Abstract
INTRODUCTION:
The Objective was to investigate the incidence of lymphedema after breast cancer treatment and to analyze the risk factors involved in a tertiary level hospital.
METHODS:
Prospective longitudinal observational study over 3 years post-breast surgery. 232 patients undergoing surgery for breast cancer at our institution between September 2013 and February 2018. Sentinel lymph node biopsy (SLNB) or axillary lymphadenectomy (ALND) were mandatory in this cohort. In total, 201 patients met the inclusion criteria and had a median follow-up of 31 months (range, 1–54 months). Lymphedema was diagnosed by circumferential measurements and truncated cone calculations. Patients and tumor characteristics, shoulder range of motion limitation and local and systemic therapies were analyzed as possible risk factors for lymphedema.
RESULTS:
Most cases of lymphedema appeared in the first 2 years. 13.9% of patients developed lymphedema: 31% after ALND and 4.6% after SLNB (p < 0.01), and 46.7% after mastectomy and 11.3% after breast-conserving surgery (p < 0.01). The lymphedema rate increased when axillary radiotherapy (RT) was added to radical surgery: 4.3% for SLNB alone, 6.7% for SLNB + RT, 17.6% for ALND alone, and 35.2% for ALND + RT (p < 0.01). In the multivariate analysis, the only risk factors associated with the development of lymphedema were ALND and mastectomy, which had hazard ratios (95% confidence intervals) of 7.28 (2.92–18.16) and 3.9 (1.60–9.49) respectively.
CONCLUSIONS:
The main risk factors for lymphedema were the more radical surgeries (ALND and mastectomy). The risk associated with these procedures appeared to be worsened by the addition of axillary radiotherapy. A follow-up protocol in patients with ALND lasting at least two years, in which special attention is paid to these risk factors, is necessary to guarantee a comprehensive control of lymphedema that provides early detection and treatment.
Introduction
The development of lymphedema after breast cancer is a direct consequence of damage to the lymphatic drainage secondary to the surgery, although there are other factors that can contribute to its development, such as a tissue reaction after radiotherapy or an increased load on the lymphatic system due to tumor involvement or metastases in the lymphatic system [5]. Axillary lymphadenectomy (ALND) causes more damage to the lymphatic system than sentinel lymph node biopsy (SLNB), but both procedures are associated with lymphedema development, and it is not clear whether the number of lymph nodes removed may also play a role [6,7]. Other risk factors that can impact the lymphedema development reported in the literature are body mass index (BMI), the use of radiotherapy (RT) or chemotherapy with taxanes [1], although the results are controversial. A better understanding of these risk factors would aid the early detection of lymphedema and, therefore, improve the effectiveness of treatments and increase patients’ quality of life.
Since the standardization of SLNB in patients with breast cancer and no clinical lymph-node involvement (cN0) in the 1990s [8], the trend has been to decrease the number of ALND procedures in favor of SLNB, even in some node-positive cases [9]. The latest St. Gallen Consensus, including work by other authors [10], recommends avoiding ALND in women with 1 or 2 sentinel-positive lymph nodes after breast-conserving surgery when adjuvant RT and systemic therapy are given, irrespective of the tumor biology [11]. Due to the increase in SLNB surgeries and the fall in ALNDs, it is necessary to reassess the follow-up protocol for the prevention of lymphedema. Our hypothesis was that in addition to ALND, SLNB also increases the risk of lymphedema, especially when more than 3 lymph nodes are removed. It was also wanted to establish the optimal follow-up period for these patients, which should include the time in which most cases of lymphedema develop.
The aim of this study was to prospectively investigate the incidence of arm lymphedema in our rehabilitation unit to 3 years after breast cancer surgery, and to determine the risk factors for its appearance, including the impact of shoulder range of motion (ROM) limitation. Secondarily, this information may help to improve the early detection of lymphedema, as it was searched for a follow-up protocol that is more adjusted to identifying high-risk patients and to increasing the effectiveness of early therapies.
Methods
The institutional ethics committee of the Bellvitge University Hospital (PR004/13) approved the present study. All procedures were performed in accordance with the ethical standards of the institutional ethics committee and national research standards, as well as those of the 1964 Declaration of Helsinki and its later amendments, or comparable ethical standards. Prior to inclusion, patients taking part in the study signed a written informed consent form.
This was a prospective longitudinal observational cohort study, performed on 232 patients with breast cancer. All patients underwent surgery at the multidisciplinary breast cancer unit of the Bellvitge University Hospital-Catalan Institute of Oncology between September 2013 and February 2018. Axillary surgery, either SLNB or ALND, was mandatory. Patients were voluntarily enrolled after surgery and assessed at the Rehabilitation Department at 1, 6, 12, 18, 24, and 36 months to track the appearance and evolution of lymphedema. The inclusion criteria were as follows: age > 18 years; male or female; unilateral primary breast cancer with a pathological diagnosis of breast carcinoma or carcinoma in situ; axillary surgery (SLNB or ALND); and conservative breast surgery or mastectomy without reconstruction. Patients with stage IV breast cancer, no axillary surgery, mastectomy with immediate reconstruction, bilateral breast cancer, or a personal history of breast cancer or shoulder pathology were excluded.
Lymphedema was diagnosed by circumferential measurements of the perimeters of both upper limbs, using an inextensible tape at pre-established points in accordance with Brorson et al.’s description [12]. With the patient seated and the arm extended, the ulnar styloid, olecranon, and two points each on the forearm (7 cm and 14 cm distal to the olecranon) and middle arm (10 cm and 20 cm proximal to the olecranon) served as anatomical landmarks. The volumes of the affected and unaffected upper extremities were then obtained using perimeter measurements and the truncated cone formula [13]. The excess volume was recorded as the difference between the affected and unaffected limbs in milliliters. Lymphedema was considered present when the excess volume in the affected limb was at least 10% greater than that in the healthy limb [3,14], and classified into three categories by severity: mild (excess volume < 20%), moderate (20%–40%), and severe (>40%) [15]. To avoid the risk of confusing lymphedema with the postoperative swelling that can occur during the first postoperative weeks, it was performed the first assessment one month after surgery. To analyze potential risk factors, patients were grouped into those with and without lymphedema.
All patients with a ≥10% excess lymphedema volume received treatment at the Bellvitge University Hospital Rehabilitation Department. Treatment consisted of exercise therapy and skin care, decongestive lymphatic therapy, pressotherapy, and a multilayer bandage during the first phase, before moving on to compression garments in the maintenance phase, in accordance with the international consensus [13] and the best practice guidelines [16].
The risk factors for lymphedema most frequently described in the literature, the most relevant oncological variables and other collateral morbidities related to axillary surgery were selected for the study. Patient age, BMI (dividing the kilograms of weight by the square of the height in meters), histological type, surrogate molecular subtype, clinical stage at diagnosis (T and N), breast and axillary surgery type, number and level of lymph nodes dissected, adjuvant therapies, and ROM of the shoulder joint limitation were assessed. Both the BMI and the shoulder range of motion limitation were evaluated at each follow-up after surgery. Immunohistological staining of the tumor samples by receptor status was classified into five surrogate subtypes according to St Gallen 2019 [11]; Luminal A-like, Luminal B-like, Luminal B Her2-positive, Her2-positive, and Triple Negative. ROM of the shoulder joint limitation were assessed in the movements of flexion, internal rotation and external rotation with a goniometer, placing one arm of the goniometer fixed and the other arm of the goniometer open as the patient performed the movement of the shoulder, and compared with the mobility of the contralateral arm. A difference of 10° was chosen as the cut-off point for diagnosing ROM limitation, as in previous studies [17].
The objective measurements were carried out by two rehabilitation doctors with more than 5 and 10 years of experience respectively. Measurements were performed twice and averaged for greater accuracy.
Surgery was planned for 3–5 weeks after a breast cancer diagnosis or the last neoadjuvant chemotherapy course. Breast-conserving surgery was offered if optimal margins were guaranteed with a good aesthetic result. SLNB was only performed in clinically N0 cases, while ALND was performed in cases with >2 positive sentinel lymph nodes (according to the ACOSOGZ0011 and AMAROS criteria [10,18]) or N-positive (N+) disease at diagnosis.
Neoadjuvant chemotherapy included anthracyclines followed by taxanes for 6 months, with anti-HER2 therapy added as appropriate. Lymphadenectomy was performed when sentinel lymph node was positive after neoadjuvant chemotherapy and for all N+ tumors. All patients who underwent breast-conserving surgery received radiotherapy. A boost to the tumor bed was administered by brachytherapy or external-beam RT in patients younger than 50 years, or in those at high risk of locoregional recurrence [10,11]. RT to the chest wall after mastectomy was administered according to local guidelines for N+ cases, those with affected surgical margins, or those with large tumors (≥T3). Nodal RT was given if >3 lymph nodes were affected. Risk factors were considered to determine the need for nodal irradiation when 1–3 lymph nodes were involved.
Analysis
Sample size was estimated taking into account that the median lymphedema risk in the literature was 14% after ALND and 2% after SLND. Using the ARCSINUS approximation and accepting an alpha risk of 0.05 and a beta risk of 0.2 in a two-sided test, 65 subjects were necessary in the first group and 130 in the second to find a statistically significant difference, expected to be of 0.14 in group 1 and 0.025 in group 2. A drop-out rate of 10% was anticipated.
Categorical variables are presented as numbers and percentages of cases, while continuous variables are presented as means and standard deviations, if normally distributed, and as medians and ranges when not. Differences between groups were analyzed by chi-square or Fisher exact tests for categorical variables and by Student t-tests for continuous variables. Axillary surgery combined with axillary radiotherapy and breast surgery was also analyzed to better assess the impact of this association. A Cox regression multivariate analysis was performed to determine the impact of each risk factor on the development of lymphedema, and hazard ratios (HRs) with 95% confidence intervals (95%CIs) were calculated. For the multivariate analysis, all variables that were significant in the univariate analysis were included. Statistical significance was arbitrarily set at 5% and statistical analyses were performed using IBM SPSS Version 23.0 (IBM Corp., Armonk, NY, USA).
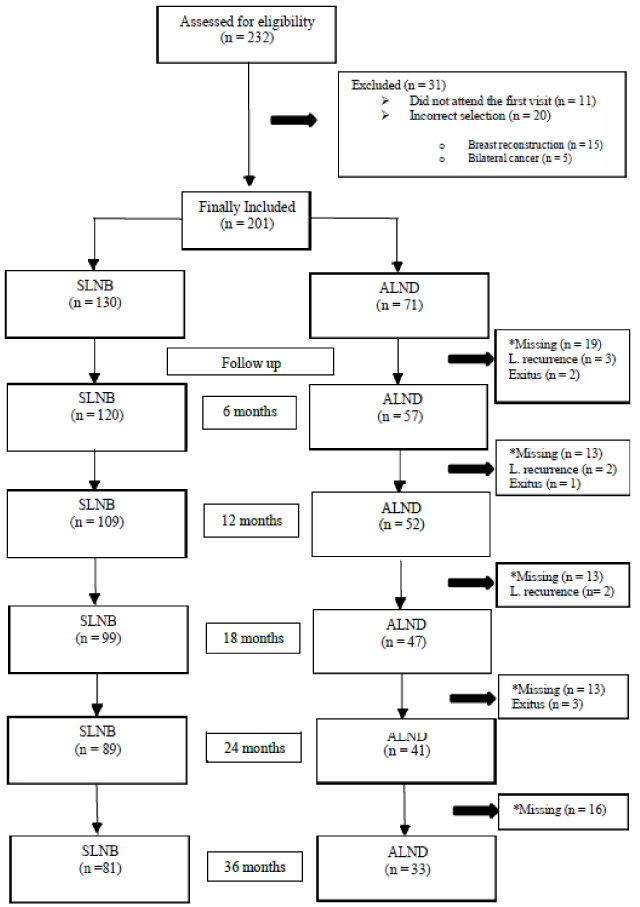
Flow chart of study participation. Abbreviations: SLNB = sentinel lymph node biopsy; ALND = axillary lymph node dissection; L = Local. *The missing patients continued follow-up by the Oncology Service, although they did not come for follow-up by the Rehabilitation Service.
Patient characteristics (n = 201)
Abbreviations: SD = standard deviation; ALND = axillary lymph node dissection; BMI = body mass index; CT = chemotherapy; DCI = ductal carcinoma invasive; Is = in situ; LCI = lobular carcinoma invasive; N = node involvement; RT = radiotherapy; SD = standard deviation; SLNB = sentinel lymph node biopsy; T = tumor size.
Informed consent was provided by 232 patients with breast cancer from September 2013 to February 2018, but only 201 (71 ALND and 130 SLNB) were included in the final sample (Fig. 1). The patient and tumor characteristics in the overall sample are shown in Table 1.
After a median follow-up of 35 months (range 1–54 months), 28 of the 201 patients developed upper limb lymphedema: 22 after ALND (31%) and 6 after SLNB (4.6%). Thus, 13.9% of patients in our sample developed lymphedema during the first 3 years of follow-up (Fig. 2), with most (24 of 28) doing so within the first 2 years. This corresponded to 86% of lymphedema cases starting during the first 2 years after surgical treatment of breast cancer. Lymphedema severity was mild in all cases after SLNB, but after ALND, it was mild in 18 cases (81.9%) and moderate in 4 cases (18.1%). All cases became stable over time; lymphedema was considered stable when it remained within its severity categories: mild, moderate or severe, thanks to the early application of therapy.
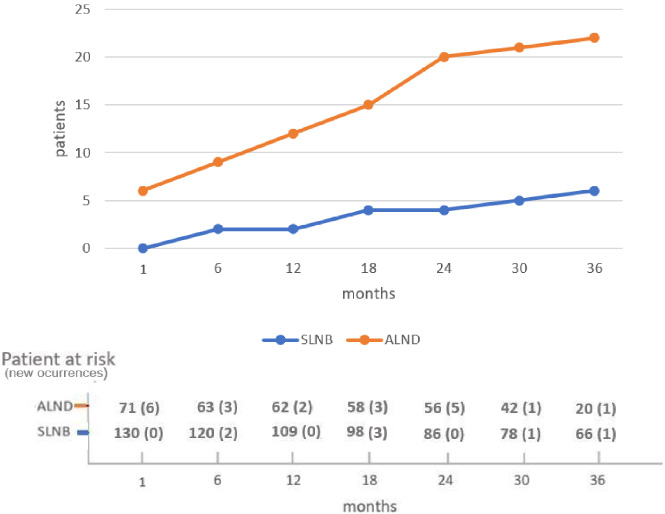
Lymphedema cumulative incidence by axillary surgery type. Abbreviations: SLNB = sentinel lymph node biopsy; ALND = axillary lymph node dissection.
The results of the univariate analyses of the risk factors for lymphedema are summarized in Table 2.
Mastectomy significantly increased the risk of developing lymphedema compared with breast- conserving surgery (HR 5.68; 95% CI, 2.40–13.39). However, 7 out of 15 mastectomies (46.7%) were performed for locally advanced tumors (T3 or T4) compared with just 12 out of 186 breast-conserving surgeries (6.5%). Mastectomies were performed in a significantly higher percentage of advanced tumors than conservative surgeries (p < 0.0001).
Regarding the type of axillary surgery, ALND also significantly increased the risk of lymphedema compared to SLNB (HR 8.55; 95%CI, 3.41–21.47). We observed more lymphedema cases when ALND included level III nodes rather than only level I and II nodes, though the differences were not statistically significant. Removing high numbers of lymph nodes (>3) in SLNB was not correlated with an increased risk of lymphedema.
RT and chemotherapy both affected the risk of lymphedema. Patients who received axillary RT to levels I–III and supraclavicular nodes presented a greater risk of developing lymphedema (HR 5.8; 95%CI, 2.55–13.27) than those who did not receive RT. The use of neoadjuvant chemotherapy was also associated with a slightly higher risk of developing lymphedema, independently of whether this included taxanes. Regarding adjuvant chemotherapy, no significant differences were found between groups, although it should be noted that all patients with lymphedema who were treated in the adjuvant setting received taxanes.
When it was analyzed the increased risk of suffering from lymphedema by grouping axillary surgical technique and RT (Table 3), the risk increased progressively with the aggressiveness of axillary treatment. Compared with patients who underwent SLNB alone, patients who underwent SLNB plus axillary RT, ALND without axillary RT, and ALND plus axillary RT had 1.5-times, 4.3-times, and 10.9-times higher risks of suffering from lymphedema respectively. Most SLNB cases received no adjuvant axillary radiotherapy (115/120), while most ALND cases received adjuvant radiotherapy (54/71), which increased the risk of lymphedema in this group.
Results of univariate analyses of risk for lymphedema
Abbreviations: ALND = axillary lymph node dissection; CT = chemotherapy; HR = hazard ratio; Is = in situ; N = node involvement; NA = not applicable; ROM = range shoulder of motion, RT = radiotherapy; SLNB = sentinel lymph node biopsy; T = tumor size; Tis = tumor in situ.
Lymphedema risk based on combined interaction factors
Abbreviations: ALND = axillary lymph node dissection; BC = breast conserving; RT = radiotherapy; SLNB = sentinel lymph node biopsy.
Limited shoulder ROM, another comorbidity related to breast cancer treatment, appeared principally during the first month after surgery and was associated with a significantly increased risk of lymphedema (HR 2.3; 95%CI, 1.00–5.26).
The risk factors significantly associated with the development of lymphedema in the multivariate analysis were mastectomy and ALND. These represented the most aggressive surgical options in both the breast and axilla. Overall, patients undergoing lymphadenectomy had a 7-fold greater risk of developing lymphedema than those undergoing SLNB, especially if they also underwent mastectomy (Table 4).
Results of multivariate analyses of risk factors for lymphedema
Abbreviations: ALND = axillary lymph node dissection; SLNB = sentinel lymph node biopsy; NA = Not applicable.
In this prospective study, the cumulative incidence of lymphedema secondary to breast cancer was 13.8% after a median follow-up of 35 months. ALND was the most important risk factor for lymphedema development (a rate of 31%, compared with 4.6% following SLNB; p < 0.01), especially when mastectomy was performed concurrently (70% following mastectomy plus ALND). Our hypothesis with regard to the associations between lymphedema and ALND and SLNB was confirmed, but the number of lymph nodes removed does not seem to influence its lymphedema development.
The strength of this study lies in its confirmation that SLNB increases the risk of lymphedema, although it does not depend on the number of lymph nodes removed. The results emphasize the idea that mastectomy associated with lymphadenectomy also has a greater impact on the appearance of lymphedema and that the rest of the risk factors described in the literature have less impact. In addition, its applicability when creating a follow-up protocol for the prevention and early diagnosis of lymphedema will improve the quality of life of patients with breast cancer.
The incidence of lymphedema in the prospective studies reported in the meta-analysis of DiSipio et al. [19] varies from 8.4% to 21.4%. Our cumulative incidence of 13.9% is inside this range. This variability may be influenced by the different definitions of lymphedema and by the methods used for measurement. Most diagnoses are made when there is a difference of ≥2 cm at any point of the circumference around the upper limb compared to the contralateral one, but this is not an absolute value. In our study, it is used the definition that lymphedema is present when there is an excess volume of >10%, which shows a good correlation with symptoms and is the absolute value referenced most often in a prominent meta-analysis [19]. According to the results of this study, most cases of lymphedema appeared during the first 2 years of follow-up; beyond this time the risk was reduced but did not disappear. Again, these results are consistent with those reported by other authors [3,5,20,21]. Most cases of lymphedema appeared in the first 2 years after axillary surgery, and so this is the period that should be included in the follow-up protocols.
Regarding lymphedema severity, it was mild in all patients with SLNB and in most patients with ALND (excess volume of <20%), and moderate in four patients after ALND (excess volume 20%–40%). These results were maintained between the measurement at the first visit and the last visit after 3 years, and are similar to those reported by other authors [20,22–24]. Probably the early detection and treatment, thanks to the better protocolized follow-up of these patients, has had a positive influence on the results.
Limited shoulder ROM appeared mainly during the first postoperative month, as reported in previous studies [3,25–27]. In our opinion, it appears during the first month because this is a period during which the patient experiences post-surgery pain. If the arm is kept immobilized, it is understandable that patients experience limited shoulder mobility when they start to use it after 2–3 weeks. However, the relationship between shoulder ROM limitations and lymphedema has not been analyzed previously. It is observed an increase in lymphedema in this cohort with shoulder ROM limitations, probably because the muscle activity that normally helps to drain the lymph vessels through its pumping action during normal use was reduced. Although the small number of cases in our multivariate analysis was insufficient to confirm this hypothesis, in the univariate analysis it is observed a significant increase in lymphedema when ROM was limited. This is consistent with advice to move the arm after surgery so as to avoid the appearance of lymphedema, and also with the recommendations in the literature [28].
In the multivariate analysis, the type of breast and type of axillary surgery had the greatest influence on the development of lymphedema. Our results show that ALND was associated with an increased risk of lymphedema, confirming the results of previous studies [19,29–32]. Interestingly, and contrary to the belief of many surgeons and some rehabilitation physicians, we did not find significant differences in the risk of developing lymphedema according to the number of SLNBs. To date, very few studies have sought to establish the risk of developing lymphedema by the number of lymph nodes removed during SLNB, but those that have been published coincide with our results [20,33–36]. There is a fine balance between preventing lymphedema and causing undue worry, and our results do not support a change the status quo. Thus, lymphedema prevention follow-up should be maintained for patients undergoing ALND, but this need not be extended to patients undergoing SLNB. Mastectomy was also associated with a higher risk of lymphedema in these analyses, albeit only when ALND was also performed. This may be because mastectomy allows for more radical lymphadenectomy compared with conservative surgery or because it is chosen over breast- conserving surgery for more advanced or larger tumors. Several other authors have also reported this association [1,4,19,37–39].
An association between axillary RT and lymphedema has been described in the literature [40–42]. In a systematic review of eight studies, Lee et al. [3] reported a combined odds ratio of 1.46 for the development of lymphedema in patients treated with RT. In a meta-analysis, DiSipio et al. also confirmed axillary RT to be a risk factor [19]. It is founded a higher risk of lymphedema for patients with axillary RT in the univariate analysis, but not in the multivariate analysis. The added risk of axillary surgery could have affected this result, and although axillary RT may increase the risk of developing lymphedema, the fundamental risk factor is the type of axillary surgery. In our study, we did not have enough cases with ALND and without RT to be able to analyze their effects independently, and so were obliged to rely on the analysis of axillary surgery plus RT together, which showed that the risk of lymphedema increased progressively with the aggressiveness of axillary surgery and the addition of axillary RT. Naoum et al. presented similar results [43].
RT to the breast did not significantly alter the risk of lymphedema in the current study. Previous studies have also shown that breast irradiation does not increase the risk of upper limb lymphedema [19,44,45]. However, as most patients in our population (197 of 201) received adjuvant breast RT, this variable lacks statistical power for independent analysis.
Neoadjuvant chemotherapy, with or without taxanes, increased the risk of developing lymphedema in the univariate analysis but not in the multivariate analysis. In the literature, chemotherapy with taxanes has been associated with the development of lymphedema due to their anti-lymphangiogenic effect; this inhibits the process of lymphatic regeneration that normally prevents lymphedema, and thus increases the volume of extracellular fluid [2,46,47].
Although no significant differences were found between groups according to the use of adjuvant chemotherapy, all patients who developed lymphedema received adjuvant chemotherapy that included taxanes. Findings on this topic in the literature vary widely, with some studies reporting no relationship with the development of lymphedema [1,19,24,42,48,49]. It may be that lymphedema develops only with more advanced tumors.
BMI was not a risk factor for lymphedema in the present study, but a clear association with a worsening of lymphedema has already been established both in the literature and in clinical practice. Although other studies have not shown a significant relationship between BMI and the risk of lymphedema [35], most, including a meta-analysis [19,40,41,44], conclude that obesity is a risk factor for its development; it is believed that lymphatic flow is impaired in obesity because the muscle pump is less active [19].
Study limitations
Few mastectomies were included in the current study. This is probably because we did not include patients with immediate reconstruction who typically have more advanced tumors. We decided to exclude these patients because breast reconstruction is associated with an increased risk of shoulder pathology and comorbidities that might bias the effect of axillary surgery, which was the focus of our study.
Second, it has not measured a preoperative ROM of the shoulder. However, all patients started the study with complete joint balance and shoulder without prior limitation.
Finally, the loss of patients over time and the small size of the subgroups precluded the evaluation of taxanes and RT. However, the present study also has several strengths. It was a prospective, single-center study with a large sample size (n = 201) which used reliable and valid methods to assess lymphedema. Moreover, it had a long follow-up time of 35 months; given that the peak incidence of arm lymphedema occurs within 24 months of surgery, we believe that our follow-up period was sufficiently long.
More prospective studies are now needed, to determine the possible increase in the appearance of lymphedema after ALND and other associated morbidities. Furthermore, the impact of adjuvant chemotherapy and axillary radiotherapy should also be better evaluated, by increasing the number of patients analyzed in these subgroups.
Conclusions
The prevalence of lymphedema during the first 3 years after breast cancer treatment was 31% when ALND was performed, but only 4.6% when SLNB was performed instead. ALND was the most important risk factor for the development of lymphedema, especially when performed concurrently with mastectomy. Surprisingly, however, the number of sentinel nodes removed was not a risk factor. We recommend close monitoring in these cases during the first 2 years, the period in which most cases of lymphedema will arise. This approach should allow the early detection and treatment of lymphedema, ultimately reducing its severity and its impact on patients’ quality of life. In addition, all breast cancer patients undergoing any type of axillary surgery should learn how to detect early symptoms of lymphedema in order to consult their doctor promptly and if necessary to be referred to a lymphedema unit.
Footnotes
List of abbreviations
ALND: axillary lymph node dissection; SLNB: sentinel lymph node biopsy; RT: radiotherapy; SD: standard deviation; CT: chemotherapy; ROM: range of motion.
