Abstract
BACKGROUND:
Lymphoma of the breast can be classified as either primary breast lymphoma (PBL) or secondary to systemic lymphoma (SBL). PBL is a rare disease with Diffuse Large B cell Lymphomas (DLBCL) being the most common subtype.
OBJECTIVES:
In the current study, we represented eleven cases diagnosed with breast lymphoma in our trust; two of them had PBL and nine had SBL. We focused mainly on the clinical presentation, diagnosis, management and outcomes.
METHODS:
We did this retrospective review for all breast lymphoma patients who were diagnosed in our trust during the periods from 2011–2022. Patients’ data were obtained from the hospital recording system. We followed up these patients thus far to identify the outcome of treatment in each patient.
RESULTS:
Eleven patients were included in our review. All patients were females. Average age of diagnosis was 66.1 ± 13 years of age. Eight patients were diagnosed with DLBCL, two patients were diagnosed with follicular lymphomas, and the last one had lymphoplasmacytic lymphoma. Chemotherapy +∕− radiotherapy was the standard treatment regimen in all patients. Four patients passed away within one year of chemotherapy, five patients achieved complete remission, one patient had two relapses and is still under treatment, while the last patient was diagnosed recently and still awaiting treatment.
CONCLUSION:
Primary breast lymphoma is an aggressive disease. The treatment for PBL is mainly systemic with chemoradiotherapy. The role of surgery is now limited to the diagnosis of the disease. Early diagnosis and proper treatment are crucial for the management of such cases.
Introduction
Malignant lymphoma can arise either within or outside the lymph nodes. Malignant lymphoma of the breast can be classified as primary (PBL); where the breast is the principle site of involvement, or secondary to systemic lymphoma (SBL) [1]; however, the distinction between PBL and SBL is usually challenging [2,3]. Primary breast lymphoma (PBL) is rare and accounts for about 0.04% to 0.5% of all breast cancer cases [4]. Diffuse Large B-cell Lymphoma (DLBCL) represents the most common subtype of PBL, other less frequent subtypes also were reported [5,6].
Primary breast lymphomas mainly present in females and roughly 11% of the cases are bilateral [7]. The mean age of diagnosis is from 60–65 years [8]. PBL could present as a solitary breast mass, multiple masses or diffuse breast enlargement. It is usually difficult to distinguish breast lymphoma from other primary breast cancers, either clinically or radiologically and the diagnosis is based mainly on histological assessment after core biopsy or surgical excision.
There are different treatment options for breast lymphoma including surgical excision, chemotherapy and/or radiotherapy [9].
In the current study, we represent eleven cases of breast lymphoma; nine cases were diagnosed with secondary breast lymphoma and two were classified as primary breast lymphomas. We focused primarily on the challenges associated with the diagnosis and treatment of such patients, particularly given the similarity in presentation to that of breast carcinoma.
Methods
We carried out this retrospective review for all patients who were diagnosed with breast lymphoma in our trust during the period from 2011 to 2022. All patients diagnosed with breast lymphoma as either primary or secondary were included in our study. The data were collected retrospectively from the hospital recording system.
The patients included in our study attended our breast clinic for the assessment of either a clinically palpable breast lump or imaging detected breast mass. Mammogram and ultrasonography were used as the initial imaging after clinical examination, (Figs 1–4). All suspicious breast masses underwent core biopsies after obtaining the appropriate verbal consent. The diagnosis of breast lymphoma was confirmed only after histopathological examination. CT and/or PET/CT scans were used to identify the stage of the disease. All cases were discussed in the breast multidisciplinary team meeting and were referred to the haematology team for further assessment and management. We included a summary of the clinical presentation, management plan and the outcome of treatment for every patient up till now.
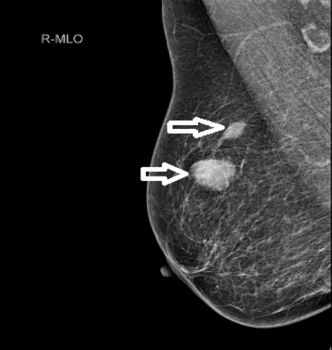
Right breast mammogram showing breast density BIRADS A with two nodules at the 12 o’clock position; 19 mm and 15 mm in diameter with no demonstrable calcification in these masses. Right M5. Right axillary lymph nodes are prominent. Final histology confirmed High-grade B-cell lymphoma.
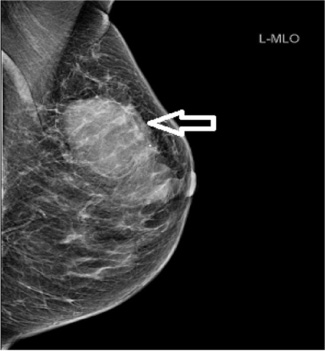
Left breast mammogram showing fairly dense tissue pattern type BIRADS B/C, with an asymmetric density in the upper central area measuring approximately 53 mm. Final histology confirmed Diffuse Large B-Cell Lymphoma, NOS.
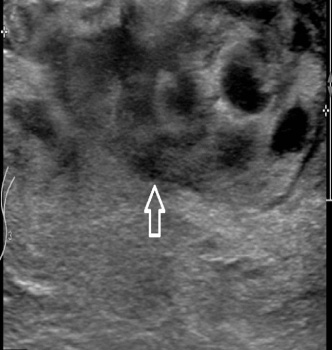
Right breast ultrasonography showing a 50 mm ill-defined hypoechoic area, U4. Final histology confirmed Diffuse Large B-Cell Lymphoma, NOS.
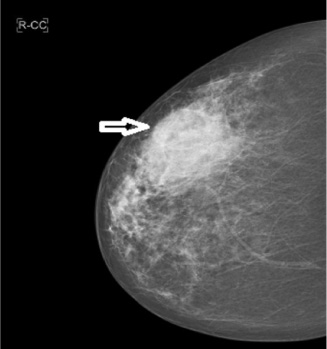
Right breast mammogram showing fatty breast with a dense irregular lesion measuring 74 mm in diameter which is 35 mm deep to the nipple, M5. Final histology confirmed Diffuse Large B-Cell Lymphoma, NOS.
Eleven patients were included in our review. Nine patients were diagnosed with systemic lymphoma (SBL) with secondary breast involvement, four of them had previous diagnosis of systemic lymphoma; three presented with a breast lump and one had an incidental finding of breast mass on a surveillance CT scan, while five patients presented initially with breast lump and a systemic disease was found on further work-up. On the other hand, two patients were diagnosed with primary breast lymphoma (PBL) where the disease was limited to the breast and the local lymph nodes. The main age of diagnosis was 66.1 ± 13 years of age.
Regarding the risk factors, four patients with SBL had previous lymphoproliferative disease and one was on immunosuppressive therapy. The two cases with PBL had no significant disease apart from Paget’s disease of the bone in one of them; both patients had left sided breast lymphoma. Of the nine patients diagnosed with SBL, six patients had right sided breast disease and it was left-sided in the remaining. Eight patients were diagnosed with B-cell lymphoma with Diffuse Large B-cell Lymphoma being the most common subtype, two patients were diagnosed with follicular lymphoma, and the last one had lymphoplasmacytic lymphoma. Immunohistochemical studies were performed and confirmed the diagnosis of DLBCL (CD20+, BCL2+) with high Ki67 expression in all patients. Of the two PBL patients, the DLBCL in the first patient is of germinal centre origin (CD10+) and in the second patient it is of activated B-cell origin (CD10−, BCL6+), (Figs 5–8).
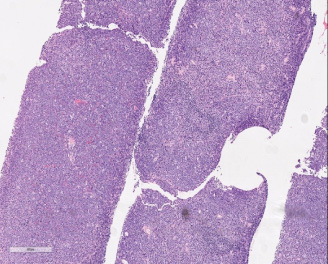
Breast core biopsy showing diffuse infiltrates of lymphoid cells, (100 H&E stain).
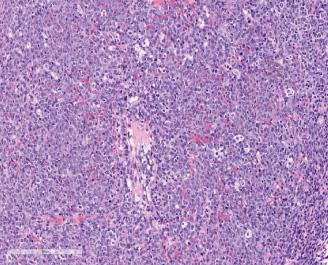
Breast core biopsy showing diffuse starry sky pattern with highly pleomorphic cells with vesicular nuclei and prominent nucleoli, (200 H&E stain).
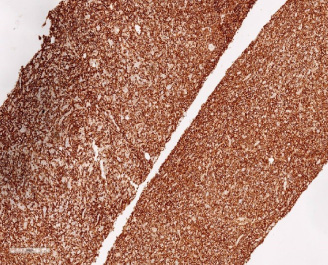
Immunohistochemical studies demonstrating diffuse and strong positivity for CD20.
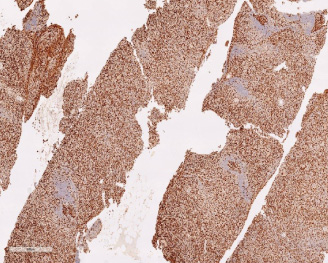
Immunohistochemical studies demonstrating High Ki67 proliferation index, approximately 95%.
All patients were referred to the haematology team for chemotherapy +∕− radiotherapy and further management with R-CHOP (Rituximab, Cyclophosphamide, Doxorubicin, Vincristine, Prednisolone) is considered the main regimen used in these cases.
Of our eleven patients, one patient passed away within one month, three patients died within one year of chemotherapy, five patients achieved complete remission, one patient had two relapse and is still under treatment, while the last patient was diagnosed recently and still awaiting treatment. Table 1 shows a summary of the presentations, diagnosis, management and outcome of our eleven cases.
Summary of the eleven cases of breast lymphoma
DLBCL: Diffuse Large B-cell Lymphomas, SCT: Stem Cell Transplant, R-CODOX-M/IVAC: (Cyclophosphamide, Doxorubicin, Vincristine, Methotrexate/Ifosfamide, Etoposide, high dose Cytarabine), R-CHOP: (Rituximab- Cyclophosphamide, Doxorubicin, Vincristine, Prednisolone), RCVP: (Rituximab, Cyclophosphamide, Vincristine Sulfate, Prednisone), IT: Intrathecal methotrexate, UOQ: upper outer quadrant.
Human breast involvement with lymphoma is relatively uncommon as the breast contains less lymphoid tissues when compared with other organs [10,11]. Breast lymphoma can be classified into primary (PBL) and secondary (SBL), based on the presence or absence of systemic involvement, respectively. Primary breast lymphoma is rare and accounts for about 1% of all non-Hodgkin’s lymphomas and 2% of extranodal lymphomas [12]. Weisman and Liao in 1972 described the following diagnostic criteria for PBL; (i) the breast is the principle site of presentation; (ii) no history of previous lymphoma or current evidence of widespread disease at the time of diagnosis; (iii) confirmed diagnosis of lymphoma in the breast tissues; (iv) ipsilateral lymph node may be involved at the same time of diagnosis of PBL [13].
Primary breast lymphoma usually presents as rapidly growing painless breast mass (es), with or without ipsilateral axillary lymph node involvement. The clinical presentation is usually similar to that of primary breast carcinomas [11,14]. The disease mainly (95–100%) affects females in the fifth to sixth decade of life [10,11,15]. The eleven patients included in our review were females, and the average age of diagnosis was 66.1 ± 13 years. It commonly affects the upper outer quadrant of the right breast [9,16]. Bilateral breast involvement was reported in 1% to 14% of the cases [17]. Both patients diagnosed with PBL in our study had left sided breast lymphoma, while from the nine patients with SBL; six patients had a right sided tumour and three had left sided lymphoma. None of our patients had bilateral breast lymphoma. The B symptoms associated with lymphoma, such as weight loss, fever and night sweats are rarely seen in patients with PBLs [10,18]. None of our patients showed any typical B symptoms at the time of diagnosis. It is difficult to differentiate between PBL and breast carcinoma clinically, and the usual manifestations of breast carcinomas are uncommon in PBL [14].
The imaging findings (mammogram and ultrasonography) of PBL are non-specific and mimic those of breast carcinoma (Figs 1–4). On the mammogram imaging, PBL usually appears as single, relatively well-circumscribed, uncalcified mass [10,14,15]. Calcification, architectural distortion, and nipple retraction are not routinely seen in case of PBL. On ultrasonography, PBL shows decreased internal echogenicity and increased posterior acoustic enhancement due to the high cell density. However, none of these manifestations are pathognomonic. MRI findings also are nonspecific [19]. CT/ PET-CT scans play an important role in tumour staging, measuring response to treatment and early detection of any recurrence [10]. All SBL patients in our review presented with stage IV disease, while the two PBL cases presented with stage IIE tumour with a limited disease to the breast and the ipsilateral axillary lymph nodes. Diagnosis is obtained only after histopathological assessment which may require further immunohistochemical analysis for further confirmation. Excisional biopsy and core biopsy are the most frequently used techniques for histopathology. Fine needle aspiration cytology (FNAC) is usually not recommended as it is difficult to obtain specific information like the subtype of lymphoma [12]. All patients involved in our review had core biopsies and the diagnosis was confirmed only after MDT discussion.
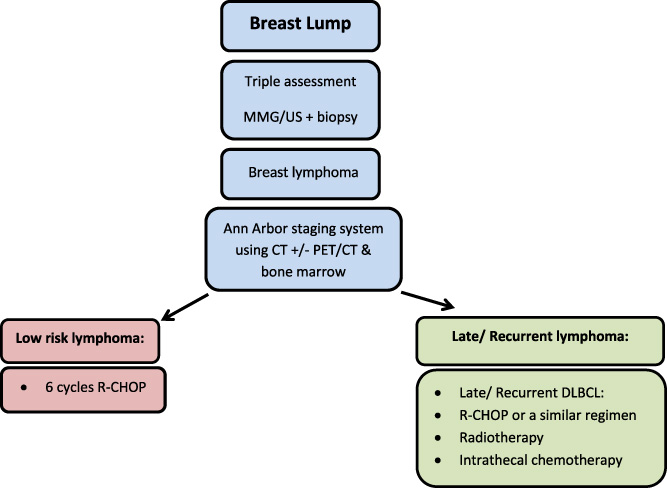
Summary of diagnosis and management tools.
The main histopathological subtypes of PBL are Non-Hodgkin’s DLBCL followed by follicular and MALT (mucosa- associated lymphoid tissue) lymphoma, with DLBCL represents 40% to 70% of all breast lymphomas [20]. Breast involvement with Hodgkin’s lymphoma or T-cell lymphoma is rare [21,22]. The prognosis of the disease depends mainly on the stage of the primary malignancy at the time of diagnosis [10,14]. Eight of the eleven patients included in our study had DLBCL, one patient had high-grade progression of lymphoplasmacytic lymphoma, and two had follicular lymphoma. The tumour cells in all our patients showed positivity for CD20 and BCL2, while CD10 and BCL6 were positive in most patients.
Favourable prognostic factors include early stage, low histological grade and a favourable international prognostic index (IPI) [15,23]. Generally, the prognosis of DLBCL is less favourable when compared with other subtypes.
The pathogenesis, management and prognosis for primary breast lymphoma are completely different from that of primary breast cancer. Unlike primary breast carcinoma, the treatment for breast lymphoma is mainly systemic. Radical surgery is not recommended and associated with higher mortality and poorer prognosis [24]. Although there is no standard treatment for primary breast lymphoma, the treatment with chemo-radiotherapy has been recommended [25].
In general, the treatment of PBLs follows the general guidelines for the management of lymphomas of the same stage in any other site of the body. R-CHOP was recommended as the first-line treatment regimen for PBL [24,26]. This may be followed with radiotherapy of the breast and regional lymph nodes [27]. Studies proved that combination therapy can improve the survival and reduce the local recurrence rates [28,29]. Patients with aggressive disease with high risk of recurrence should be considered for CNS prophylaxis [18,24,30]. Figure 9 summarizes the steps of diagnosis and management.
In our study, the two PBL patients were referred for chemotherapy; the first patient received R-CHOP regimen and achieved complete remission, and the second patient is still waiting for treatment. Four patients with SBL died within one year of treatment. Four patients achieved remission; one of them had previous DLBCL and was treated with CHOP with radiotherapy and stem cell transplant (SCT), the relapsed lymphoma in the breast was treated with four cycles of R-CODOX-M/IVAC (Cyclophosphamide, Doxorubicin, Vincristine, Methotrexate/Ifosfamide, Etoposide, and high dose Cytarabine) and achieved complete remission. The second patient had two relapses of follicular lymphoma despite initial treatment with RCVP (Rituximab, Cyclophosphamide, Vincristine Sulfate, and Prednisone); the first relapse was treated with R-CHOP and the second relapse was treated with radiotherapy and R-Bendamustine, the patient is still in remission. The third patient was treated with R-CHOP plus IT therapy, and the fourth one was treated with R-CHOP plus local radiotherapy. The last patient had two relapses of DLBCL and is waiting to commence on CHOP plus radiotherapy. Three patients had radiotherapy, all achieved complete remission. Three patients with aggressive disease had IT treatment; one achieved complete remission and two passed away.
Generally, this study included a series of breast lymphoma patients. We included eleven patients; two with primary breast lymphoma and nine with secondary breast lymphoma. We included a detailed summary of the presentation, diagnosis, management and outcome for every patient. However, we had some limitations in our review as the data were collected retrospectively and the majority of included patients had secondary breast lymphoma.
Primary breast lymphoma is an aggressive disease with an estimated 5-year overall survival rate of 50%–60% [31]. There are no specific criteria for the diagnosis of PBL, and the diagnosis is confirmed only after histopathological assessment. Accurate diagnosis of PBL is essential to avoid unnecessary surgery as the treatment is mainly systemic with chemo-radiotherapy being the standard regimen for the management of PBL.
Footnotes
Ethical approval
Not required as the study was retrospective with no breach to any patient’s confidentiality.
Conflicts of interest
Authors declare there are no conflicts of interest.
Funding
None.
