Abstract
BACKGROUND:
Current guidelines define primary and secondary endocrine resistance according to the periods of adjuvant endocrine therapy (adj-ET); however, the relationship between adj-ET period and endocrine resistance remains unclear.
OBJECTIVE:
We examined progression-free survival (PFS) after primary ET for recurrent hormone receptor-positive/HER2-negative breast cancer, and evaluated the relationship between endocrine resistance and the periods of adj-ET.
METHODS:
We assessed PFS among 183 patients who received ET as primary treatment for the first recurrence, according to the period of adj-ET (adj-ET < 1 year, 1–2 years, ≥2 years, and completion).
RESULTS:
Patients who relapsed during the first year of adj-ET had the significantly shortest PFS. PFS did not significantly differ between patients who relapsed at 1–2 years of adj-ET and patients who relapsed while on adj-ET but after the first 2 years.
CONCLUSIONS:
Relapse at 1–2 years after adj-ET initiation might be better classified as secondary endocrine resistance rather than primary endocrine resistance.
Keywords
Introduction
Breast cancer is the most frequently diagnosed cancer in women and a major cause of cancer-related mortality worldwide [1,2]. Approximately 70–80% of breast cancer patients have hormone receptor-positive (HR+) tumors, and are considered candidates for endocrine therapy (ET), which significantly improves survival [3–5]. However, although the majority of HR+ patients initially respond to ET, 15–20% exhibit intrinsic resistance to ET, and an additional 30–40% will acquire ET resistance over years of treatment [6,7]. Endocrine resistance inevitably results in relapse and metastasis, thus leading to a poor prognosis, and is a major clinical problem regarding the treatment of HR+ breast cancer. Endocrine resistance can occur at disease inception (primary or intrinsic resistance) or develop during treatment (secondary or acquired resistance) [8,9]. According to ESMO guidelines [10], endocrine resistance is clinically classified in terms of the period of adjuvant ET (adj-ET)—with primary endocrine resistance defined as relapse during the first 2 years of adj-ET, and secondary endocrine resistance as relapse while on adj-ET but after the first 2 years. The majority of expert opinions support the guidelines, and the period of adj-ET may be associated with endocrine resistance development. However, there is little evidence regarding the length of adj-ET needed to determine whether endocrine resistance is primary or secondary.
In the present retrospective study, we aimed to examine progression-free survival (PFS) among patients who received primary ET for recurrent HR+/HER2-negative (HER2-) cancer, and to evaluate the possible relationship between endocrine resistance and the period of adj-ET. Our results may be valuable for identifying optimal treatment options in cases of recurrent HR+/HER2- breast cancer.
Methods
Study design and patients
This study was conducted using a retrospective longitudinal cohort design with the use of hospital electronic patient records. Patients with a first diagnosis of recurrent HR+/HER2- breast cancer made between January 2000 and December 2015 were identified from Sakai City Medical Center and Kindai University Hospital. Of these patients, we analyzed 183 patients who received ET as primary treatment for the first distant recurrence that occurred after primary breast cancer removal and standard adjuvant treatment initiation. According to current guidelines, all patients received standard adjuvant treatment and were followed up with through a regular physical examination 1–4 times a year and annual mammography; if necessary, blood exams, ultrasonography, computed tomography (CT), bone scintigraphy, magnetic resonance imaging, or position emission tomography/CT were added for the diagnosis of recurrence. Recurrence was defined as the occurrence of distant metastasis after removal of the primary breast cancer. Patients with only locoregional recurrence and distant metastasis at initial diagnosis (de novo Stage IV metastatic disease) were excluded from this analysis. Ipsilateral breast tumor recurrence and ipsilateral axillary, infraclavicular, internal mammary, and supraclavicular lymph node metastasis were defined as locoregional recurrence. TNM staging was based on the criteria of the 8th Union for International Cancer Control. The adjuvant and metastatic treatment strategies (treatment protocol after recurrence) were all decided at the experts’ conference in the institutions based on current guidelines. This study was approved by the institutional review board of the two hospitals, and all enrolled patients provided informed consent.
Subtype classification
Positivity for estrogen receptor (ER) or progesterone receptor (PR) was defined as a score ≥3 using the Allred scoring system [11]. HR-positivity was defined as ER and/or PR-positivity. HER2-negativity was defined as an immunohistochemistry score of 0, 1+, or 2+ and negative fluorescence in situ hybridization (ratio < 2.0).
All patient characteristics
ER: Estrogen receptor.
All patient characteristics
ER: Estrogen receptor.
Recurrent patients were divided into four categories according to their period of adj-ET: relapse during the first year of adj-ET (adj-ET < 1 y), relapse at 1–2 years after adj-ET initiation (adj-ET : 1–2 y), relapse while on adj-ET but after the first 2 years (adj-ET ≥ 2 y), and relapse more than one year after adj-ET completion (adj-ET:NR; no resistance).
Patients’ characteristics according to the periods of adjuvant endocrine therapy
ET: Endocrine therapy; NR: no resistance; PFS: progression-free survival; OS: overall survival; ER: estrogen receptor.
Patients’ characteristics according to the periods of adjuvant endocrine therapy
ET: Endocrine therapy; NR: no resistance; PFS: progression-free survival; OS: overall survival; ER: estrogen receptor.
Among the enrolled patients, we assessed PFS for the first recurrence, overall survival (OS), and chemotherapy-free duration (CFD) according to the periods of adj-ET. PFS was defined as the time from the date of ET initiation for the first distant recurrence to disease progression. OS was defined as the time from the date of the first distant recurrence to death or last follow-up. CFD was defined as the time from the date of the first distant recurrence to the first initiation of chemotherapy.
Statistical analysis
PFS, OS, and CFD plots were calculated using the Kaplan–Meier method. Survival curve distributions were compared using the log-rank test. The Cox proportional hazard regression model was used to examine survival according to the period of adj-ET. A 95% confidence interval was calculated for all hazard ratios (HRs) in the Cox regression analysis. HRs > 1.0 indicated an increased risk of disease progression and chemotherapy initiation. All tests were two-tailed, and
Results
Patients characteristics
Our analysis included 183 patients with HR+/HER2-recurrent metastatic breast cancer (Table 1). Table 2 shows patients’ characteristics according to the periods of adj-ET. The adj-ET < 1 y group exhibited a greater frequency of low ER expression compared to any other group.
Survival outcomes
Table 2 presents the median OS, PFS, and CFD after recurrence according to the periods of adj-ET. Compared to the other groups, the adj-ET < 1 y group exhibited the shortest median OS after recurrence (log-rank test
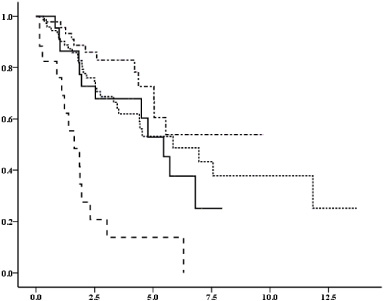
Overall survival (years) according to the periods of adjuvant endocrine therapy (adj-ET). Vertical axis: probability of survival (%), horizontal axis: years. The graph legend: − ⋅ − ⋅ − adj-ET : NR,. . . . . . . adj-ET ≥ 2 y,
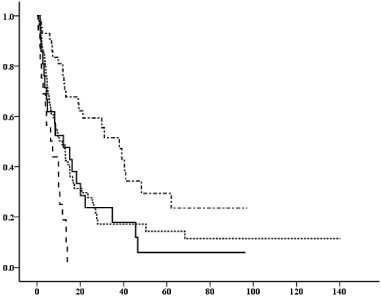
Progression-free survival (months) of primary endocrine therapy for first recurrence according to the periods of adjuvant endocrine therapy (adj-ET). Vertical axis: probability of survival (%), horizontal axis: months. The graph legend: − ⋅ − ⋅ − adj-ET : NR,. . . . . . . adj-ET ≥ 2 y,
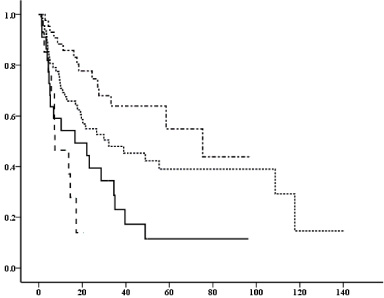
Chemotherapy-free duration after recurrence (months) according to the periods of adjuvant endocrine therapy (adj-ET). Vertical axis: probability of survival (%), horizontal axis: months. The graph legend: − ⋅ − ⋅ − adj-ET : NR, . . . . . . . adj-ET ≥ 2 y,
Progression-free survival of primary endocrine therapy (ET) for first recurrence and chemotherapy-free duration after recurrence according to the periods of adjuvant endocrine ET
Adj-ET: Adjuvant endocrine therapy; PFS: progression-free survival; HR: hazard ratio; CI: confidence interval; CFD: chemotherapy-free duration.
Breast cancer is a heterogeneous disease that includes several biologic subtypes with different clinical behaviors and treatment responses [5,12]. The most common type is HR+/HER2- breast cancer, belonging to the luminal subtype [3–5]. One major clinical problem faced in the treatment of HR+/HER2- cancer is endocrine resistance, which reduces the therapeutic effect of anti-estrogen agents [6–9]. ESMO guidelines clinically define primary and secondary endocrine resistance based on the period of adj-ET [10], but scarce data are available to support the selected cut-off length of adj-ET for determining primary versus secondary endocrine resistance. In the present retrospective study, we identified a significant relationship between endocrine resistance and the periods of adj-ET among cases of recurrent HR+/HER2- breast cancer. Our results suggested that relapse at 1–2 years after adj-ET initiation is not equivalent to primary endocrine resistance, but may rather be regarded as secondary endocrine resistance. This is an important distinction since these patients with secondary resistance might not have endocrine resistance strong enough to require a cytotoxic agent as compared to patients with primary resistance. Our present findings contradict the current ESMO guidelines, suggesting a need to reconsider the current clinical criterion.
The literatures include hypotheses regarding multiple mechanisms of endocrine resistance, such as low or loss of ER expression, disrupted tamoxifen metabolism, converted microRNA expression, HER2 upregulation, genetic mutations, and activation of various intracellular pathways involved in signal transduction pathways [13–24]. Analysis of these mechanisms may lead to significant improvements in treating patients with endocrine resistance; however, we do not yet fully understand the molecular mechanisms underlying primary and secondary ET resistance. To our knowledge, this is the first clinical evaluation of the relationship between endocrine resistance and periods of adj-ET in cases of HR+/HER2- breast cancer. Primary endocrine resistance is intense enough to prevent responses to both first-line ET and subsequent courses of ET [8,9], such that patients who relapse during the first year of adj-ET might require primary treatment other than endocrine monotherapy. Our present results suggested that patients who relapse at 1–2 years after adj-ET initiation may not have endocrine resistance strong enough to require primary treatment with a cytotoxic agent. The ability to identify the intrinsic presence of primary endocrine resistance through various means, such as based on clinical courses, could help us to select more advantageous treatment strategies for these patients.
Our study had some limitations. First, our present study is a retrospective chart review without validation, and thus it was impossible to avoid a selection bias. Second, the sample size of our study was small, and the results should be interpreted with caution. However, the selection of patients with HR+/HER2- recurrent breast cancer who received ET as primary treatment for their first recurrence and exclusion of HER2+, triple-negative, and de novo breast cancer patients may have allowed the recruitment of a patient population with relative homogeneity, regardless of the small sample size. Despite these limitations, the present study with a uniform population enabled us to focus on the relationship between endocrine resistance and the periods of adj-ET, and reveal important clinical significance based on real-world data from actual clinical practice. Future prospective studies with larger and independent cohorts could provide more conclusive evidence regarding the relationship between endocrine resistance and adj-ET, and potentially validate our present findings.
Conclusion
Our present results suggest that relapse at 1–2 years after adj-ET initiation might be unlikely to represent primary endocrine resistance, but should rather be considered secondary endocrine resistance. This distinction is important because these patients with secondary resistance might not require cytotoxic agents as a primary treatment. Thus, our present study provides an important clue to guide the development of new approaches to improve the treatment of patients with endocrine resistance.
Footnotes
Acknowledgements
The authors thank Dr. Tetsuya Mitsudomi, MD, PhD, Prof, Division of Thoracic Surgery, Department of Surgery, Kindai University Faculty of Medicine, Japan, for his excellent advice and support.
Conflict of interest
The authors declare no conflict of interest.
Funding sources
This study was supported by no funding source.
Author contributions
Yamamura J and Komoike Y conceived of the presented idea and design, and the analysis and interpretation of data for the work. All authors discussed the results and contributed to the final manuscript.
