Abstract
Standard operative management for breast carcinoma has significantly shifted from extensive procedures to minor interventions.
Although axillary dissection was a fundamental component of operative management, sentinel biopsy is an actual process for axillary staging. Axillary dissection may be postponed for cases that have negative SLNs or 1 or 2 infiltrated lymph nodes undergoing breast or axillary radiation. Contrarily, axillary dissection is still the conventional management for patients with clinically positive nodes.
Arm lymphedema is a frequent and overwhelming complication of axillary dissection, with a worse impact on the patient’s life.
Axillary reverse mapping was recently introduced to map and conserve the lymph drain of the upper limb throughout axillary dissection or sentinel biopsy. A technique based on the theory that the breast’s lymphatic drainage differs from those that drain the arm, so preserving lymphatic drainage of the upper limb can prevent lymphedema, thereby not raising the risk of axillary recurrence.
Therefore, this technique is the reverse of sentinel biopsy, which remove the lymph nodes that drain the breast.
Introduction
Breast carcinoma is the most common carcinoma among women worldwide and the second most common cause of cancer-related deaths after lung cancer [1]. Despite the significant prevalence of breast cancer over the preceding few years, breast cancer fatality rates appear to be declining due to early identification and more effective treatment strategies [2].
The primary treatment strategy for breast cancer is surgical intervention. It started with radical mastectomy, which Halsted proposed as an extended radical procedure introduced by Marogottini and Urban [3], and consequently modified radical mastectomy introduced by Patey and Dyson [4]. In recent years, the surgical treatment of this malignancy has developed from more invasive surgery to less invasive, conservative breast surgery. Meanwhile, with a high early-stage breast cancer detection rate. 50% of breast cancer cases do not have an axillary spread, leading to more extensive treatment if all undergo axillary dissection (ALND). Recently, the concept of removing the breast and its lymph nodes extensively was changed in surgical treatment. When sentinel biopsy results are negative, the axillary dissection may be postponed [5].
Nevertheless, the subsequent arm lymphedema has the worst impact on patients’ quality of life who underwent axillary intervention, either with sentinel biopsy or axillary dissection. Axillary dissection was initially reported to lead to a 7–77% risk of lymphedema [6], while sentinel biopsy, which only removed a few axillary nodes, was also associated with a 3–13% risk [7]. Accordingly, an axillary mapping procedure is meant to prevent this complication since it can identify and preserve the lymphatic tissue that drains the arm throughout ALND and SLNB.
Pathophysiology of lymphedema
Disturbance in normal homeostasis, while the lymphatic system is adversely affected, leads to a collection of lymphatic fluid. T cell-mediated inflammatory response in the involved tissues accumulated in response to the aggregation of protein-rich fluid [8]. So the result is the formation of a pressure gradient that increases fluid accumulation. Additionally, it restricts their capability to recycle fluid caused by chronic inflammation [9]. Eventually, This process contributes to these vessels being irreversibly affected and not functioning.
Throughout an unexplained mechanism, adipocyte differentiation increases, leading to fibro-adipose changes of lymphedema: skin thickening, dermal elasticity loss, hyperkeratosis, hyperpigmentation, and elephantiasis [10,11]. Weight gain is an essential issue of lymphedema and is the cause in cases without further lymphatic impairment [12]. Lower limb lymphedema may be observed once a patient’s BMI exceeds 50 kg/m2, and it is the leading cause of lymphedema when a BMI is more than 60 [13].
Manifestations of lymphedema
Lymphedema is an overwhelming complication for patients experiencing cancer management. The rate of lymphedema will increase with any options of cancer treatments involving dissection of lymph nodes, radiotherapy, and chemotherapy [14]. Consequently, the most prevalent etiology of lymphedema is breast cancer therapy. Approximately 90% of patients will have the manifestations of lymphedema in the first 3y of therapy; also, some cases may experience lymphedema throughout the first 8 m of operation [15,16].
To date, no globally accepted guideline-concordant staging system classifies this disease according to its severity. The research studies the International Society of Lymphology (ISL) staging system is the commonly published staging system for lymphedema [17]. This method categorizes the grades of lymphedema from zero to three, varying from subclinical, mild, moderate, or severe. Therefore, the most immediate stage of lymphedema (stage zero) regarding ISL shows subjective manifestations without significant swelling. Also, clinical presentations may include heaviness, tightness, stiffness, aching, or weakness of the impaired limb.
Some data show that bioimpedance may help recognize minor differences in tissue components at this stage [18]. Accumulating the fluid causes early edema with an enlarged arm circumference as the condition advances. This swelling becomes reversible by the rise or squeezing of the limb at the early stages. Then, it will proceed to the irreversible stage revealing pitting edema followed by non-pitting edema, which occurs when the lymphatic tissues are flooded by permanent fluid stagnation. Skin manifestations will happen in mild to severe lymphedema due to excessive fibrin deposit and repeated cellulitis. Advanced lymphedema stages have multiple previously mentioned manifestations with noticeable skin signs. Additionally, symptoms include brown or black pigmentation, tissue hypertrophy, enlargement, and woody fibrosis of the affected area with papules and verrucous lesion formation, splitting, ulcer formation, and seldom Stewart Treves syndrome within the late stages [19]. Furthermore, with malformation and disabling manifestations, considerable swelling with boosted arm girth leads to reduced limb movement with a deficiency of function [20,21].
Axillary LN dissection & sentinel LN biopsy
Halsted proposed radical breast removal more than 100 years ago [22]. Axillary dissection was necessary throughout radical breast removal surgery, significantly improving survival rates [23–25]. Despite this, axillary dissection is associated with terrible morbidities, such as upper limb lymphedema, seroma formation, arm dysfunction, paresthesia, and discomfort [26]. The recorded frequency of lymphedema after arm lymph node dissection varied from 7 to 77% [27–29]. So, many researchers have studied this invasive surgery and raised their interest in the sentinel biopsy procedure [24]. This technique depends on the premise that the sentinel lymph nodes are the first locations primary cancer cells are expected to drain, thus acting as a filter for cancer cells [28]. Lymphatic drainage occurs in an orderly pattern through the subareolar plexus of Sappey to the same lymph node(s) in all breast quadrants. SLNB is performed using radioactive dye (Tc-99 sulfur colloid) and a visible blue dye (isosulfan blue or methylene blue) to detect proximal draining lymph nodes. These lymph nodes are then removed. So with SLNB, the lymphedema rate is 1–7% [30–33].
Donald L. Morton, Alistair J. Cochran, and their colleagues introduced the sentinel lymph node approach in 1992 to treat melanoma [34]. It was introduced into breast cancer treatment in 1993 after being proven useful in melanoma [35]. Consequently, several clinical experiments established that sentinel biopsy was a reliable and valid method to identify cancerous cells in axillary lymph nodes. Despite skipping unremoved affected nodes did not markedly raise the risk of a recurrence or breast-cancer-related fatality [5,27,36,37].
A sentinel biopsy was therefore considered the most appropriate treatment when there is no evidence of cancer in the lymph nodes of the axilla [38]. In comparison, axillary dissection maintains the most appropriate management for patients with axillary metastasis, which is detected on sentinel biopsy [39]. A study conducted by the IBCSG 23-01 concluded that patients with micrometastases should be treated with axillary dissection only if they had SLN involvement [40]. Consequently, the 2011 St-Gallen Consensus Conference recommended against interpreting micrometastases in only one SLN indicative of ALND regardless of treatment method [41].
According to the Z-0011 study, regional recurrence risk did not vary between women with early-stage (T1 or T2) breast cancer with a positive sentinel node who underwent conservative breast surgery with or without axillary dissection [42].
Accordingly, the redesigned 2014 American Society of Clinical Oncology guidelines encourage oncologists not to suggest ALND in breast cancer cases with one or two positive sentinel lymph nodes [43]. There is incomplete data to perform SLNB in females with end-stage breast cancer with large or locally invasive (T3-T4) inflammatory breast cancer. Females with sentinel LN metastases who will perform mastectomy should be doing axillary dissection if they do not have axillary radiation [44,45].
Axillary dissection in females with clinically node-positive axilla, axillary recurrence, or ineffective previous lymph node dissection and positive SLN in females who fall outside the Z0011 selection criteria ( >2 positive SLNs, matted nodes, mastectomy, or breast conservation without radiation therapy) is recommended [46].
Axillary reverse mapping
The ARM depends on how the arm’s lymphatic drainage (Fig. 1) differs from the lymphatics that drain the breast. As a result, ARM can reduce arm lymphedema by sensing the upper limb lymphatics during ALND or SLNB and keeping them intact [47]. This procedure can conserve the upper limb’s lymphatics and thus reduce lymphedema without missing lymph nodes harboring cancer cells [47].
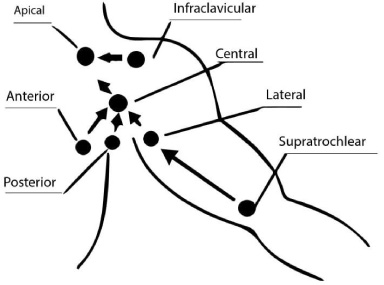
The arm lymphatic drainage.
In their initial study using hydrogen peroxide to visualize lymphatic arm vessels, Suami et al. noted that almost all of the blood flow from these lymphatic vessels led to one primary lymph node (sentry). The “sentry” node may be bypassed by some lymphatic vessels in the forearm, and passages along its posterior aspect skip it [48].
In 2007, two-spectral color fluorescence lymphangiography was a new method of identifying lymphatic drainage circuits emptying the breast and upper limb [49]. Then, in the corresponding year [50], Klimberg introduced ARM to the US [51].
Therefore, To distinguish ARM, three methods are used, i.e., methylene dye, radionuclide, and fluorescent dye.
Thompson and coworkers initially characterized the ARM procedure with blue dye in 2007. ARM lymphatics were recognized in 61% of patients [50]. Afterward, many researchers described the feasible practice with blue dye in ARM [52–54]. The lymphatic vessel and blue nodes were examined through the axillary fascia during ALND. An injection of 2–4 ml methylene blue dye was made subcutaneously 5 cm distal to the axilla within the uppermost inner ipsilateral limb. Give injection thirty minutes sometime recently ALND, taken after by a massage at the infusion area with an arm elevation for 5 minute. The patients who identified the bluish color lymph nodes were stripped and assigned autonomously for histopathology assessment. Consecutive sectioning and hematoxylin and eosin staining were evaluated in these lymph nodes to detect metastasis [55].
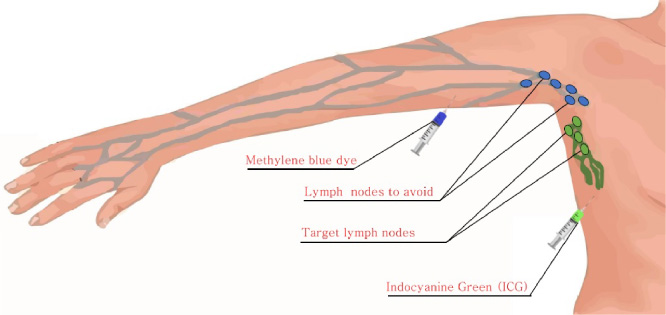
ARM using methylene blue dye + SLN mapped by ICG.
As the ARM nodes are mainly located lower than the SLN, it is difficult to identify them during the surgical procedure. As a result, isotope scans are needed to increase the probability of accurate detection and reduce the tattoos after the infusion of methylene dye.
Direct injection of the isotope can be performed in the webspace of the ipsilateral grip. After detecting the radioactive ARM nodes with a gamma probe, the detection ratios increased from 75 to 100%. Using different isotopes to detect ARM nodes via a scintillation counter, i.e., 111-in-human polyclonal immunoglobulin G [56]. In this sense, radioisotope appears to identify ARM nodes more significantly than methylene dye only. Despite enhancing node identification, radioisotopes do not allow the perception of the lymphatic vessels.
Thus, several methods are used to improve the detection of lymph nodes and lymphatic vessels [57,58]. Consequently, Tc99 sulfur colloid into the breast in 1.0 mCi injected is. The method is comparable to that for the SLNB and Isosulfan Blue injections. The patients received 2–5 ml injections into the upper ipsilateral limbs in the medial bicipital sulcus. As a result, the lymphatic channel fluid changes into a blue or blue-green color, making it possible to visualize directly during surgery.
Through split-mapping (radiotracer injected into the breast and blue dye injected into the upper extremity), can identify crossover lymph nodes (draining the breast and upper extremity). Afterward, SLNB or ALND is performed (depending on which technique is more appropriate), with special care to identify and spare the blue lymphatics [59–61].
Mapping by near-infrared fluorescent technique
An undetectable near-infrared fluorescent technique was used with 0.1 ml (0.25 mg) of indocyanine green (ICG) injection in the upper internal arm subdermally to identify the ARM lymph nodes. The lantern returned off within the working room; besides, bright fluorescent nodes and lymphatics were seen within the axillary area throughout ALND or SLN biopsy [37]. Via fluorescence procedure, the detection of ARM nodes may reach 32.0 to 89%.
Via fluorescent technique, there is a possibility to do Sentinel biopsy and mapping procedures in the same process and at the same time. Despite this, using isotope scans in ARM is somewhat sophisticated and time-needed, raising the detection rate [62]. Also, the value of that procedure is very sheep, and just several moments are needed to recognize the lymph system. Furthermore, identifying the paths is uncomplicated, and it can perform at any institution with any fluorescent imaging camera [63].
Anatomical location of ARM nodes
Many types of research have proven that axillary reverse mapping nodes are present beneath the axillary vein level [64,65]. Most lymph nodes that drain the ARM are located more profoundly than the SLN; therefore, the recognizable percentage of these lymph nodes signifies more importance during ALND than SLNB [54,66,67].
The axillary LN is divided into five anatomical locations, including:
Area A, between the 2nd intercostobrachial nerve and axillary vein. Area B, medially area A. Area C, below area A. Area D, medially to area C. Area E, the space over the axillary vein [68].
Approximately 62–98% of this LN are present in Area A, located in the uppermost external one-fourth of the axillary area just caudal to the axillary vein [67]. Many studies reported no lymphatics for the arm inferior to the 2nd intercostobrachial nerve [63].
SLN-ARM (crossover) lymph nodes
Depending on the anatomical information, the reverse mapping technique considered the lymphatics of the breast and lymphatics of the arm are drained in different axillary lymph nodes; the oncologic safety regarding that approach necessity be concerned [69]. Preserving mapping lymph nodes in sentinel biopsy will be more challenging if the sentinel biopsy that drains the breast is like the lymph node at which the reverse mapping node drains the arm. Studies reported that the lymph vessels arising in the inferior portion of the upper extremities had a reliable connection with the breast tissues through certain radiological studies of the lymph drainage of the upper limbs [70]. About one-fourth of cases might have this connection [71].
When using distinct pair techniques to detect SLN and ARM nodes simultaneously, the corresponding lymph node was seen by both approaches called SLN-ARM nodes. The occurrence of this condition indicates that the sentinel biopsy beside the upper limb node has the same lymphatic channels; hence physicians will consider SLN-ARM nodes with a great opportunity for metastatic affection. The SLN-ARM crossover occurred in roughly 4.2% of 348 cases regarding some novel research [72]. Another researcher noted that the SLN-ARM crossover risk varied from 0 to 28% [56,67,73]. Crossover nodes have a couple of various assumptions.
Primary, eliminating sentinel nodes may moderately disturb the lymph drain of the arm [48]; thus, it demonstrates how sentinel biopsy has an episode of lymphedema yet [62]. Second, if the sentinel nodes harbor spreading malignant cells, these cells may give spreading to crossover nodes. It will not be easy to conserve these lymph nodes. Different studies have revealed that cases with prominent complex nodes metastatic affection have a high chance of normal lymphatic flow and backflow alteration to the ARM nodes [74]. Almost metastatic ARM nodes exclusively happened in cases with SLN-ARM crossover, Deng and co-workers described [75]. Just 0–32% of crossover nodes are associated with malignancy [51,62,66,75,76], and the spreading risk for ARM nodes was related to the nodal (N) stage [72]. Cases with malignant affection of the ARM nodes most have pN3 stage, Noguchi et al. [66] and Nos et al. reporeted [65].
The cancer cells in the late stages are more prone to spread to nearby tissues, thus reducing the efficacy of ARM. Therefore, the N3 lymphatic stage is not an indication of ARM. It is crucial to identify the affected nodes in SLN-ARM nodes and eliminate them. Despite positive ARM nodes being further recognized in cases with less positive or negative nodes [64,68]. FNAC and biopsy of incomplete removal of SLN-ARM nodes may contribute vital data, in deciding which SLN-ARM node to remove and which one to persist.
Factors affecting the success of ARM
On the other hand, some factors do not affect the detection of axillary mapping nodes: patient age, mass extent, the appearance of extended lymph node spread, and concurrent sentinel biopsy [84–86].
The effect of lymphedema prevention after ARM
Breast cancer-related lymphedema (BCRL) is a common complex dilemma of breast cases encountered by axillary surgery. Studies predicted that 1 in 5 patients had lymphedema [87]. The existence of arm lymphedema following axillary dissection or sentinel biopsy is related to several agents, such as raised BMI > 25 kg/m2, adjuvant radiotherapy, infection or lymphangitis, and remaining lymphatic affection following preoperative chemotherapy [85,86,88].
The incidence of BCRL with ARM procedure significantly decreased. Yue et al. reported that the frequency of lymphedema in cases experiencing ARM ALND is 5.93%, which is less than that of patients without ARM 33.07% [89]. Abdelhameed et al. reported that the frequency of lymphedema using ARM is 6%, while it was 20% for the patients without ARM [90].
Many other studies confirmed similar findings without compromising the oncological outcome [91–94]. This differences regarding the incidence of lymphedema could be objectively measured, by physiatrist assessment, lymphoscintigraphy, and excess arm volume [95].
Complications of ARM
However, the ARM procedure is not complex; many complications are unrelated to the tumor after the operation. The most frequent problem is the dye staining at the infusion region; this staining might pass for some days or persist for many years [50,51]. Pain may also occur at the injection site is the other common ARM complication, and most patients suffer from mild pain [85]. Furthermore, systemic allergic or anaphylactic reactions may occur after dye injection [96].
Conclusion
ARM can be used with ALND or SLNB for breast cancer patients to minimize arm lymphedema. The technique is easy, applicable, and can offer improvement in the rates of upper limb lymphedema after axillary surgery, without compromising the oncological outcome.
Footnotes
12.
The authors declare no conflict of interest. No funding source.
