factor for BC [25].
A case-control study of 238 women with breast cancer and 153 healthy women as the control group done by Bothuo et al. in 2020 also showed that there is a statistically significant positive correlation between the age of the father and breast cancer risk (p-value < 0.001); the majority of breast cancer patients had fathers over 37 years old at the time of birth [32].
Unlike the studies mentioned above, three studies concluded no significant relationship between the age of patient’s fathers at the time of their birth and BC risk.
Colitz et al. examined the relation between parental age at birth and the risk of BC in a population of 118,309 women in 1991. As the result, paternal age was not significantly related to the risk of breast cancer (p-value = 0.76) [15]. The study of Janerich et al. in 1994 also had the same results and no association was found between breast cancer risk and paternal age (p-value = 0.59) [16].
The study by Newcomb et al. in 1997 also found no association between paternal age and breast cancer risk (p-value = 0.98) [18].
Paternal diet and breast cancer in daughters
Five studies investigated the father’s diet as a breast cancer risk factor. These five animal studies investigated nutritional state factors such as fat intake, protein intake, and micronutrients. None of the studies investigated the effect of carbohydrate intake. Dietary factors investigated in each study fall into two categories: Nutrition and Fat intake.
The effect of paternal fat intake on breast cancer incidence in offspring
The effect of paternal fat intake on breast cancer incidence in offspring
N = sample size.
The effect of paternal obesity inducing diet on breast cancer incidence in offspring
N = sample size.
In the same year, they (Fontelles et al.) studied the effect of an obesity-inducing diet (OID) of fathers on breast cancer risk with the same method on two groups of mice: they induced BC in offspring of two groups (case group with obesity-inducing diet and control groups with normal diet), and after 20 weeks 80 percent of the mice in OID group developed BC which was significantly higher than the control group with 50 percent rate of breast cancer development (p-value < 0.05) (Table 4), OID in fathers also led to a higher incidence of palpable mammary tumors and Tumor multiplicity in their offspring (p-value < 0.05) [33].
The effect of paternal malnutrition on breast cancer incidence in offspring
N = sample size.
Besides macronutrients, Guido et al. studied the father’s selenium deficiency and its potential role in increasing breast cancer risk among their offspring. Three groups of 24 mice were fed a selenium-deficient diet, a selenium-deficient diet with supplementation, and a normal diet. Paternal selenium deficiency increased the number of terminal end buds, epithelial elongation, and cell proliferation in the mammary gland of the female rat offspring. These effects were associated with increased breast tumor incidence and higher-grade tumors, as only 0.08 percent of the offspring with selenium-deficient fathers were cancer-free after induction (p-value < 0.05). On the other hand, paternal selenium supplementation did not influence these parameters (Table 6) [28].
The effect of paternal selenium deficiency on breast cancer incidence in offspring
N = sample size.
Weiss-Salz et al. concluded that paternal education is not significantly related to BC incidence risk; in this cohort study with 71 BC patients, fathers of 33 of them had below eight years of education, 20 had fathers with 9–12 years of education, and fathers education of 14 of them were more than 13 years; however, these differences were not statically significant (p-value > 0.05) (Table 7) [22].
The effect of paternal educational status on breast cancer incidence in offspring
The effect of paternal educational status on breast cancer incidence in offspring
N = sample size, n = number of cases in each group, % = percentage of cases in each group.
The study of Pudrovska et al. reported that although the father’s education does not significantly affect the incidence of breast cancer in children, women whose fathers had a high school education or higher have an 88% lower risk of breast cancer death. However, this effect becomes less significant when the effect of the women’s own education is considered [27].
Carren et al. conducted a human cohort study on 76 women whose fathers had exposure to dibutyl phthalate during their serving in Malaya (1948–1960); 3 of these women developed BC, which shows a statistically significant positive association between this exposure and breast cancer incidence (Table 8) [26].
The effect of paternal occupational exposure on breast cancer incidence in offspring
The effect of paternal occupational exposure on breast cancer incidence in offspring
N = sample size, n = number of cases, % = percentage of cases in sample size.
An animal study done by Da Cruz et al. revealed that paternal exposure to DDT slightly increases BC incidence, shortens the latency period, and considerably increases tumor growth (Table 9). In this case-control study, 25 mice were examined in two groups, one of which was exposed to DDT. Finally, the offspring of the exposed group had 20% more breast cancer incidence than the control group [31].
The effect of paternal occupational exposure on breast cancer incidence in offspring
In a long-term study using a random sample of 10,317 women and men, Pudrovska et al. concluded that the father’s occupation, education, and occupational prestige were unrelated to the daughter’s onset of BC (Table 10) [27].
In another human cohort study held by de Kok et al., 209 breast cancer patients were classified according to their father’s occupational type (high professional, lower grade professional, self-employed, high manual, and low manual), and no significant association was observed between types of occupation and BC risk (Table 10) [24].
The effect of paternal types of occupation on breast cancer incidence in offspring
The effect of paternal types of occupation on breast cancer incidence in offspring
N = sample size, n = cases in each group, % = percentage of cases in each group.
Some of the studies mentioned in this article also investigated the epigenetic changes due to considered paternal factors that can be responsible for changes in the next-generation breast cancer risk. Among five studies considering the abovementioned points, one was a human study, and the others were animal studies. The most commonly discovered epigenetic changes in studies concluded in this article are telomere length alternations, DNA methylations, and alternations in microRNA expression.
In another study conducted by Da Cruz et al. on the impact of the father’s exposure to DDT and breast cancer in the next generation, a comparison of sperm samples was also performed using a similar method. The results showed that the sperm of DDT-exposed males showed a larger quantity of miRNAs (6.7%) than controls (1.4%). Furthermore, 16 miRNAs had expression alternations, with nine being down-regulated and seven being upregulated, in the sperm of DDT-exposed mice compared to controls [31].
According to the study of Fontelles et al. on mouse models, it is reported that paternal overweight around the time of conception and the increased mammary cancer risk were associated with epigenetic changes in the miRNA profile of paternal sperm and their daughters’ mammary tissue due to an increase in body weight. Certain miRNAs that have been altered can regulate genes that are commonly deregulated in cancer, particularly those related to the hypoxia signaling pathway. This pathway was modified in mammary tissue and mammary tumors of offspring whose fathers were overweight [33].
It was also found in another study by Fontelles et al. that male rats who consumed corn oil had 89 downregulated (p-value < 0.05) miRNAs in the sperm compared with male rats fed lard. Furthermore, female offspring of corn oil-fed male rats had 21 downregulated (p-value < 0.05) and two upregulated (p-value < 0.05) miRNAs in their mammary glands compared with female offspring of lard-fed male rats [29].
Table 2 summarizes the factors explored in each study and the obtained results.
Discussion
Evidence from recent studies suggests that parent’s environmental experiences can affect their offspring’s risk of breast cancer by creating a molecular memory through epigenetic changes and genetic material are not the only things that transmit to the offspring at conception [8,11]. It has been experimentally shown that the risk of environmental disease is transmitted from one generation to another through epigenetic mechanisms through the male and female germlines [34–36].
Although most of the evidence for this mode of disease inheritance comes from maternal exposures in pregnancy, it seems that the importance of paternal factors should not be underestimated, since the zygote, which is the first cell forming human body, has the genomic and epigenomic content of both parents due to the process of fertilization [37,38].
In this study, we tried to collect the existing evidence and studies investigating paternal factors. One of these factors is age. Aging and cancer formation may seem like opposite processes; however, they share many common characteristics that may indicate an association between them [39]. Studies show that for each year of increase in father’s age at the birth of the offspring, leukocyte telomere length in the daughters and sons were longer by 15.5 bp and 13.7 bp, respectively [25]. Longer telomeres are known to be associated with an increased risk of cancer development overall and several common cancer types, including breast cancer [40].
Another two of these common characteristics are increased genomic instability and methylation. Methylation has been suggested as the predictor of human age in many genome-wide methylation studies [41], but this relationship is complex. Aging is associated with increased global hypomethylation, but many loci, like many tumor suppressor genes, are hypermethylated [42,43]. In addition, evidence suggests that microRNA is involved in regulating many essential pathways in aging and cancer [41].
Two other factors are diet and overweight, which can program the sperm epigenome, particularly sperm non-coding RNAs. An obesity-inducing diet alters the expression of miRNAs in sperm. Studies show differences in sperm miRNAs due to diets with two different types of fat (animal or vegetable origin). A low protein diet also alters the distribution and content of the small RNA species including microRNAs in paternal sperm [8].
Other studies indicate that placental development changes have worth exploring as another paternally induced mechanism of epigenetic inheritance [44,45]. The placenta connects the fetus to the mother and provides the nutrient and gas exchanges necessary for proper development of the fetus and arises from the trophectoderm that forms the outer cell layer of the embryo in the blastocyst stage [46]. It attaches the placenta to the mother, provides the nutrients and gas exchanges necessary for the proper development of the fetus, and forms the structure of the outer cell layer of the fetus in the blastocyst stage through the trophyctoderm. Improper placental development disrupts the passage of nutrients and oxygen to the fetus and has been proven to have implications of future disease for the offspring, such as hypertension, type 2 diabetes, and cancer [47]. According to the studies, the patterns of gene expression are different between placentas from offspring of obese and non-obese fathers, because the obesity of the father considerably increased overall DNA methylation amounts in female placentas. Thus, the role of the placenta could be an answer to how poor paternal nutrition can result in increased risk of breast cancer and other diseases in offspring [30].
Regarding socioeconomic status, occupational exposures play a significant role. While there is a clear link between maternal DDT exposure and breast cancer in the next generation, no human study has directly examined the link between father DDT exposure and breast cancer in their offspring. However, there is evidence for the role of related paternal encounters. For example, a link between father’s exposure to pesticides and several childhood cancers has been reported in children [48,49]. Epidemiological studies also show that father exposure to DDT and other pesticides is associated with low birth weight [50]. Low birth weight is more common in minority populations [51,52] which are more exposed to environmental toxins such as DDT and develop more invasive breast tumors [53]. The study of Da Cruz et al. demonstrates that the exposure of the father before pregnancy with DDT pesticide modulates the load of sperm non-coding RNA, especially miRNAs, and programs the growth of female offspring breast cancer in a pre-clinical animal model [54]. Recent studies show that phthalates can disrupt human placental function by epigenetically regulating important placental genes and altering microRNA expression in paternal sperm [55,56].
Conclusion
Intrauterine factors, which may increase cancer risk in individuals by leaving genetic and epigenetic changes, are not only influenced by maternal environmental conditions. However, the father’s environmental condition can also be important in this regard. According to the literature, four major paternal characteristics can influence the BC risk in offspring: the age of the father at the time of birth, the diet of the father, paternal education, and type of occupation. These factors can cause epigenomic alternations in the paternal germline, such as telomere length alternations, DNA methylations, and alternations in microRNA expression that can be the reason for the association between paternal environmental condition and BC risk in offspring. Given the importance of breast cancer, conducting more extensive human studies in this field can provide more substantial evidence to prove the relationship between paternal factors and breast cancer.
Footnotes
Ethics approval
This is a systematic review study and no ethical approval is required.
Consent to publish
All authors had given their consent for the publication of identifiable details, which can include photograph(s) and/or videos and/or case history and/or details within the text (“Material”) to be published in the above journal and article. Therefore, anyone can read material published in the Journal.
Competing interests
The authors have no relevant financial or non-financial interests to disclose.
Funding
The authors declare that no funds, grants, or other support were received during the preparation of this manuscript.
Author contributions
All authors contributed to the study conception and design. Material preparation and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
