Abstract
In 2016, the World Health Organization added Breast Implant-Associated Anaplastic Large Cell lymphoma as a provisionally recognized lymphoma to the family of existing Anaplastic Large Cell lymphomas. Current estimates of the lifetime risk of the disease in women with textured breast implants range from 1:1,000 to 1:30,000. The mean interval from implant placement to diagnosis is 10.7 ± 4.6 years and the most common clinical symptom at presentation is breast swelling. A high level of clinical suspicion is recommended in patients presenting with breast symptoms and/or peri-implant fluid collection occurring more than 1 year after breast implant placement. Ultrasound is the imaging modality of choice, with a high sensitivity for peri-implant fluid and a high specificity for peri-implant mass. When ultrasound is inconclusive, breast MRI is indicated. As of today, all confirmed cases have tested positive for CD30 immunohistochemistry and the disease has shown to have an excellent prognosis when it is diagnosed earlier (localized disease), and when complete surgery, consisting of explantation, capsulectomy, and removal of any associated capsule mass, is performed. This overview summarizes the available epidemiological and clinical data of Breast Implant-Associated Anaplastic Large Cell lymphoma, with an emphasis on imaging features.
Introduction
In 1997, Keech & Creech published the first case report of anaplastic T-cell lymphoma in proximity to a saline-filled breast implant [1]. Following the initial published report, additional case reports and case series of this entity were published [2–4], and in 2016, the World Health Organization (WHO) added Breast Implant-Associated Anaplastic Large Cell lymphoma (BIA-ALCL) as a provisionally recognized lymphoma to the family of existing ALCL [5]. In 2019 the US Food and Drug Administration (FDA) issued a safety communication stating, “all individuals who are considering a breast implant of any type be informed of the risk of developing BIA-ALCL” [6]. The aim of this article is to present a brief overview of the epidemiological and clinical data of BIA-ALCL, with an emphasis on its imaging features.
Epidemiology
The current causal hypothesis for BIA-ALCL is based on the reported link of this disease to textured breast implants [7]. Shell texturing of breast implants was developed in the 1980s to reduce implant contractures. Texturing of the implant shell may lead to a greater inflammatory response of the surrounding fibrous tissue capsule eliciting an increased chronic antigenic stimulation, which in turn could potentially be responsible for the development of ALCL [8].
Current estimates of the lifetime risk of the disease in women with textured breast implants range from 1:1,000 to 1:30,000, with a reported geographic variation of the risk likely due to variable reporting and less likely to geographic or genetic predisposition [9–13].
The number of BIA-ALCL reported cases worldwide reached 573 in July 2019, with 320 cases in US [7]. At the time, the number of reported deaths reached 100, with 36 of them in the US [7].
A previous subset data analysis of cause of death showed direct extension into the chest wall, ultimately causing respiratory failure, as the most common cause of death [14]. Other less common causes of death were stem cell transplant complication and development of a second unrelated lymphoma [14]. Importantly, this subset analysis showed that none of the patients who died had received complete surgical excision or targeted therapy, and that there was delay in diagnosis or any treatment for an average of 1–2 years from onset of symptoms [14].
BIA-ALCL has shown to have an excellent prognosis when the disease is diagnosed earlier (localized disease), and when complete surgery, consisting of explantation, capsulectomy, and removal of any associated capsule mass, is performed [15].
Clinical presentation
The mean age at diagnosis is 53.2 ± 12.3 years and the mean interval from implant placement to diagnosis is 10.7 ± 4.6 years [16].
Type of breast implant surface, clinical presentation, and CD30 status at diagnosis of BIA-ALCL
Type of breast implant surface, clinical presentation, and CD30 status at diagnosis of BIA-ALCL
(a) History of prior textured implants. (b) List more than one clinical presentation, e.g., seroma and peri-implant mass/lump, in which two presentations were counted. N/A = Not Available. Source: US Food and Drug Administration. Medical Device Reports of Breast Implant-Associated Anaplastic Large Cell Lymphoma. https://www.fda.gov/medical-devices/breast-implants/medical-device-reports-breast-implant-associated-anaplastic-large-cell-lymphoma. Accessed February 18, 2020.
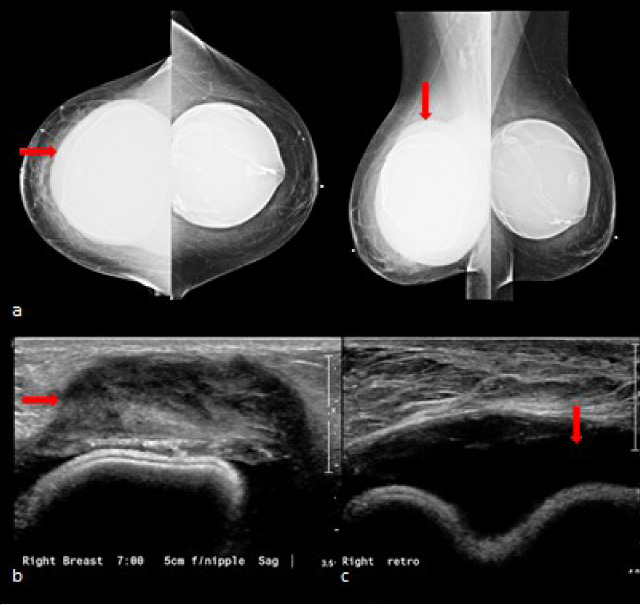
63-year-old female with history of saline breast implants with textured surface placed 19 years ago presented with pain and swelling throughout the right breast and a palpable abnormality in the posterior third of the right breast at 7 o’clock for 1 month. (a) Bilateral digital mammogram showed bilateral retro glandular saline breast implants. The right breast is larger than the left and shows a homogeneous and circumferential area of increased density (arrow) surrounding the implant, including the area of palpable abnormality noted by triangular marker in the posterior third of the right breast lower quadrants. (b) Grey scale ultrasound shows a 4 × 1.4 × 2.4 cm mixed echogenicity, oval, partially, indistinct mass (arrow) abutting the fibrous capsule in the right breast at 7 o’clock, 5 cm from the nipple, corresponding to palpable abnormality. (c) Grey scale ultrasound shows right breast peri-implant fluid collection (arrow). Breast MRI (d) axial T2-weighted fat-saturated sequence shows right breast peri-implant fluid collection (arrow), (e) axial T1-weighted fat saturated postcontrast subtraction shows a 4 × 1.7 × 2 cm oval heterogeneously enhancing mass (arrow) arising from the fibrous capsule in the right lower outer quadrant. (f) Screen capture of a whole-body 18F-FDG PET/CT shows FDG activity of SUV 10.56, corresponding to a soft tissue mass (arrow) in the lower outer quadrant of the right breast adjacent to the implant measuring 3.2 × 4.8 cm × 2.5 cm. (g) Screen capture of follow-up PET/CT obtained 6 months after bilateral breast explantation and total capsulectomy shows no abnormality.
TNM classification of BIA-ALCL
Source: BIA-ALCL, breast implant-associated anaplastic large cell lymphoma; NCCN, National Comprehensive Cancer Network.
Table 1 shows that the most common clinical symptom at presentation is breast swelling while peri-implant fluid collection (seroma) is the most common imaging finding [7]. A high level of clinical suspicion is recommended in patients presenting with breast symptoms and/or peri-implant fluid collection occurring more than 1 year after breast implant placement.
In general, mammography findings include nonspecific capsular thickening, circumferential asymmetry around the implant, or irregular mass change (without calcification) may be detected in some cases [17]. Overall, mammography has a lower sensitivity and specificity the both ultrasound and MRI for any abnormality due to BIA-ALCL, at 73% and 50% respectively [18].
Ultrasound is the imaging exam of choice and findings most commonly include a homogeneous peri-implant effusion with inflammatory changes in the periprosthetic breast tissue. In cases where a mass (or masses) is present it most commonly appears as an oval, hypoechoic, and well-circumscribed solid mass, without hypervascularity, although a complex-cystic mass has also been observed [17]. Abnormal ipsilateral axillary lymph nodes with corical thickening or diffusely hypoechoic without evident fatty hilus may be present in some cases. Ultrasound has the highest sensitivity and specificity for peri-implant fluid collection and solid mass at 84% and 100% respectively [18].
Breast MR is the second imaging test of choice after US and is particularly indicated when US yields indeterminate results. Findings include peri-implant tissue edema and effusion, as well as peri-implant mass lesions, including small-volume mass components not detected with US. MR also serves to evaluate for the presence of implant rupture when there is a silicone implant [17]. MRI is the imaging modality with the second highest sensitivity for peri-implant fluid collection and the second highest specificity for mass at 82% and 93% respectively [18].
Two case examples demonstrating the appearance of BIA-ALCL on mammography, ultrasound, and MRI, or shown in Fig. 1a–e and Fig. 2a–e.
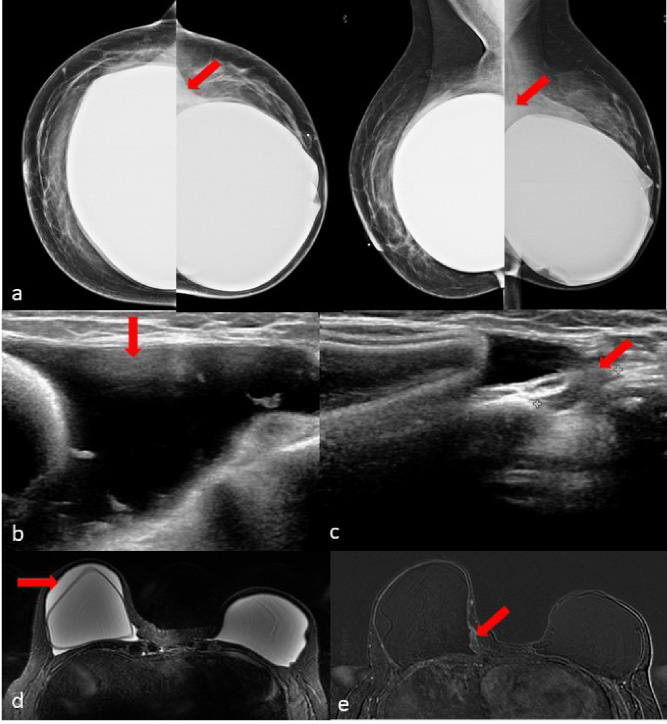
54-year-old female status post left breast mastectomy for DCIS and right breast prophylactic mastectomy, followed by immediate bilateral breast silicone implant with textured surface reconstruction 11 years ago presented with sudden onset of right breast swelling and enlargement with associated discomfort. She denied fever or general symptoms. (a) Bilateral mammogram shows irregular contours of the right breast silicone implant with associated focal-peri-implant increased density (arrows). (b) Grey scale ultrasound shows right breast peri-implant fluid collection (arrow) containing debris (arrow). (c) Grey scale ultrasound shows a 2.7 × 0.7 × 1 cm irregular mixed echogenicity, indistinct mass along the medial aspect of the right breast implant capsule at approximately 3 o’clock, in parasternal location. (d) MRI axial T2-weighted fat-saturated sequence shows right breast peri-implant fluid collection (arrow); (e) MRI axial T1-weighted fat saturated postcontrast subtraction shows a 2.3 cm in maximum extension focal asymmetric area of heterogeneous mild enhancement within the fibrous capsule in the posterior right breast lower inner quadrant.
In patients who present with peri-implant fluid collection a fine needle aspiration with a minimum of 30–50 ml fluid sample should be obtained for testing, while core needle biopsy is recommended if suspicious mass or abnormal lymph node are detected. Both procedures can be performed under ultrasound guidance.
As of today, all confirmed cases of BIA-ALCL have tested positive for CD30 immunohistochemistry (Table 1). Diagnostic evaluation should include cytological evaluation of fluid or mass with Wright Giemsa stained smears and cellblock immunohistochemistry/flow cytometry testing for cluster of differentiation (CD30) and Anaplastic Lymphoma Kinase (ALK) markers [6,7]. Figure 3 shows an example of the appearance of BIA-ACLC on hematoxylin and eosin and on immunocytochemistry for CD30.
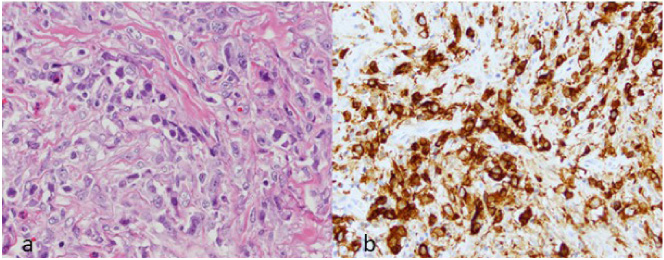
Photomicrographs of ultrasound-guided core needle biopsy samples of solid mass showed in Fig. 1b show (a), large and pleomorphic neoplastic cells with irregular nuclei, large prominent nucleoli, conspicuous mitotic activity, and moderate cytoplasm (hematoxylin and eosin [H&E], ×60), and (b) a majority of the cells to be strongly and uniformly positive for CD30 (CD30 immunohistochemistry, ×60).
Clonal T-cell receptor (TCR) gene rearrangements have been identified in nearly all BIA-ALCLs reported, which renders polymerase chain reaction (PCR) for gene rearrangement a valuable diagnostic tool. Mutations which have been reported include: STAT3, JAK1, JAK3, DNMT3A, and TP53. However, because both false-positive and false-negative results may be obtained, it is important to interpret the results of TCR PCR in the context of other clinical and pathologic features of BIA-ALCL [19].
Once the diagnosis of BIA- ALCL is established, patients should be referred to oncology, and a multidisciplinary evaluation by surgical oncology, plastic surgery, and radiation oncology is recommended (14). Filing a report of the case with an existing case registry is encouraged [20,21].
A lymphoma workup, including whole-body PET/CT (Image 1 g), should also be obtained [15]. TNM classification for BIA-ALCL is shown in Table 2.
The disease is considered extended (not localized) (Table 3) if there is tumor invasion beyond the fibrous capsule, spread to one or more regional lymph nodes, or spread to any organs/distant sites [22].
Currently, there is no data from prospective clinical trials on the treatment of BIA-ALC and treatment recommendations are based on case reports and on extrapolation from trials for peripheral ALCL [15].
The recommended surgical treatment for patients with localized disease consists of explantation, capsulectomy, and removal of any associated capsule mass. The goal is to achieve complete surgical excision, which is associated with excellent long-term and disease-free survival [10]. Local recurrence is most common following incomplete resection or partial capsulectomy [15].
Stage classification and extension of BIA-ALCL
Stage classification and extension of BIA-ALCL
Source: BIA-ALCL, breast implant-associated anaplastic large cell lymphoma; NCCN, National Comprehensive Cancer Network.
Systemic therapy with anthracycline-based chemotherapy and brentuximab vedotin combination is recommended for patients with disease that is not localized [15].
Local or involved site radiation therapy with 24 to 36 Gray (Gy) is recommended for patients with local residual disease, positive margins, or unresectable disease with chest wall invasion [15].
Surveillance of patients with complete response to treatment should include history and physical exam and either a CT with contrast of chest, abdomen, and pelvis or a whole-body PET-CT (Fig. 1h), every 6 months for 2 years, and then as clinically indicated [15].
BIA-ALCL has been linked to textured shell breast implants, independent of implant fill. Peri-implant fluid formation and/or breast symptoms occurring more than 1 year after placement of breast implant should raise suspicion of BIA-ALCL. While mammography may demonstrate subtle abnormalities, ultrasound and MRI have higher sensitivity and specificity. Diagnosis requires sampling of peri-implant fluid or mass/lymph node, the pathologist should be alerted of the suspicion of BIA-ALCL, and immunohistochemistry for CD30 should be ordered. Once BIA-ALCL is diagnosed, oncology referral and multi-specialty team care including plastic surgery and radiation therapy is recommended. With prompt diagnosis and complete treatment, the disease appears to have an excellent prognosis.
Key messages
New onset of peri-implant fluid formation and/or breast symptoms occurring more than 1 year after placement of breast implant should raise suspicion of BIA-ALCL. While mammography may demonstrate subtle abnormalities, ultrasound and MRI have higher sensitivity and specificity. Diagnosis requires sampling of peri-implant fluid or mass/lymph node, the pathologist should be alerted of the suspicion of BIA-ALCL, and immunohistochemistry for CD30 should be ordered.
