Abstract
BACKGROUND:
Breast cancer is a common women's disease. Usually, oestrogen is blamed in the aetiology and correlated with the prognosis; however, androgens are recently raising concern about its role in the breast cancer treatment and prognosis.
METHODS:
In this study we retrieved archival paraffin blocks of breast cancer patients and stained it for androgen. Thereafter, we compared clinico-epidemiologic parameters, histopathology, neoadjuvant response and recurrence rate and pattern among patients with and without androgen receptor (AR) expression.
RESULTS:
In total, 119 patients fulfilled enrolment criteria; AR expression were present in 77.3% of the patients. AR expression was associated with less grade III (6.8% versus 36.4%), and less triple negative (6.2% versus 25%), but similar overall recurrence rate (25% versus 22.2%). However, distant recurrence was significantly higher in androgen positive patients (91.3% versus 33.3% of all recurrences).
CONCLUSION:
Androgen expression appears to be common among breast cancer, but with no clear implication in tumour aggressiveness or effect on the rate of recurrence. However, being commonly associated with distant spread may have an impact on survival of the patients.
Background
Breast cancer is a highly heterogeneous disease. The current clinical classification of breast cancer is based on the expression of oestrogen receptor (ER), progesterone receptor (PR), human epidermal growth factor 2 (HER2) and the proliferative index Ki67, thus resulting in broad division into 4 molecular subtype groups, Luminal A, Luminal B, Her2-enriched and triple negative breast cancer. ER and HER2 serve as prognostic markers, guide to molecular subtyping and the more important a direct therapeutic strategies [1].
AR receptor status has been explored in literature aiming at aiding in prognostication [2] and addition of a potential new therapeutic target. Targeting the androgen receptors had result in a confusing data as some implemented testosterone (androgen agonist therapies) [3,4] and other authors implemented flutamide and anti-androgen/selective androgen receptor modulators extrapolated from prostate cancer protocols [5].
Furthermore, the worst prognostic group of breast cancer—those with triple negative disease—was subdivided in accordance of Lehman classification into 7 categories, one of them is the luminal androgen receptor [6]. Other clinical classification divided triple negative cancer into quadruple negative (ER, PR, HER2neu & AR negative) and triple negative androgen positive type with better prognosis [7].
The signalling effect of AR is likely to be different across breast cancer subtypes, and particularly important is its interaction with ER signalling [8]. In a Norwegian study, AR was an independent predictor of good prognosis, particularly in grade 3 and luminal A tumours. Discordant AR-expression between primary tumour and lymph node metastasis (LNM) was observed in 21.4% of the cases and most often there was a switch from AR−ve primary tumour to AR+ve axillary LNM [2].
This study aims at studying the rate and pattern of recurrence among patients with breast cancer in accordance with their androgen receptor status as a primary outcome. In addition, secondary outcome includes the distribution of AR expression among different molecular subtypes and its correlation with primary disease aggressiveness and the response to neoadjuvant therapy. To our knowledge, this is the first study to explore the incidence and prognostic value of androgen receptor status in Egyptian women.
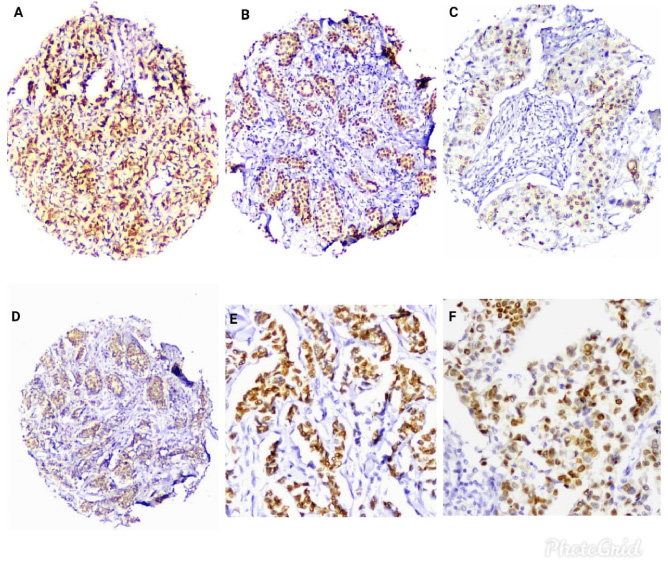
A, B, C and D: Androgen receptors immunohistochemical expression in different cases (H&E, 200×). E and F: Higher power views show the nuclear positivity for AR (H&E, 400×).
This is a retrospective study on the archival material of specimens from females with breast cancer, attending a tertiary cancer hospital within the period from May 2017 till May 2020.
Inclusion criteria: Pathologic proven breast cancer. Patients receiving curative surgical treatment.
Exclusion criteria: Patients with metastatic disease on presentation.
The patients’ paraffin blocks of the primary tumour were retrieved from the pathology archive. Tissue microarray was constructed manually as previously published [9]. Briefly, a representative slide for each tumour was selected and an area of the tumour was circled. Using the manual tissue microarrayer (MTA-1) produced by Beecher Instruments Inc., Sun Prairie, WI, USA, the selected areas of interest of the donor block were cored with a 0.6-mm diameter cylinder and transferred to a recipient paraffin block. Three cores were taken from each tumour.
Immunohistochemistry (Fig. 1): Tissue sections from the microarray were cut at thickness of 4 μm. After that, they were stained with antibodies against androgen receptors (AR) (Rabbit, monoclonal, Cell Marque, USA) according to the instructions of the manufacturers. The positive control for AR in this study will be human prostatic tissue.
Evaluation of the staining: Slides were examined by two independent pathologists blinded to patient characteristics and outcomes. For AR, the average of AR expression across the three cores was manually assessed. A cut-off of 1% nuclear expression or greater was used to define AR positivity. For assessment of the intensity and percentage of expression, H-score was estimated for each case. It was determined by adding the results of multiplication of the percentage of cells with staining intensity ordinal value (scored from 0 for “no signal” to 3 for “strong signal”) with 300 possible values. Expression of ER, PR, HER2 and Ki67 was routinely previously evaluated on the whole sections by immunohistochemistry (IHC) and by fluorescent in situ hybridization for equivocal HER2 score.
Response to neoadjuvant therapy: In patients who received neoadjuvant therapy, the response was reported using RECIST 1.1.
The epidemiologic, pathologic, treatment and follow up data of those patients were collected. Patients after completing treatment were followed up as per the hospital policy by history and physical examination biannually and sonomammography annually for 5 years unless there is newly developed symptoms and signs of recurrence that warrant further investigations. Recurrence and mortality were recorded.
The data of these patients were analysed using SPSS version 22 (Inc, Chicago, IL). Continuous variables are presented as mean when symmetrical or median and range when asymmetrical. Categorical variables are presented as proportions. P-value <0.05 was considered significant.
Results
In this study, 119 patients were enrolled. The mean age was 54.4 + ∕− 11.6 years old. 92 patients (77.3%) were AR positive. The majority of the patients (104) had infiltrating duct carcinoma (IDC). Neoadjuvant therapy was used for only 15 (12.6%) patients. Most of the enrolled patients (115, 96.6%) were treated by mastectomy. 99 (83.2%) patients were node positive. 79 (66.4%) patients were luminal, 14 (11.8%) were HER2neu enriched and 11 (9.2%) were triple negative. The patients were followed-up for a median of 46 months. Recurrence occurred in 29 (24.4%) patients, being distant in about 2/3 (65.5%) of them (Table 1).
Clinico-epidemiologic parameters of the studied patients
Clinico-epidemiologic parameters of the studied patients
∗Some missing data. #Valid percent.
Dividing the patients into group I androgen receptor over-expression (AR+ve) and group II androgen receptor non-expression (AR−ve) patients, both groups were comparable in age (p-value = 0.67), type of surgical treatment to both the breast (p-value = 1) and the axilla (p-value = 0.32), and in the rate of receiving neoadjuvant therapy (p-value = 0.33) (Table 2).
Comparison of androgen receptor positive and negative patients according to age and treatment received
Regarding the primary outcomes, the rate of recurrence (22.2% versus 25%, p-value = 1) and the time span to recurrence (median 18.2 versus 26 months, p-value = 0.56) were comparable in AR−ve and AR+ve groups; however, the pattern of recurrence was different with a tendency to distant recurrence in AR+ve patients (33.3% versus 91.3% of all recurrences in each group, p-value = 0.008) (Table 3). The overall (OAS) and disease-free survival (DFS) were again comparable between both groups (p-value = 0.13 and 0.93, respectively) (Fig. 2).
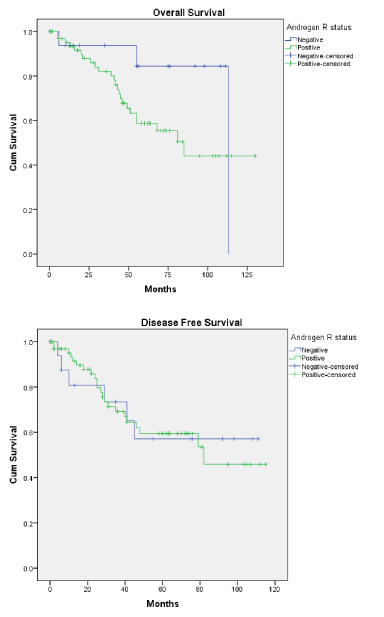
Kaplan Meier curve showing overall and disease-free survival among androgen receptor positive and negative patients.
Comparison of androgen receptor positive and negative patients according to the outcomes
Looking at the molecular types, although luminal type was the most common in both groups, there was a significant difference in molecular pattern among the groups where AR+ve patient tend to have more luminal (78.7% versus 66.7%) and Her2enriched (15% versus 8.3%), and less triple negative (6.2% versus 25%) cancers (Table 3).
As for aggressiveness, they are comparable in stage (p-value = 0.8), LVI (p-value = 0.8) and PNI (p-value = 1), but AR−ve tumours tend to be more grade III (36.4% versus 6.8%, p-value = <0.001). Finally, the response to neoadjuvant treatment was the similar (p-value = 0.58) (Table 3).
Subgroup analysis according to the molecular type, still shows similar recurrence rate in AR negative/positive patients of luminal (p-value = 0.503), HER2neu-enriched (p-value = 0.505) and triple negative (p-value = 1) cancers.
Also, androgen score distribution was similar among the recurrence groups (p-value = 0.098).
Androgen receptor is expressed in approximately 80 and 60% of primary and metastatic breast tumours, respectively [8]. Androgen receptor was positive in 77.3% of our random study population, coping with numbers from previous literature.
Immunohistochemical studies have demonstrated that AR expression differs between the various subtypes, with a higher positivity in luminal A and a lower positivity in triple negative breast cancers [10,11]. This is again similar to our study were there is a significant difference in distribution among AR groups with less triple negative patients expressing androgen receptor.
In comparison to previous study by Hwang et al. [12] and to Vera-Badillo et al. [13] earlier metanalysis, most of our recruited patients were node positive (about 83%) and more than half of patients were locally advanced (about 60% stage III). In addition, only 12.5% of this study population received neoadjuvant therapy, a low percentage that can be explained by the fact that more than three quarters of them had a luminal breast cancer, a factor that probably result in a higher tendency of the multidisciplinary team (MDT) to refer for upfront surgery.
The expression of AR in early breast cancer was associated with better overall and disease free survival irrespective of co-expression of oestrogen receptor (ER) in a metanalysis in 2014 [13], this was later confirmed in a large cohort regarding overall survival, but with a tendency to better disease free survival only in luminal HER2 negative breast cancers [12]. However, in another study, AR expression was neither associated with prognosis, nor altering the treatment effect with letrozole versus tamoxifen. As such, Kensler et al. suggested that AR expression may not be an informative biomarker for the selection of adjuvant endocrine therapy for postmenopausal women with ER+ breast cancers [14].
In the current study, the main endpoint was the recurrence rate, which was comparable in patients with androgen receptor over-expression and those non-expressing, among all patients and among different molecular types analysed separately. Also, the overall and disease-free survival were equivalent among both groups of patients. However, we surprisingly found a higher tendency of distant distribution of the recurrence among those with AR over-expression that may signify a more aggressive pattern.
Also, higher rate of AR expression was observed in grade I/II versus grade III tumours, was associated with older age >50, and was found in node negative versus node positive disease; while, no association was seen with menopausal status, clinical stage, or lymphovascular invasion (LVI) [15]. Similarly, our data showed tendency for lower grade in AR+ve patients, but no age or nodal status difference were evident. In addition, our results coincide with Vidula et al. finding that there is no implication of androgen expression on stage or LVI.
In a Turkish study, AR positivity was associated with poor response to neoadjuvant chemotherapy (NACT) in breast cancer patients [16]. In contrast, another study showed that higher AR expression was correlated to good clinical response to NACT [17]. In contrast to both aforementioned studies, there was no significant correlation between response to neoadjuvant therapy and androgen receptor expression in our study.
Finally, we need to highlight that the retrospective nature together with the relatively small number of patients represent a limitation for the current study. However, as a strength point to the study, involved patients had in the majority locally advanced and node positive breast cancer with minimal interference in outcomes by neoadjuvant therapy. In addition, to ameliorate the prognostic effect of different molecular types we reanalysed outcomes in each type separately. Moreover, we studied the effect of androgen receptor score not only the negative/positive expression on recurrence.
Conclusions
Androgen receptor expression may be present in about three quarters of the breast cancer patients. Androgen receptor over-expressing tumours tend to be of lower grade and less commonly triple negative. Androgen expression does not affect the response to neoadjuvant therapy. Finally, androgen expression does not appear to affect the recurrence rate, overall or disease-free survival, however; the recurrences in androgen positive patients tend to present as a metastatic disease.
List of abbreviations
AR: androgen receptor, ER: oestrogen receptor, PR: progesterone receptor, HER2neu: human epidermal growth factor 2. LNM: lymph node metastasis, IHC: immunohistochemistry, RECIST: response evaluation criteria in solid tumours, IDC: infiltrating duct carcinoma, LVI: lymphovascular invasion, NACT: neoadjuvant chemotherapy.
Ethical approval and consent to participate
All procedures performed in the study involving human participants were in accordance with the ethical standards of the institutional research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. The study protocol was approved by the institutional review board at faculty of medicine, Mansoura University with code number R.20.05.848.
Consent for publication
This was study on archival material and no identifiable patient data included.
Availability of data and materials
All the clinical, radiological & pathological data used in this manuscript is available on Mansoura University database medical system (Ibn Sina Hospital management system). http://srv137.mans.edu.eg/mus/newSystem/
Competing interests
None to declare.
Funding
No funding was received.
Authors’ contributions
SE: conceptualization, data curation, investigation, methodology. AH: conceptualization, investigation, supervision. AE: investigation, data curation. IHM: Data analysis, writing draft, validation. AMS, MJ, and ES: data collection, methodology, revision. MA: conceptualization, methodology, project administration. All authors have read and approved the manuscript.
Footnotes
Acknowledgements
None.
